 Back
BackComprehensive Study Notes on Enzymes
Study Guide - Smart Notes

Introduction to Enzymes
Definition and Importance
Enzymes are biological catalysts that accelerate the rate of biochemical reactions in living organisms without being consumed in the process. Most enzymes are globular proteins, though some, such as ribozymes, are RNA molecules with catalytic activity. Enzymes are essential for processes such as respiration, photosynthesis, digestion, and biosynthesis of macromolecules.
Classification of Enzymes
Enzymes are classified based on the type of reaction they catalyze. The main classes include:
Oxidoreductases: Catalyze oxidation-reduction reactions (e.g., dehydrogenases, oxidases).
Transferases: Transfer functional groups between molecules (e.g., kinases, phosphorylases).
Hydrolases: Catalyze hydrolysis reactions (e.g., sucrase, lipases, proteases).
Lyases: Remove groups from substrates without hydrolysis (e.g., decarboxylases).
Isomerases: Rearrange atoms within a molecule (e.g., isomerases).
Ligases: Join two molecules using energy from ATP (e.g., synthetases, ligases).
Enzyme names typically end with '-ase'.
Characteristics of Enzymes
General Properties
Specificity: Enzymes are highly specific for their substrates due to the unique structure of their active sites.
Efficiency: Enzymes have high turnover rates, meaning a small amount can catalyze the conversion of large amounts of substrate.
Unchanged by Reaction: Enzymes remain chemically unchanged after catalysis and can be reused.
Affected by Environmental Factors: Enzyme activity is influenced by substrate concentration, enzyme concentration, temperature, and pH.
Cofactor Requirement: Some enzymes require non-protein cofactors (inorganic ions, coenzymes, or prosthetic groups) to function. The enzyme-cofactor complex is called a holoenzyme, while the protein part alone is an apoenzyme.
Regulation: Enzyme activity is tightly regulated by activators and inhibitors.
Equilibrium: Enzymes accelerate the attainment of equilibrium but do not alter the equilibrium position.
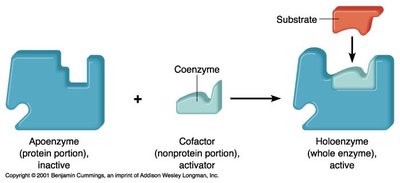
Types of Cofactors
Inorganic ions (e.g., Zn2+ for carbonic anhydrase)
Coenzymes: Organic molecules that bind loosely (e.g., NAD+ for dehydrogenases)
Prosthetic groups: Organic molecules tightly bound (e.g., haem in catalases)
Mode of Action of Enzymes
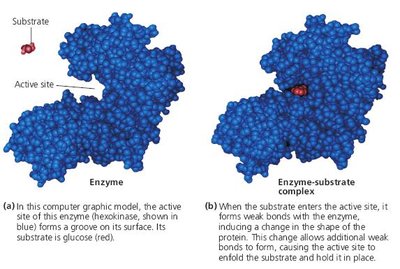
Active Site Structure
The active site of an enzyme is a specific region formed by the three-dimensional folding of the protein, comprising the substrate-binding site and the catalytic site. The active site is responsible for substrate recognition and catalysis.
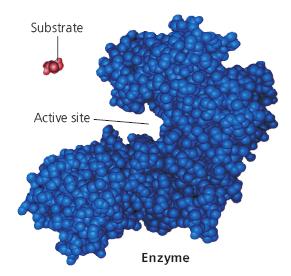
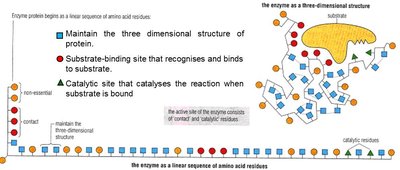
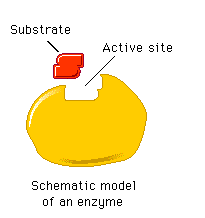
Enzyme Specificity
Enzyme specificity arises from the precise fit between the enzyme's active site and its substrate, determined by shape, size, charge, and orientation. This specificity is a direct result of the enzyme's unique amino acid sequence and three-dimensional conformation.
