Back
BackCell Junctions and Stem Cells: Structure, Function, and Regulation
Study Guide - Smart Notes

Cell Junctions
Overview of Cell Junctions
Cell junctions are specialized structures that connect adjacent cells or cells to the extracellular matrix, providing mechanical strength and facilitating communication. They are essential for maintaining tissue integrity and regulating cellular processes.
Adherens Junctions: Connect actin filaments between cells via cadherins.
Desmosomes: Link intermediate filaments between cells using specialized cadherins.
Tight Junctions: Seal neighboring cells to prevent leakage of molecules.
Gap Junctions: Allow direct communication between cells through channels.
Hemidesmosomes: Anchor cells to the extracellular matrix via integrins.
Cell Membrane Adhesion Proteins
Adhesion proteins mediate cell-cell and cell-matrix interactions, crucial for tissue structure and signaling.
Cadherins: Mediate calcium-dependent cell-cell adhesion; essential for adherens junctions and desmosomes.
Integrins: Mediate cell-matrix adhesion; connect the cytoskeleton to the extracellular matrix.
Cadherin Structure and Function
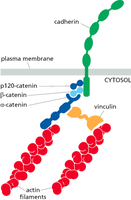
Cadherins are transmembrane proteins that require calcium ions (Ca2+) to maintain their rigid, adhesive conformation. Their extracellular domains interact with cadherins on adjacent cells, while their cytoplasmic tails bind to catenins, linking to the actin cytoskeleton.
Low Affinity Binding: Individual cadherin interactions are weak, but collectively provide strong adhesion ("Velcro" principle).
Catenins: Link cadherins to actin filaments, facilitating signal transduction and mechanical stability.
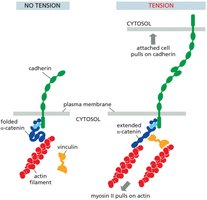
Mechanotransduction at Cell Junctions
Mechanotransduction is the process by which cells sense and respond to mechanical forces through junctional proteins. Forces are balanced across the junction, and tension can induce conformational changes in proteins like α-catenin, affecting downstream signaling.
Vinculin: Recruited under tension to reinforce the linkage between cadherins and actin filaments.
Biochemical Signals: Mechanical stress can trigger signaling pathways that regulate cell behavior.
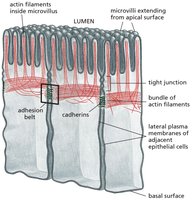
Adherens Junctions in Epithelial Cells
Adherens junctions are prominent in epithelial tissues, where they form adhesion belts that encircle cells and connect to actin filaments, maintaining tissue integrity and facilitating coordinated movement.
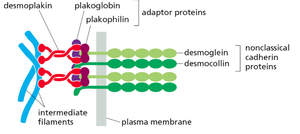
Desmosomes
Desmosomes are specialized adhesive structures similar to adherens junctions but connect to intermediate filaments (e.g., keratin) instead of actin. They provide strong mechanical attachment between cells, especially in tissues subject to stress, such as the skin and heart.
Desmoglein and Desmocollin: Nonclassical cadherins specific to desmosomes.
Adaptor Proteins: Plakoglobin, plakophilin, and desmoplakin link cadherins to intermediate filaments.
Tight Junctions
Tight junctions seal the space between adjacent epithelial cells, preventing the passage of molecules and maintaining distinct cellular compartments. They are crucial for the barrier function of epithelial layers.
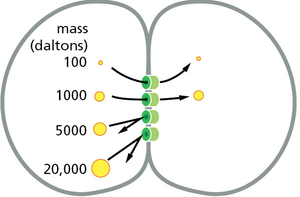
Gap Junctions
Gap junctions are intercellular channels that allow the direct transfer of ions and small molecules between neighboring cells, facilitating electrical and metabolic coupling.
Connexins: The protein subunits that form gap junction channels.
Size Selectivity: Only molecules below a certain size (typically <1 kDa) can pass through.
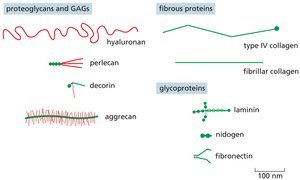
Extracellular Matrix (ECM)
The ECM is a complex network of proteins and polysaccharides that provides structural support to cells and tissues. It also regulates cell behavior through biochemical and mechanical signals.
Proteoglycans and Glycosaminoglycans (GAGs): Hydrated gel-forming components that resist compression.
Fibrous Proteins: Collagens provide tensile strength; elastin allows tissues to stretch and recoil.
Glycoproteins: Laminin, fibronectin, and nidogen mediate cell-ECM adhesion and organization.
