 Back
BackChapter 18: Cell Death – Apoptosis and Necrosis
Study Guide - Smart Notes

Cell Death: Apoptosis and Necrosis
Overview of Cell Death
Cell death is a fundamental biological process essential for development, tissue homeostasis, and the removal of damaged or dangerous cells. There are two primary forms of cell death: apoptosis (programmed cell death) and necrosis (uncontrolled cell death). Understanding the mechanisms and consequences of these processes is crucial in cell biology.
Apoptosis: A regulated, energy-dependent process leading to cell elimination without causing inflammation.
Necrosis: An unregulated, often pathological process resulting from acute cellular injury, leading to cell lysis and inflammation.
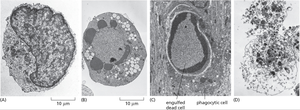
Apoptosis vs. Necrosis
Apoptosis and necrosis differ in their triggers, morphological features, and biological outcomes.
Apoptosis: Characterized by cell shrinkage, chromatin condensation, membrane blebbing, and formation of apoptotic bodies, which are phagocytosed by neighboring cells.
Necrosis: Characterized by cell swelling, loss of membrane integrity, and release of cellular contents, often triggering inflammation.
Physiological Role: Apoptosis is essential for normal development and tissue maintenance, while necrosis is typically associated with injury or disease.
Apoptosis in Development and Homeostasis
Role of Apoptosis in Development
Apoptosis is crucial for shaping tissues and organs during development. It removes unnecessary or misplaced cells, ensuring proper morphogenesis.
Example: Removal of interdigital cells during limb formation in vertebrates, allowing for the separation of fingers and toes.
Apoptosis in Homeostasis
In adult organisms, apoptosis maintains tissue homeostasis by balancing cell proliferation and cell death. It eliminates damaged, infected, or potentially cancerous cells.
Molecular Mechanisms of Apoptosis
Caspases: Central Effectors of Apoptosis
Caspases are a family of cysteine proteases that orchestrate the apoptotic process by cleaving specific cellular substrates. They exist as inactive precursors (procaspases) and are activated in a cascade.
Initiator Caspases: Activated by apoptotic signals, they cleave and activate executioner caspases.
Executioner Caspases: Responsible for the cleavage of cellular proteins, leading to the morphological and biochemical features of apoptosis.
Initiator Caspase Activation
Initiator caspases are activated by dimerization and cleavage upon assembly into adaptor protein complexes in response to apoptotic signals.
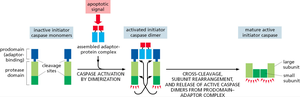
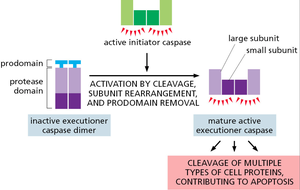
Executioner Caspase Activation
Executioner caspases are activated by cleavage, subunit rearrangement, and removal of the prodomain, enabling them to cleave multiple cellular substrates.
DNA Fragmentation During Apoptosis
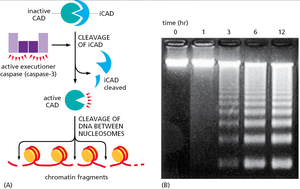
One hallmark of apoptosis is the fragmentation of nuclear DNA. This is mediated by the activation of CAD (caspase-activated DNase), which is released upon cleavage of its inhibitor (iCAD) by executioner caspases.
Result: DNA is cleaved between nucleosomes, producing a characteristic "ladder" pattern on agarose gels.
Pathways of Apoptosis
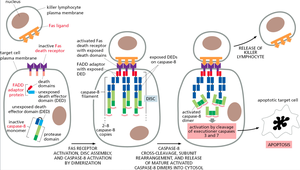
Extrinsic Pathway: Fas Receptor-Mediated Apoptosis
The extrinsic pathway is triggered by the binding of extracellular death ligands (e.g., Fas ligand) to their receptors (e.g., Fas receptor) on the cell surface. This leads to the formation of the death-inducing signaling complex (DISC) and activation of initiator caspase-8.
Key Steps: Ligand binding, DISC assembly, caspase-8 activation, executioner caspase activation, apoptosis.
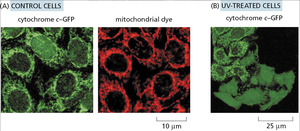
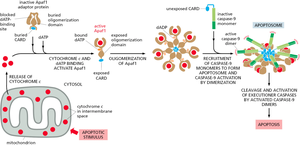
Intrinsic Pathway: Mitochondrial (Cytochrome c) Pathway
The intrinsic pathway is activated by intracellular stress signals (e.g., DNA damage, oxidative stress) and involves the release of cytochrome c from mitochondria into the cytosol. Cytochrome c binds Apaf-1, leading to apoptosome formation and activation of initiator caspase-9.
Key Steps: Mitochondrial outer membrane permeabilization, cytochrome c release, apoptosome assembly, caspase-9 activation, executioner caspase activation.
Regulation of Apoptosis
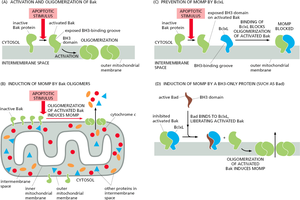
Bcl-2 Family Proteins
The Bcl-2 family of proteins tightly regulates the intrinsic pathway by controlling mitochondrial outer membrane permeabilization. Members include both pro-apoptotic (e.g., Bax, Bak) and anti-apoptotic (e.g., Bcl-2, Bcl-xL) proteins.
Pro-apoptotic: Promote cytochrome c release by forming pores in the mitochondrial membrane.
Anti-apoptotic: Inhibit pore formation and prevent apoptosis.
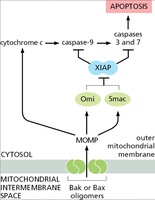
Inhibition of Apoptosis: XIAP and IAPs
Inhibitor of apoptosis proteins (IAPs), such as XIAP, block caspase activity and prevent apoptosis. Mitochondrial proteins (e.g., Smac/DIABLO, Omi) released during apoptosis can neutralize IAPs, allowing caspase activation to proceed.
Summary Table: Apoptosis vs. Necrosis
Feature | Apoptosis | Necrosis |
|---|---|---|
Regulation | Highly regulated (programmed) | Unregulated (accidental) |
Cell Morphology | Shrinkage, blebbing, apoptotic bodies | Swelling, lysis |
DNA Fragmentation | Ladder pattern (nucleosomal) | Random degradation |
Inflammation | None | Often present |
Physiological Role | Development, homeostasis | Pathological (injury, infection) |
Key Terms
Apoptosis: Programmed cell death involving caspase activation and DNA fragmentation.
Necrosis: Uncontrolled cell death due to injury, causing inflammation.
Caspases: Proteases that mediate apoptosis by cleaving cellular proteins.
Bcl-2 Family: Proteins that regulate mitochondrial pathway of apoptosis.
Cytochrome c: Mitochondrial protein released to activate apoptosome in apoptosis.
XIAP: Inhibitor of apoptosis protein that blocks caspase activity.
Additional info: Apoptosis is also implicated in various diseases, including cancer (where it is suppressed) and neurodegenerative disorders (where it is excessive).
