 Back
BackCellular Junctions, Cytoskeletal Systems, and Regulation of Muscle Contraction
Study Guide - Smart Notes

Cellular Junctions
Types of Cellular Junctions
Cellular junctions are specialized structures that connect cells to one another or to the extracellular matrix, playing critical roles in tissue integrity, communication, and motility. The main types include:
Adhesive Junctions (Adherens): These junctions provide strong mechanical attachments between adjacent cells.
Transmitting Junctions: Facilitate communication and transport between cells.
Desmosomes: Spot-like adhesions that anchor intermediate filaments, providing mechanical strength.
Tight Junctions: Seal adjacent cells, preventing passage of molecules between them.
Adhesion Belt: Composed of tight junctions, encircling cells and maintaining tissue structure.
Gap Junctions: Channels that allow direct communication between cytoplasms of adjacent cells.
Plasmodesmata: Specialized junctions in plant cells for cytoplasmic exchange.
Focal Adhesions & Hemidesmosomes: Attach cells to the substrate and facilitate motility.
Plasmodesmata Structure and Function
Plasmodesmata are unique to plant cells, enabling transport and communication between cells through the cell wall. They consist of a plasma membrane-lined channel containing a central desmotubule derived from the endoplasmic reticulum.
Desmotubule: A central rod-like structure facilitating molecular transport.
Cytoplasmic Sleeve: Surrounds the desmotubule, allowing passage of ions and small molecules.
Spoke Proteins: Connect the desmotubule to the plasma membrane.
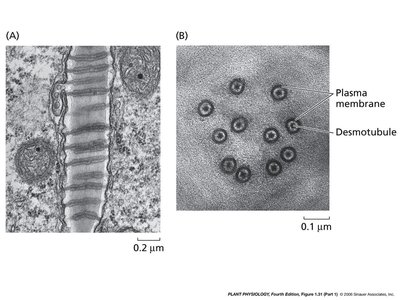
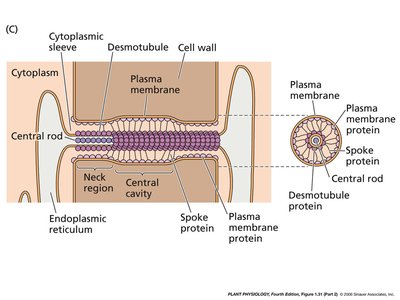
Example: Plasmodesmata are essential for the transport of nutrients and signaling molecules in plant tissues.
Cytoskeletal Systems and Motor Proteins
Motility & Motor Proteins
The cytoskeleton is a dynamic network of protein filaments that provides structural support, facilitates intracellular transport, and enables cell movement. Motor proteins such as kinesin and dynein move along microtubules, transporting vesicles and organelles.
Kinesin: Moves toward the plus end of microtubules, typically transporting cargo away from the cell center.
Dynein: Moves toward the minus end, carrying cargo toward the cell center.
ATP Hydrolysis: Provides energy for motor protein movement.
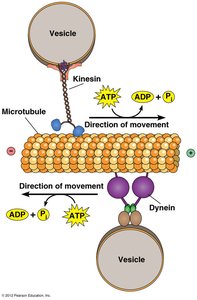
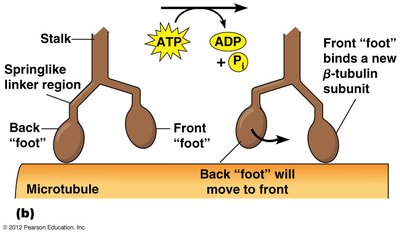
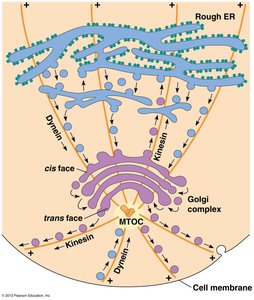
Example: Kinesin transports vesicles from the Golgi apparatus to the cell membrane, while dynein returns them toward the nucleus.
Thin Filaments and Muscle Contraction
Structure of Thin Filaments
Thin filaments are essential components of muscle fibers, interdigitating with thick filaments to enable contraction. They are composed of:
F-actin: Helical polymer of G-actin monomers.
Tropomyosin: Rod-shaped protein that blocks myosin binding sites on actin.
Troponin Complex: Consists of three subunits: TnT (binds tropomyosin), TnC (binds calcium), and TnI (inhibitory).
