 Back
BackCell Cycle, Apoptosis, and Cancer: Key Concepts in Cell Biology
Study Guide - Smart Notes

Cell Cycle: Structure and Regulation
Overview of the Cell Cycle
The cell cycle is a series of phases that cells undergo to grow and divide. It consists of interphase (G1, S, G2) and mitosis (M phase), each with distinct molecular events. - G1 phase: Cell growth and preparation for DNA replication. - S phase: DNA synthesis and replication. - G2 phase: Preparation for mitosis. - M phase: Mitosis and cytokinesis, leading to cell division. 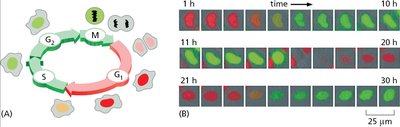
Cell Cycle Analysis by DNA Content
Flow cytometry can be used to analyze cell cycle phases based on DNA content. - G1 phase: Cells have a single set of DNA. - S phase: Cells are actively replicating DNA. - G2/M phase: Cells have double the DNA content. 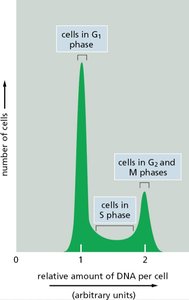
Cell Cycle Control: Cyclins and Cdks
Cyclin-Dependent Kinases (Cdks) and Cyclins
Cyclin-dependent kinases (Cdks) are key regulators of the cell cycle. They require binding to cyclins for activation. - Cdk: Enzyme that phosphorylates target proteins to drive cell cycle progression. - Cyclin: Regulatory protein whose levels fluctuate during the cell cycle. 
Sequential Activation of Cdks
Different cyclin-Cdk complexes are activated at specific cell cycle stages, ensuring orderly progression. - G1/S-Cdk: Initiates DNA replication. - S-Cdk: Drives DNA synthesis. - M-Cdk: Triggers mitosis. 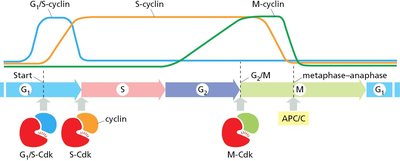
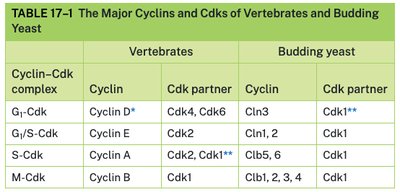
Major Cyclins and Cdks in Vertebrates and Yeast
The table below compares the main cyclin-Cdk complexes in vertebrates and budding yeast.
Cyclin–Cdk complex | Vertebrates: Cyclin | Vertebrates: Cdk partner | Budding yeast: Cyclin | Budding yeast: Cdk partner |
|---|---|---|---|---|
G1-Cdk | Cyclin D* | Cdk4, Cdk6 | Cln3 | Cdk1** |
G1/S-Cdk | Cyclin E | Cdk2 | Cln1, 2 | Cdk1 |
S-Cdk | Cyclin A | Cdk2, Cdk1** | Clb5, 6 | Cdk1 |
M-Cdk | Cyclin B | Cdk1 | Clb1, 2, 3, 4 | Cdk1 |
Activation of Cdks
Cdk activation is a multi-step process involving cyclin binding and phosphorylation. - Inactive: Cdk alone, T-loop blocks active site. - Partly active: Cyclin binding exposes active site. - Fully active: Cdk-activating kinase (CAK) phosphorylates Cdk, fully activating it. 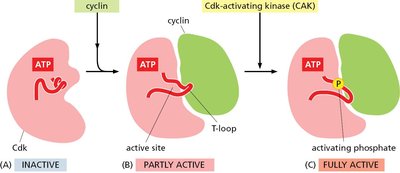
