 Back
BackChemotrophic Energy Metabolism: Glycolysis and Fermentation – Study Notes
Study Guide - Smart Notes

Chemotrophic Energy Metabolism: Glycolysis and Fermentation
Introduction to Chemotrophic Energy Metabolism
Chemotrophic energy metabolism encompasses the cellular processes by which organisms extract energy from chemical compounds, primarily through the breakdown of organic molecules. This energy is essential for cellular activities such as movement, transport, and biosynthesis.
Chemotrophs obtain energy by ingesting or engulfing food, which is then catabolized to release energy.
All cellular chemical reactions constitute metabolism, organized into metabolic pathways.
Metabolic Pathways
Anabolic and Catabolic Pathways
Metabolic pathways are classified as anabolic or catabolic based on their function and energy requirements.
Anabolic pathways synthesize complex molecules from simpler ones, increasing order and decreasing entropy. These are endergonic (require energy).
Catabolic pathways break down complex molecules into simpler ones, decreasing order and increasing entropy. These are exergonic (release energy).
Catabolic reactions produce metabolites and can be aerobic (with oxygen) or anaerobic (without oxygen).
ATP: The Primary Energy Molecule in Cells
Structure and Function of ATP
Adenosine triphosphate (ATP) is the main energy currency in cells, powering movement, transport, and enzymatic reactions.
ATP consists of adenine, ribose, and three phosphate groups linked by phosphoanhydride bonds.
Hydrolysis of ATP to ADP and Pi is highly exergonic: kcal/mol.
Other high-energy molecules include GTP, creatine phosphate, and reduced coenzymes like NADH.
Why ATP Hydrolysis Is Exergonic
Charge repulsion between phosphate groups strains bonds.
Resonance stabilization is greater in hydrolysis products than in ATP itself.
Increased entropy and solubility of products further drive the reaction.
ATP as an Intermediate Energy Carrier
ATP occupies an intermediate position among phosphorylated compounds, allowing it to both donate and accept phosphate groups in cellular reactions.
The ATP/ADP system is a reversible means of conserving and releasing energy.
Chemotrophic Energy Metabolism and Biological Oxidation
Oxidation and Reduction in Cells
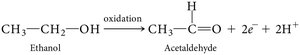
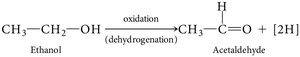
Energy-yielding oxidative reactions are central to chemotrophic metabolism. Biological oxidations typically involve the removal of both electrons and protons (dehydrogenation).
Oxidation: Removal of electrons (often as hydrogen atoms).
Reduction: Addition of electrons (often as hydrogen atoms).
Enzymes called dehydrogenases catalyze these reactions.

Role of Coenzymes
Coenzymes such as NAD+ serve as electron acceptors in biological oxidations, cycling between oxidized and reduced forms (NADH).
They are present in low concentrations and are recycled.
Glucose Catabolism: Glycolysis and Fermentation
Overview of Glycolysis
Glycolysis is a universal pathway that converts glucose to pyruvate, generating ATP and NADH. It occurs in the cytosol and is common to both aerobic and anaerobic organisms.
Net yield: 2 ATP and 2 NADH per glucose molecule.
Divided into three phases: preparation and cleavage, oxidation and ATP generation, pyruvate formation and ATP generation.
Fate of Pyruvate: Aerobic vs. Anaerobic Conditions
Pyruvate is a key metabolic branch point. In the presence of oxygen, it is further oxidized to acetyl-CoA and enters the citric acid cycle. In the absence of oxygen, it undergoes fermentation to regenerate NAD+.
Lactate fermentation: Pyruvate is reduced to lactate (in animals and some bacteria).
Alcoholic fermentation: Pyruvate is converted to ethanol and CO2 (in plants, yeast, and some microorganisms).

Types of Fermentation
Lactate fermentation: Catalyzed by lactate dehydrogenase, important in muscle cells under low oxygen.
Alcoholic fermentation: Involves decarboxylation of pyruvate to acetaldehyde, then reduction to ethanol.
Other fermentations: Propionate, butylene glycol, acetone, and butyrate fermentations occur in various bacteria.
Energy Yield and Efficiency of Fermentation
Fermentation yields only 2 ATP per glucose, but conserves energy efficiently (about 30%).
Most free energy remains in the fermentation end products (lactate or ethanol).
Regulation of Glycolysis and Gluconeogenesis
Allosteric Regulation
Key enzymes in glycolysis and gluconeogenesis are regulated allosterically by effectors such as AMP, acetyl-CoA, and fructose-2,6-bisphosphate (F2,6BP).
AMP activates glycolysis, inhibits gluconeogenesis.
Acetyl-CoA activates gluconeogenesis, inhibits glycolysis.
F2,6BP is a potent activator of phosphofructokinase-1 (PFK-1) and inhibitor of fructose-1,6-bisphosphatase.
Hormonal Regulation
cAMP, regulated by hormones like glucagon and epinephrine, modulates F2,6BP levels and thus glycolytic/gluconeogenic flux.
Alternative Substrates and Pathways
Other Sugars and Glycerol
Cells can catabolize sugars other than glucose and glycerol by converting them into glycolytic intermediates.
Monosaccharides and disaccharides are converted to glycolytic intermediates after phosphorylation.
Glycerol enters glycolysis as dihydroxyacetone phosphate.
Polysaccharide Breakdown
Starch (plants) and glycogen (animals) are cleaved by phosphorolysis to yield glucose-1-phosphate, which enters glycolysis.
Gluconeogenesis
Pathway and Regulation
Gluconeogenesis is the synthesis of glucose from non-carbohydrate precursors, mainly pyruvate and lactate. It is not a simple reversal of glycolysis; key steps are bypassed by alternative reactions.
Bypass reactions circumvent the most exergonic steps of glycolysis (Gly-1, Gly-3, Gly-10).
Regulation ensures that glycolysis and gluconeogenesis do not occur simultaneously in the same cell.
Clinical and Biotechnological Relevance
Cancer Cell Metabolism
Cancer cells often rely on aerobic glycolysis (Warburg effect), fermenting glucose to lactate even in the presence of oxygen. This supports rapid growth and biosynthesis.
Detected clinically by PET scans using radiolabeled glucose analogs.
Summary Table: Key Differences Between Glycolysis and Fermentation
Process | Oxygen Requirement | End Product | ATP Yield (per glucose) |
|---|---|---|---|
Glycolysis (aerobic) | Yes | Pyruvate (further oxidized) | ~38 (with full respiration) |
Glycolysis (anaerobic) | No | Lactate or Ethanol + CO2 | 2 |
Fermentation | No | Lactate or Ethanol + CO2 | 2 |
Additional info: The above notes integrate and expand upon the provided lecture content, including definitions, regulatory mechanisms, and clinical applications relevant to cell biology students.
