 Back
BackCell Membranes, Membrane Transport, Endomembrane System, and Cytoskeleton: Study Guide
Study Guide - Smart Notes

Cell Membranes
Major Biological Functions of Cellular Membranes
Cellular membranes are essential for maintaining the integrity and functionality of cells. They:
Define cell boundaries and separate the internal environment from the external environment.
Regulate transport of substances into and out of the cell.
Facilitate communication via receptors and signaling molecules.
Provide sites for biochemical reactions and energy transduction.
Anchor the cytoskeleton and help maintain cell shape.
Development of the Fluid Mosaic Model
The fluid mosaic model describes the structure of cell membranes as a mosaic of components that gives the membrane a fluid character. Key steps in its development:
Early models proposed a lipid bilayer based on the amphipathic nature of phospholipids.
Freeze-fracture electron microscopy revealed proteins embedded within the bilayer.
Fluorescence recovery after photobleaching (FRAP) demonstrated lateral mobility of membrane proteins.
The model was refined to include the dynamic, heterogeneous distribution of proteins and lipids.
Major Components of the Cell Membrane
Lipids: Phospholipids, glycolipids, and sterols (e.g., cholesterol).
Proteins: Integral, peripheral, and lipid-anchored proteins.
Carbohydrates: Attached to lipids (glycolipids) or proteins (glycoproteins).
Main Classes of Membrane Lipids and Their Structures
Phospholipids: Glycerol backbone, two fatty acid tails, and a phosphate-containing head group.
Glycolipids: Lipids with carbohydrate groups attached, important for cell recognition.
Sterols: Such as cholesterol, with a rigid ring structure.
Phospholipids
Phospholipids are the most abundant membrane lipids and are classified by their phosphate-containing head groups (e.g., phosphatidylcholine, phosphatidylethanolamine).
Cholesterol: Functions and Properties
Cholesterol is a sterol that modulates membrane fluidity and stability.
It is amphipathic, with a hydrophilic hydroxyl group and a hydrophobic steroid ring structure.
Factors Influencing Membrane Fluidity
Fatty acid composition (saturated vs. unsaturated).
Cholesterol content.
Temperature.
Lipid Rafts
Lipid rafts are microdomains enriched in cholesterol, sphingolipids, and certain proteins.
They serve as organizing centers for signaling molecules.
Glycoproteins and Glycolipids
Glycoproteins: Proteins with carbohydrate chains attached, found on the extracellular surface of the membrane.
Glycolipids: Lipids with carbohydrate groups, also on the extracellular surface.
Human Blood Types: Molecular Basis
Blood type is determined by specific glycosylation patterns on red blood cell membranes.
Type O lacks A or B antigens, making it a universal donor; type AB has both, making it a universal recipient.
Detergents and Integral Membrane Proteins
Detergents are amphipathic molecules that solubilize membrane proteins by surrounding their hydrophobic regions, keeping them soluble in aqueous solutions.
Cell Walls in Different Organisms
Plants: Cellulose-based cell walls.
Fungi: Chitin-based cell walls.
Bacteria: Peptidoglycan-based cell walls.
Techniques: SDS-PAGE and FRAP
SDS-PAGE: Separates proteins by size using a detergent (SDS) that denatures proteins and gives them a uniform negative charge.
FRAP (Fluorescence Recovery After Photobleaching): Measures the lateral mobility of membrane proteins by bleaching a fluorescently labeled area and observing recovery.
Transport Across Membranes
Types of Membrane Transport
Simple diffusion: Movement of small, nonpolar molecules down their concentration gradient.
Facilitated diffusion: Movement of molecules via transport proteins, still down the gradient.
Passive transport: Includes simple and facilitated diffusion; does not require energy.
Active transport: Moves substances against their gradient; requires energy (ATP or ion gradients).
Primary active transport: Direct use of ATP (e.g., Na+/K+ pump).
Secondary active transport: Uses energy from an existing gradient (e.g., symporters, antiporters).
Symport: Two substances move in the same direction.
Antiport: Two substances move in opposite directions.
Carrier proteins: Bind and transport specific molecules.
Channel proteins: Form pores for passive movement of ions or water.
Osmosis: Diffusion of water across a semipermeable membrane.