Inactivation of Cdks
Cdks can be inactivated by inhibitory phosphorylation (Wee1 kinase) and reactivated by dephosphorylation (Cdc25 phosphatase). 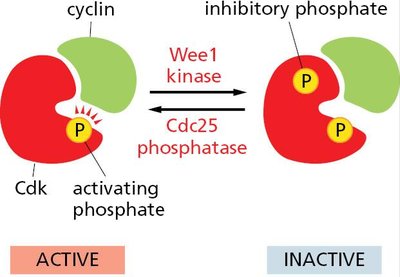
Cdk Inhibition by p27
The protein p27 binds to cyclin-Cdk complexes, inhibiting their activity and halting cell cycle progression. 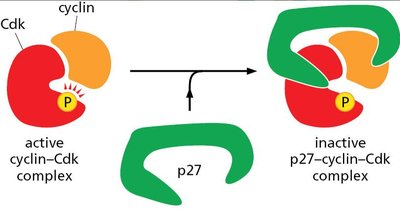
Cell Cycle Checkpoints
Cell cycle checkpoints ensure proper progression and prevent errors. - G1 checkpoint: Monitors extracellular environment and DNA integrity. - G2 checkpoint: Prevents entry into mitosis if DNA is damaged or unreplicated. - M checkpoint: Ensures chromosomes are properly attached to spindle before anaphase. 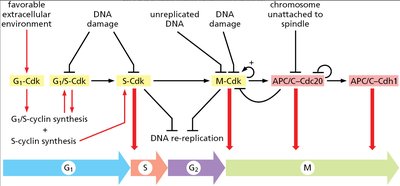
Chromosome Cohesion and Condensation
Cohesin Proteins
Cohesins are protein complexes that hold sister chromatids together after DNA replication, ensuring accurate chromosome segregation. 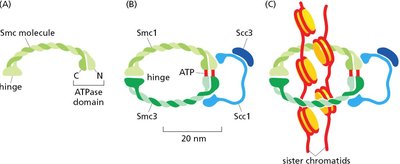
Condensin Proteins
Condensins help condense chromosomes during mitosis, facilitating their segregation. 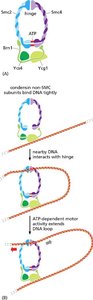
Mitosis: Key Events
Prophase
During prophase, chromosomes condense, the nuclear envelope remains intact, and the mitotic spindle begins to form. 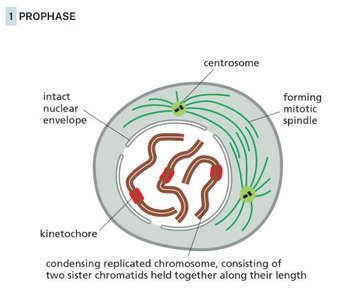
Metaphase
Chromosomes align at the metaphase plate, attached to spindle microtubules via kinetochores. 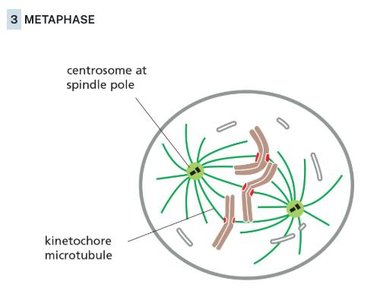
Cytokinesis
Cytokinesis is the final step of cell division, where the cytoplasm splits, forming two daughter cells. 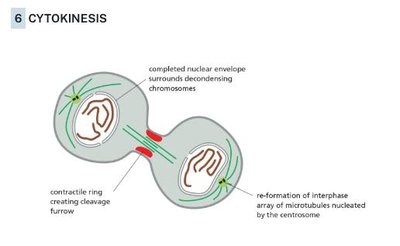
Centrosome Cycle
The centrosome duplicates and matures, organizing microtubules for mitosis. 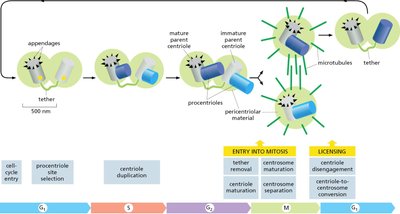
Kinetochore Attachment
Proper attachment of kinetochores to spindle poles is essential for chromosome segregation. 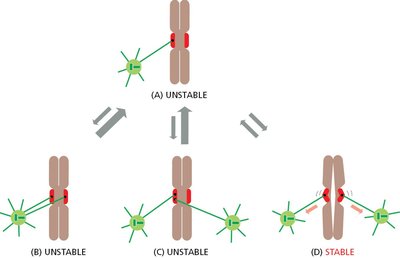
Sister Chromatid Separation
The anaphase-promoting complex (APC/C) triggers the degradation of securin, activating separase and allowing cohesins to be cleaved, leading to sister chromatid separation. 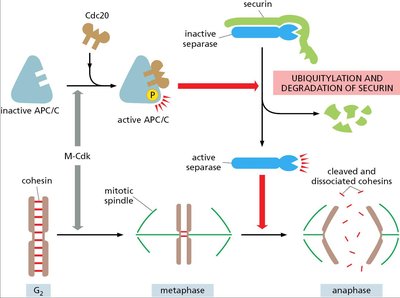
Cell Cycle Arrest and DNA Damage Response
DNA Damage-Induced Cell Cycle Arrest
Cells can halt the cell cycle in response to DNA damage, preventing propagation of mutations. - Checkpoints: G1, S, and G2 phases are monitored for DNA integrity.