Enzyme-Substrate Complex
When a substrate binds to the active site, an enzyme-substrate (E-S) complex forms. The substrate is converted to product, which then leaves the active site, allowing the enzyme to catalyze further reactions.
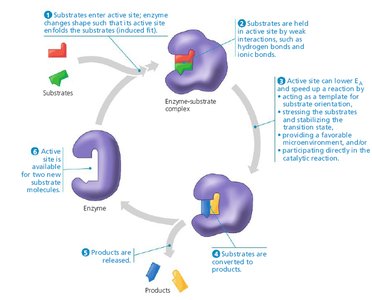
Lock-and-Key and Induced Fit Hypotheses
Lock-and-Key Hypothesis: The active site is perfectly complementary to the substrate, allowing precise binding (more likely for enzymes with a single substrate type).
Induced Fit Hypothesis: The active site is flexible and molds around the substrate upon binding, allowing for a broader substrate range.
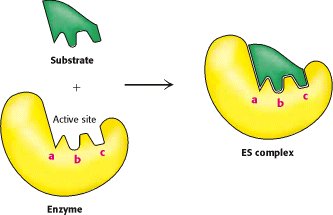
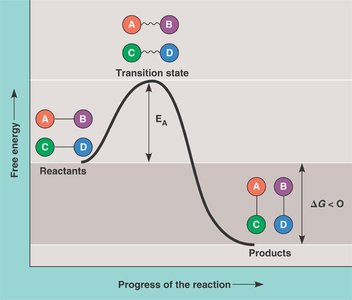
Activation Energy
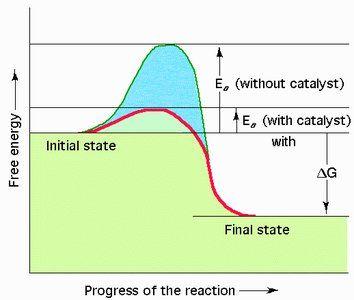
Activation energy (EA) is the minimum energy required for reactants to reach the transition state and undergo a chemical reaction. Enzymes lower the activation energy, providing an alternative pathway and increasing the reaction rate without raising the temperature.
Enzyme Kinetics
Measurement of Enzyme Kinetics
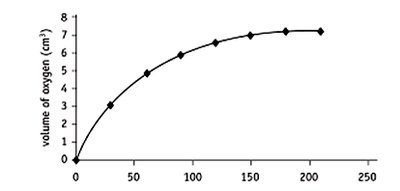
Enzyme kinetics studies the rates of enzyme-catalyzed reactions. Reaction rates can be measured by the amount of product formed or substrate depleted over time. The initial rate is determined from the linear portion of the reaction curve.
Product Formation: Example—catalase breaking down hydrogen peroxide, measuring oxygen produced.
Substrate Disappearance: Example—amylase hydrolyzing starch, measuring decrease in starch via colorimetry.
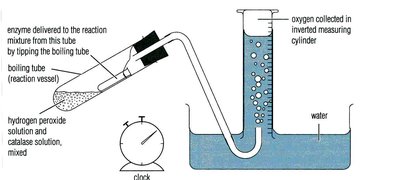
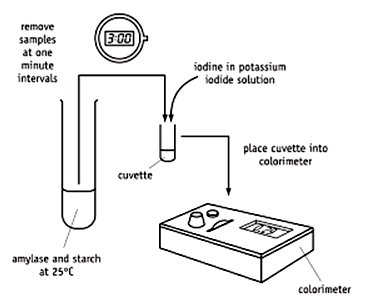
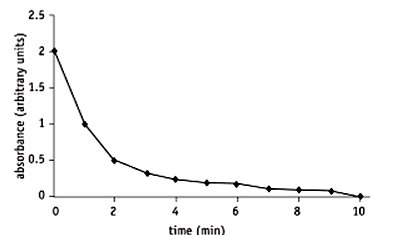
Measurement of Enzyme Affinity (Michaelis Constant, Km)
The Michaelis constant (Km) is the substrate concentration at which the reaction rate is half its maximum (Vmax). It indicates the affinity of the enzyme for its substrate: low Km means high affinity, high Km means low affinity.