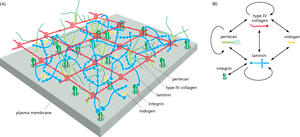
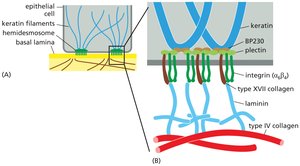
Integrins and Hemidesmosomes
Integrins are transmembrane receptors that mediate cell-ECM adhesion. Hemidesmosomes are specialized structures that anchor epithelial cells to the basal lamina via integrins and intermediate filaments.
Integrin Structure: Heterodimers composed of α and β subunits.
Hemidesmosomes: Link keratin filaments inside the cell to the ECM outside, providing strong adhesion to the basal lamina.
Cell Hierarchy and Stem Cells
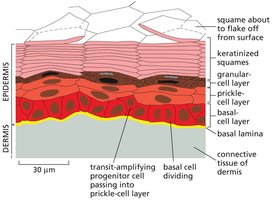
Epidermal Structure and Cell Renewal
The epidermis is a multilayered tissue with a well-defined hierarchy of cells, from dividing basal cells to terminally differentiated squames. Cell renewal is maintained by stem cells located in the basal layer.
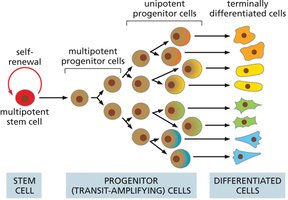
Cell Hierarchy
Cellular differentiation follows a hierarchical model, with multipotent stem cells giving rise to progenitor cells, which then differentiate into specialized cell types. This hierarchy ensures tissue maintenance and repair.
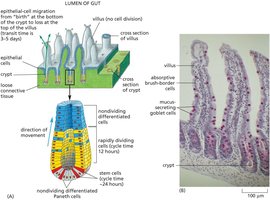
Gut Epithelial Lining Renewal
The gut epithelium is continuously renewed by stem cells located in the crypts. These stem cells divide to produce progenitor cells, which migrate upwards and differentiate as they move toward the villus tip.
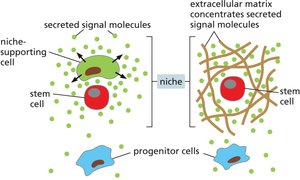
Stem-Cell Niche and Regulation
The stem-cell niche is a specialized microenvironment that maintains stem cell properties and regulates their proliferation and differentiation. The size of the niche and local signals control the number of stem cells.
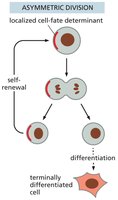
Asymmetric Division: Produces one stem cell and one differentiated cell, maintaining the stem cell pool.
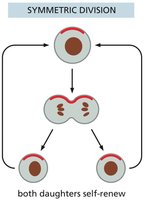
Symmetric Division: Can expand or deplete the stem cell population depending on the fate of daughter cells.
Pluripotency and Induced Pluripotent Stem Cells (iPS Cells)
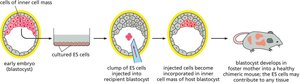
Embryonic Stem Cells and Pluripotency
Embryonic stem (ES) cells are totipotent or pluripotent, meaning they can give rise to all or most cell types in the organism. Pluripotency is a defining feature of ES cells, enabling their use in developmental biology and regenerative medicine.
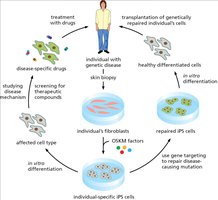
Induced Pluripotent Stem Cells (iPS Cells)
iPS cells are generated by reprogramming differentiated somatic cells (e.g., fibroblasts) using defined transcription factors (OKSM: Oct4, Klf4, Sox2, Myc). iPS cells exhibit properties similar to ES cells, including pluripotency and self-renewal.

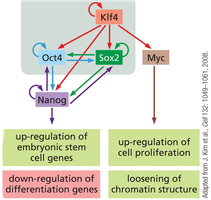
Reprogramming and Differentiation of iPS Cells
The process of reprogramming involves sequential activation of pluripotency genes and silencing of somatic genes. iPS cells can be differentiated into various cell types for research and therapeutic applications.
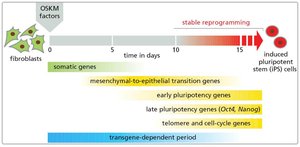
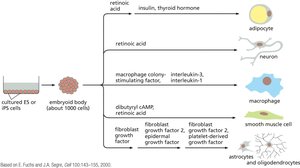
Applications of iPS Technology
iPS technology enables disease modeling, drug screening, and personalized cell therapy. Patient-specific iPS cells can be genetically corrected and differentiated into healthy cells for transplantation.