Cell Growth Stimulation by Extracellular Factors
Growth factors and extracellular signals can stimulate cell cycle entry and progression.
Apoptosis: Programmed Cell Death
Apoptosis vs. Necrosis
Apoptosis is a regulated, energy-dependent process of cell death, distinct from necrosis, which is uncontrolled and often results from injury. - Apoptosis: Cell shrinkage, DNA fragmentation, membrane blebbing. - Necrosis: Cell swelling, membrane rupture, inflammation. 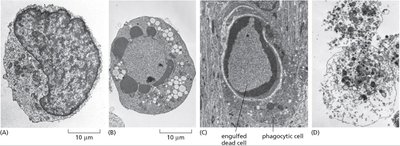
Initiator Caspase Activation
Initiator caspases are activated by apoptotic signals, leading to dimerization and cleavage. 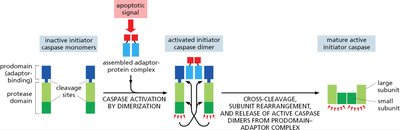
Executioner Caspase Activation
Executioner caspases are activated by initiator caspases, cleaving cellular proteins and driving apoptosis. 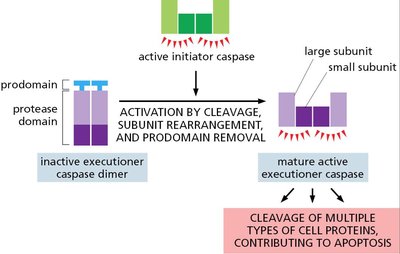
DNA Fragmentation During Apoptosis
Caspase-activated DNase (CAD) cleaves DNA between nucleosomes, resulting in characteristic DNA laddering. 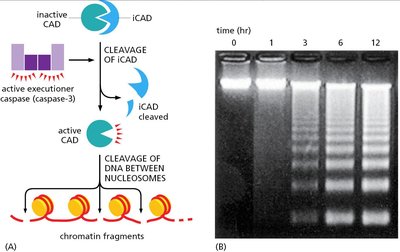
Extrinsic Apoptosis Pathway: Fas Receptor
The extrinsic pathway is triggered by death receptors such as Fas, leading to caspase activation. 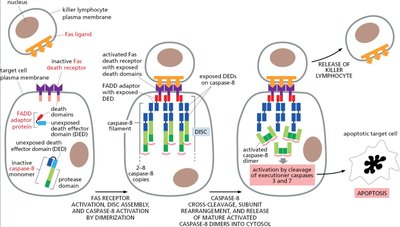
Intrinsic Apoptosis Pathway
The intrinsic pathway is initiated by mitochondrial release of cytochrome c, forming the apoptosome and activating caspase-9. 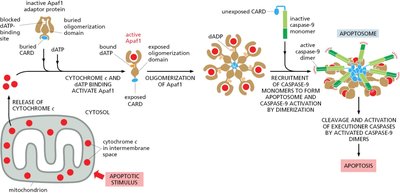
Bcl2 Proteins in Apoptosis
Bcl2 family proteins regulate mitochondrial membrane permeability and apoptosis. - Anti-apoptotic: Bcl2, Bcl-XL. - Pro-apoptotic: Bax, Bak, Bad. 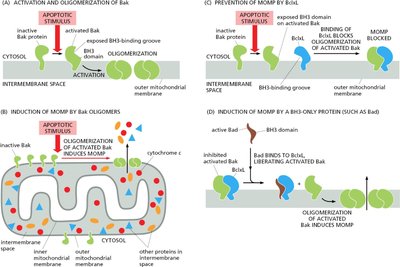
Inhibition of Apoptosis
Survival factors can inhibit apoptosis by upregulating anti-apoptotic proteins or inactivating pro-apoptotic proteins. 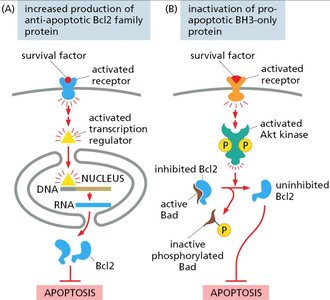
Cancer: Cell Cycle Dysregulation and Tumorigenesis
Benign vs. Malignant Tumors
Benign tumors remain localized, while malignant tumors invade surrounding tissues and can metastasize. 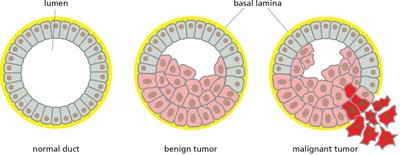
Clonal Evolution in Tumors