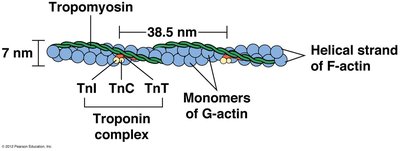
Example: The troponin-tropomyosin complex acts as a calcium-sensitive switch for muscle contraction.
Mechanism of Muscle Contraction
Muscle contraction is regulated by the interaction between actin and myosin, controlled by calcium ions. The process involves:
Cross-Bridge Formation: Myosin head binds to actin.
Power Stroke: Myosin head pivots, pulling actin filament.
ATP Binding: Causes myosin to detach from actin.
ATP Hydrolysis: Energizes myosin for the next cycle.
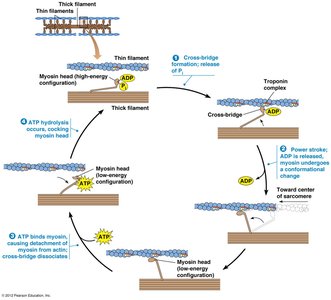
Example: The sliding filament model explains how muscles contract by the movement of actin and myosin filaments.
Regulation of Muscle Contraction by Calcium
Calcium ions play a pivotal role in muscle contraction by regulating the availability of myosin binding sites on actin filaments. When calcium concentration is low, tropomyosin blocks these sites; when calcium is high, it binds to TnC, causing tropomyosin to shift and exposing the binding sites.
Low Calcium: Tropomyosin blocks myosin binding sites.
High Calcium: Calcium binds TnC, tropomyosin shifts, myosin binds actin.
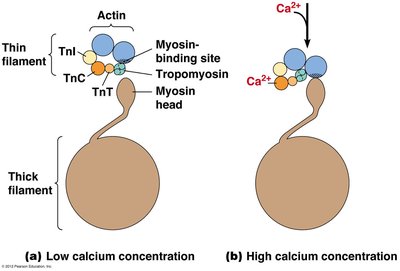
Example: Calcium release from the sarcoplasmic reticulum triggers muscle contraction.
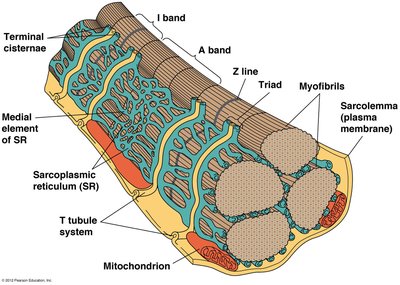
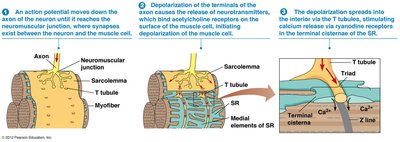
Regulation of Calcium Levels in Skeletal Muscle Cells
Calcium levels in muscle cells are tightly regulated by nerve impulses. The sarcoplasmic reticulum (SR) stores calcium and releases it in response to action potentials at the neuromuscular junction. Calcium ATPase pumps return calcium to the SR, allowing muscle relaxation.
Neuromuscular Junction: Site where motor neuron contacts muscle cell.
Acetylcholine Release: Triggers action potential in muscle cell.
Calcium Release: Depolarization opens channels, releasing calcium into sarcoplasm.
Calcium ATPase: Pumps calcium back into SR for relaxation.
Example: Rapid changes in calcium concentration enable quick muscle contraction and relaxation.
Focal Adhesions and Cellular Movement
Role of Focal Adhesions in Cell Motility
Focal adhesions are dynamic structures that anchor cells to the extracellular matrix and regulate cell movement. They act as "molecular clutches," promoting protrusion at the leading edge and converting actin dynamics into traction forces.
Membrane Protrusion: Focal adhesions resist actin retrograde flow, promoting forward movement.
Traction Forces: Myosin pulling forces are converted into movement against the extracellular matrix.
Clutch Engagement: Organization of actin and adhesion components increases protrusion rate.
Example: Focal adhesions are essential for cell migration during wound healing and development.
Primary Cilia
Structure and Function of Primary Cilia
Primary cilia are sensory organelles with a "9+0" microtubule arrangement, lacking the central pair. They play important roles in development and sensory perception. Defects in primary cilia can lead to disorders such as deafness and left-right asymmetry reversals.
9+0 Structure: Nine microtubule doublets without a central pair.
Sensory Function: Detect environmental signals.
Developmental Role: Essential for proper tissue patterning.
Example: Mutations affecting primary cilia can cause ciliopathies, impacting organ development and function.