Hypertonic solution: Higher solute concentration outside the cell; cell shrinks.
Hypotonic solution: Lower solute concentration outside; cell swells.
Membrane Transport in Erythrocytes
Examples include glucose transport via GLUT1 (facilitated diffusion) and anion exchange via band 3 protein (antiport).
Endomembrane System
Components of the Endomembrane System
Includes the nuclear envelope, endoplasmic reticulum (ER), Golgi apparatus, lysosomes, endosomes, and transport vesicles.
Functions of Rough ER and Smooth ER
Rough ER: Protein synthesis and modification.
Smooth ER: Lipid synthesis, detoxification, and calcium storage.
Cotranslational Translocation
Process by which nascent polypeptides are translocated into the ER lumen during translation.
Protein Synthesis: ER vs. Cytosol
Proteins destined for secretion, membranes, or lysosomes are synthesized on the ER.
Cytosolic, nuclear, and mitochondrial proteins are synthesized in the cytosol.
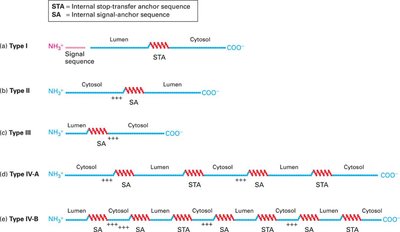
Membrane Protein Topology and Anchor Sequences
Membrane protein topology is determined by the presence and arrangement of signal-anchor (SA) and stop-transfer anchor (STA) sequences, which are hydrophobic regions that direct membrane insertion and orientation.
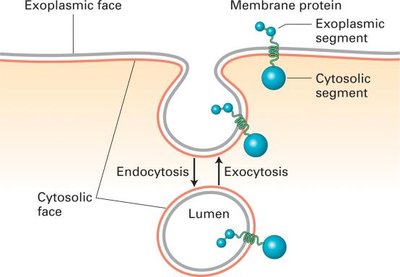
Membrane Orientation During Budding and Fusion
Membrane orientation (cytosolic vs. non-cytosolic faces) is preserved during vesicle budding and fusion, ensuring that the topology of membrane proteins and lipids remains consistent.
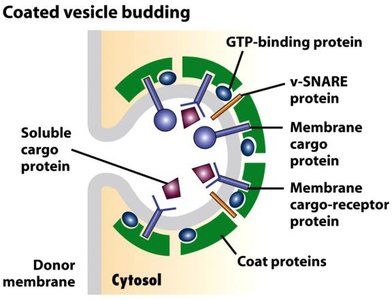
Vesicle Budding: Components and Functions
GTP-binding proteins: Regulate vesicle formation and uncoating.
v-SNARE proteins: Mediate vesicle fusion with target membranes.
Coat proteins: Shape the vesicle and select cargo.
Membrane cargo proteins: Proteins to be transported.
Membrane cargo protein receptors: Recognize and bind cargo proteins.
Vesicular Transport Pathways
Vesicular transport involves the movement of cargo between organelles via coated vesicles, with specific coat proteins (e.g., COPI, COPII) and SNAREs ensuring correct targeting and fusion.
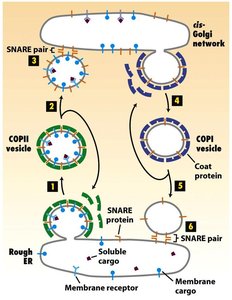
Vesicle Coat Shedding and SNARE Exposure
The vesicle coat is shed after budding to expose v-SNAREs, which are necessary for vesicle docking and fusion with the target membrane.
Vesicle Target Recognition
Vesicles recognize their target organelles via specific interactions between v-SNAREs (on vesicles) and t-SNAREs (on target membranes).
Retrograde Vesicular Transport
Retrograde transport returns escaped ER proteins and recycles membrane components from the Golgi back to the ER.
KDEL Signal
The KDEL sequence is a retrieval signal for ER-resident proteins. If a KDEL-containing protein reaches the Golgi, it is recognized by KDEL receptors and returned to the ER.
Receptor-Mediated Endocytosis
Specific uptake of molecules into the cell via receptors and clathrin-coated vesicles.
