 Back
BackCytoskeletal Structure and Function: Microtubules, Microfilaments, and Intermediate Filaments
Study Guide - Smart Notes

Cytoskeletal Structure
Overview of Cytoskeletal Elements
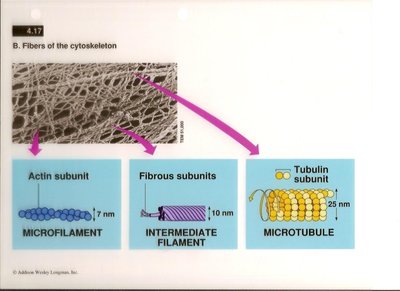
The cytoskeleton is a dynamic network of protein filaments that provides structural support, facilitates cellular movement, and organizes the contents of the cell. It consists of three main types of fibers: microfilaments, intermediate filaments, and microtubules, each with distinct protein subunits, diameters, and functions.
Microfilaments: Composed of actin subunits, approximately 7 nm in diameter.
Intermediate Filaments: Composed of fibrous proteins, approximately 10 nm in diameter.
Microtubules: Composed of tubulin subunits, approximately 25 nm in diameter.
Microtubules
Structure and Assembly
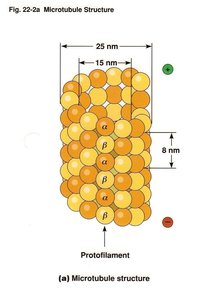
Microtubules are the largest cytoskeletal components, formed by polymerization of α- and β-tubulin dimers into hollow tubes. They are essential for maintaining cell shape, intracellular transport, and cell division.
Protofilaments: Linear chains of tubulin dimers that associate laterally to form the microtubule wall.
Microtubule diameter: 25 nm; wall thickness is about 15 nm.
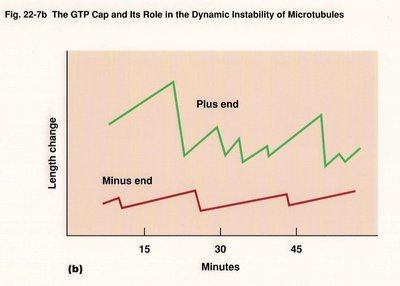
Polarity: Microtubules have a plus end (fast-growing) and a minus end (slow-growing).
Microtubule Assembly and Dynamics
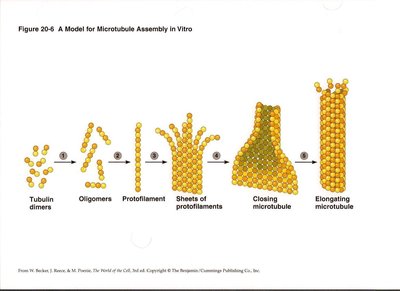
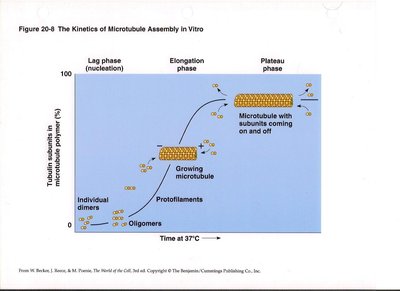
Microtubule assembly occurs in distinct phases: nucleation (lag phase), elongation, and plateau. Tubulin dimers first form oligomers, then protofilaments, which assemble into sheets and close into tubes. Microtubules exhibit dynamic instability, alternating between growth and shrinkage.
Nucleation: Formation of small tubulin oligomers.
Elongation: Addition of tubulin dimers to protofilaments.
Plateau: Steady-state with subunits coming on and off.
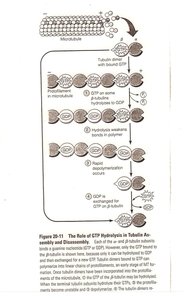
Role of GTP in Microtubule Dynamics
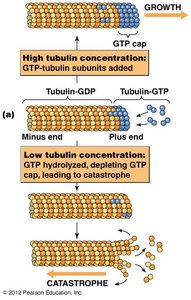
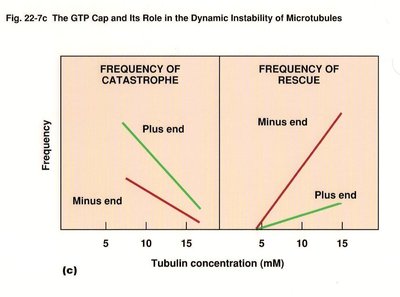
GTP binding and hydrolysis regulate microtubule assembly and disassembly. Tubulin dimers with GTP bind to the plus end, and GTP hydrolysis leads to instability and rapid depolymerization (catastrophe).
GTP cap: Stabilizes growing microtubule ends.
Catastrophe: Loss of GTP cap causes rapid shrinkage.
Rescue: Regaining GTP cap allows regrowth.