Tumors evolve through accumulation of mutations, leading to increased proliferation and invasiveness. 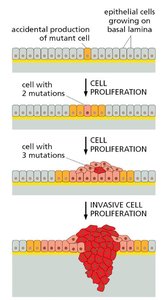
Genomic Instability and Aneuploidy
Cancer cells often exhibit genomic instability, including aneuploidy (abnormal chromosome number), which drives tumor progression. 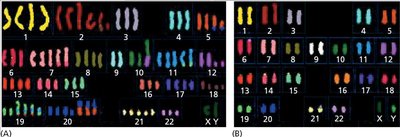
Chromosome Segregation Defects
Errors in mitosis can lead to chromosome mis-segregation, micronucleus formation, and chromothripsis. 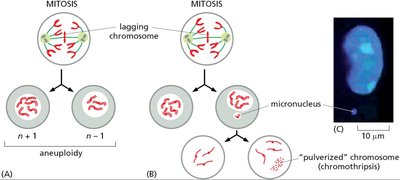
Cancer Risk and Proliferative Cells
Highly proliferative cells are at greater risk for cancer due to increased opportunities for mutation accumulation.
Cancer Hallmarks
Cancer cells display hallmarks such as sustained proliferation, evasion of apoptosis, genomic instability, and altered metabolism.
Contact Inhibition in Cell Culture
Normal cells stop dividing when they contact each other, but cancer cells lose this property, leading to uncontrolled growth.
Cancer Cell Metabolism
Cancer cells often exhibit altered metabolism, such as increased glycolysis (Warburg effect).
Oncogenes and Tumor Suppressor Genes
- Oncogenes: Genes that promote cell proliferation when mutated or overexpressed. - Tumor suppressor genes: Genes that inhibit cell cycle progression or promote apoptosis; loss of function leads to cancer.
Tumor Suppressor p53
p53 is a key tumor suppressor, known as the "guardian of the genome," regulating cell cycle arrest and apoptosis in response to DNA damage.
Heterogeneity of Subclones
Tumors are composed of heterogeneous subclones, each with distinct genetic mutations.
Genetic Mutations in Cancer Progression
Cancer progression involves accumulation of mutations in key genes, often driven by carcinogens.
Targeting Cancer Cells for Therapy
Therapies target cancer cells' genomic instability, upregulated pathways, and tumor-specific antigens.
Immune System and Cancer
The immune system can recognize and attack tumor-specific antigens; immune checkpoint inhibitors are used in cancer immunotherapy.