Lysosome Functions
Lysosomes are acidic organelles containing hydrolytic enzymes for degradation of macromolecules, cellular debris, and pathogens.
Cytoskeleton
What is the Cytoskeleton?
The cytoskeleton is a dynamic network of protein filaments that provides structural support, facilitates cell movement, and organizes organelles within the cell.
Microfilaments: Components and Polarity
Composed of actin monomers.
Polar structures with distinct plus (barbed) and minus (pointed) ends.
Microfilaments vs. Microtubules
Microfilaments: Actin-based, involved in cell shape and movement.
Microtubules: Tubulin-based, involved in intracellular transport and cell division.
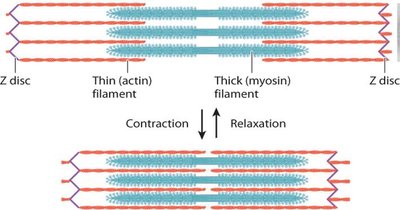
Myosin and Muscle Contraction
Myosin: Motor protein that interacts with actin to generate force.
Myosin I, II, and V move toward the plus end of actin filaments; myosin II forms bipolar filaments in muscle.
Muscle contraction involves sliding of actin (thin) and myosin (thick) filaments.
Calcium ions (Ca2+) trigger contraction by enabling myosin-actin interaction.
Microtubules: Structure and Polarity
Composed of α- and β-tubulin heterodimers arranged into protofilaments.
Microtubules are polar, with a plus end (fast-growing) and a minus end (anchored at the MTOC).
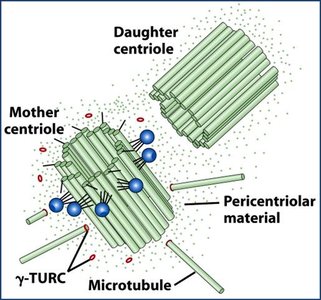
Microtubule-Organizing Centers (MTOCs) and Centrosomes
MTOCs nucleate and anchor microtubules; the centrosome is the primary MTOC in animal cells.
Centrosomes contain a pair of centrioles and pericentriolar material, including γ-tubulin ring complexes (γ-TuRC) for microtubule nucleation.
Non-mitotic cells have one centrosome; mitotic cells have two.
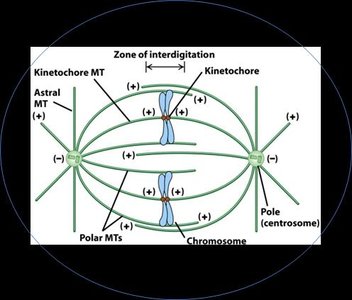
Mitotic Spindle and Microtubule Classes
Mitotic spindle contains kinetochore, polar, and astral microtubules.
Kinetochore MTs attach to chromosomes; polar MTs overlap at the spindle midzone; astral MTs anchor the spindle to the cortex.
Dynamic Instability and Treadmilling
Dynamic instability: Rapid switching between growth and shrinkage of microtubules.
Treadmilling: Addition of subunits at one end and loss at the other, resulting in movement of the filament.
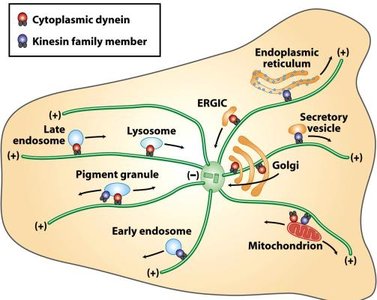
Microtubule Motor Proteins
Kinesins: Move toward the plus end of microtubules (anterograde transport).
Dyneins: Move toward the minus end (retrograde transport).
Both use ATP hydrolysis for energy.
γ-Tubulin Ring Complex (γ-TuRC)
γ-TuRC nucleates microtubule assembly at the centrosome.
Intermediate Filaments
Intermediate in size between microfilaments and microtubules.
Composed of various proteins (e.g., keratins, lamins).
Non-polar and provide mechanical strength.
No associated motor proteins.
HIV Intracellular Trafficking
HIV utilizes the cytoskeleton for movement within host cells, exploiting microtubule-based transport for efficient infection.
Application of Cytoskeletal Concepts
Understanding cytoskeletal dynamics is essential for interpreting cell movement, division, and intracellular transport in health and disease.