$K_m = [S] \text{ when } V_i = \frac{1}{2} V_{max}$
Factors Affecting Enzyme Activity
Substrate Concentration
At low substrate concentrations, the reaction rate increases proportionally with substrate concentration.
At high substrate concentrations, the rate plateaus as all enzyme active sites become saturated.
Enzyme Concentration
At low enzyme concentrations, the reaction rate increases proportionally with enzyme concentration.
At high enzyme concentrations, the rate plateaus if substrate becomes limiting.
Temperature
Increasing temperature increases reaction rate up to an optimum, beyond which the enzyme denatures and activity drops sharply.
Q10 coefficient: For many enzymes, the rate doubles for every 10°C rise up to the optimum.
$Q_{10} = \frac{\text{rate at } (x+10)^{\circ}C}{\text{rate at } x^{\circ}C}$
pH
Each enzyme has an optimum pH. Deviations can reduce activity or denature the enzyme.
pH effects are often reversible unless extreme conditions cause denaturation.
Enzyme Inhibition
Reversible Inhibition
Reversible inhibitors bind non-covalently and can dissociate from the enzyme, restoring activity. Two main types:
Competitive Inhibition: Inhibitor resembles substrate and binds to the active site, competing with the substrate. Increases Km, but Vmax can be reached at high substrate concentrations.
Non-competitive Inhibition: Inhibitor binds to a site other than the active site, altering enzyme conformation. Lowers Vmax, but Km remains unchanged.
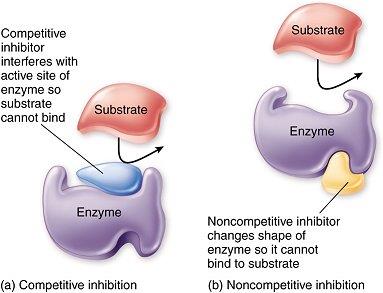
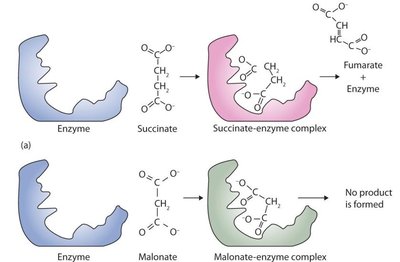
Irreversible Inhibition
Irreversible inhibitors bind covalently, permanently inactivating the enzyme (e.g., heavy metals disrupting disulfide bonds).
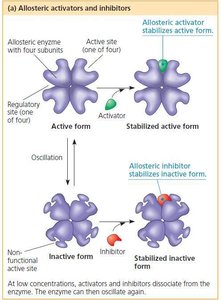
Allosteric Regulation of Enzymes
Allosteric Activation and Inhibition
Allosteric enzymes have multiple subunits and regulatory sites. Binding of an activator or inhibitor at an allosteric site stabilizes the enzyme in its active or inactive form, respectively. This regulation can be positive (activation) or negative (inhibition).
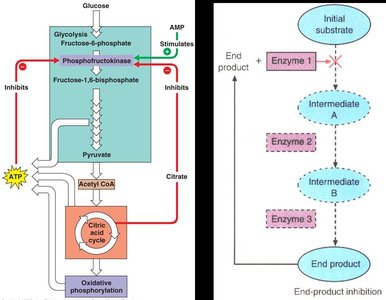
End-Product (Feedback) Inhibition
In metabolic pathways, the end product can inhibit an earlier enzyme, preventing overproduction. For example, ATP inhibits phosphofructokinase in glycolysis.
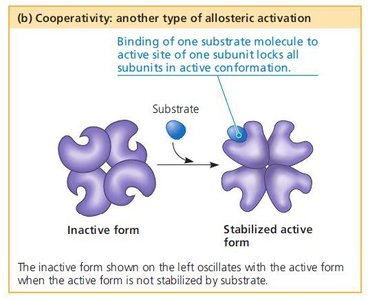
Cooperativity
Cooperativity is a form of allosteric regulation where binding of a substrate to one active site increases the affinity of other active sites for the substrate, enhancing enzyme activity (e.g., hemoglobin oxygen binding).
Additional info: These notes provide a comprehensive overview of enzyme structure, function, kinetics, regulation, and inhibition, suitable for college-level cell biology students preparing for exams.