Microtubule-Binding Proteins
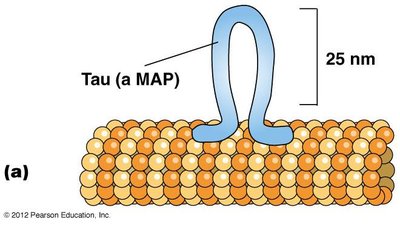
Microtubule-associated proteins (MAPs) regulate microtubule stability, bundling, and interactions with other cellular structures. Tau and MAP2 are examples of MAPs that control microtubule spacing and bundling in neurons.
Tau: Promotes tight bundling in axons.
MAP2: Promotes looser bundling in dendrites.
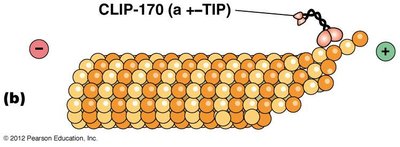
+–TIP proteins stabilize microtubule plus ends, decreasing the likelihood of catastrophic loss.
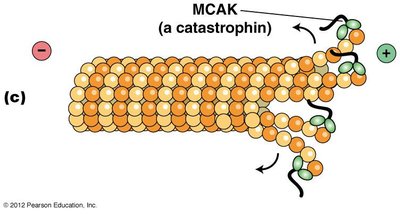
Catastrophins, such as MCAK, promote microtubule depolymerization at the ends.
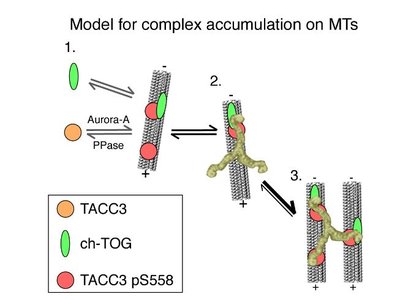
The Mesh Organelle and Microtubule Stabilization
The Mesh is a network of microtubule connectors that stabilizes kinetochore fibers during mitosis. Clathrin, TACC3, and ch-TOG proteins form complexes that bridge and stabilize adjacent microtubules, ensuring proper chromosome attachment and segregation.
Clathrin: Bridges between parallel microtubules.
TACC3: Recruits ch-TOG to microtubules; regulated by Aurora-A kinase.
Mutations: May cause cancer due to improper spindle stabilization.

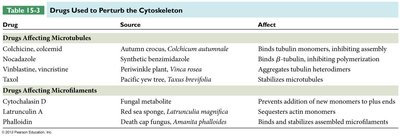
Drugs Affecting Microtubules
Several drugs target microtubules, affecting their polymerization and stability. These drugs are important in research and cancer therapy.
Drug | Source | Affect |
|---|---|---|
Colchicine, colcemid | Autumn crocus | Binds tubulin monomers, inhibiting assembly |
Nocodazole | Synthetic benzimidazole | Binds β-tubulin, inhibiting polymerization |
Vinblastine, vincristine | Periwinkle plant | Aggregates tubulin heterodimers |
Taxol | Pacific yew tree | Stabilizes microtubules |
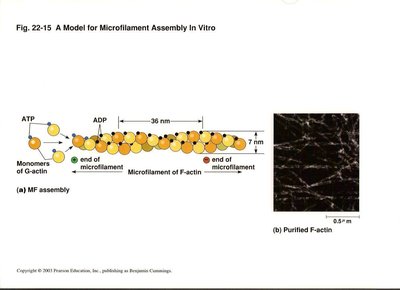
Microfilaments
Structure and Assembly
Microfilaments are the smallest cytoskeletal fibers, composed of actin. They are crucial for muscle contraction, cell migration, and maintenance of cell shape.
G-actin: Globular actin monomers that polymerize to form F-actin (filamentous actin).
Microfilament diameter: 7 nm.
Polarity: Plus (barbed) end and minus (pointed) end.
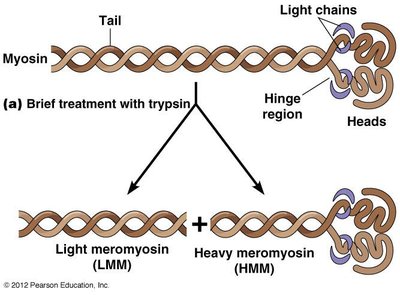
Microfilament Polarity and Decoration
Myosin S1 fragments bind to actin filaments, creating a distinctive arrowhead pattern that reveals filament polarity. The barbed end is the plus end, and the pointed end is the minus end.
Drugs Affecting Microfilaments
Specific drugs can perturb microfilament dynamics, affecting cell movement and structure.
Cytochalasin D: Prevents addition of new monomers to plus ends.
Latrunculin A: Sequesters actin monomers.
Phalloidin: Binds and stabilizes assembled microfilaments.

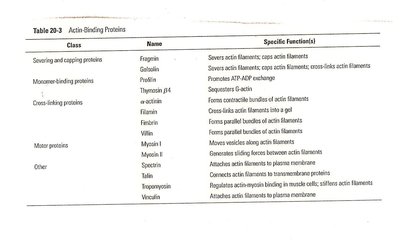
Actin-Binding Proteins
Actin-binding proteins regulate the polymerization, length, and organization of actin filaments. They control nucleation, elongation, severing, and association into networks.
Class | Name | Specific Functions |
|---|---|---|
Severing and capping proteins | Fragmin, gelsolin | Severs and caps actin filaments |
Monomer-binding proteins | Thymosin β4, profilin | Sequesters G-actin, promotes ATP-ADP exchange |
Cross-linking proteins | Filamin, α-actinin, fimbrin | Forms bundles and cross-links actin filaments |
Motor proteins | Myosin I, II | Generates sliding forces between actin filaments |
Other | Spectrin, tropomyosin, vinculin | Anchors actin filaments to membranes, attaches to plasma membrane |
Intermediate Filaments
Structure and Assembly
Intermediate filaments are the most stable and least soluble cytoskeletal components. They are not polarized and provide mechanical strength to cells and tissues.
Diameter: 10 nm.
Assembly: Involves formation of dimers, tetramers, protofilaments, and mature filaments.
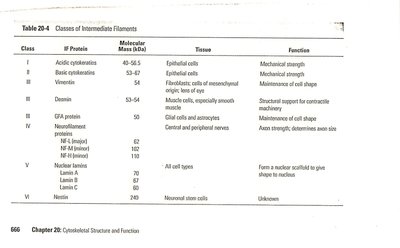
Classes of Intermediate Filament Proteins
IF proteins are tissue-specific and grouped into six classes:
Class | IF Protein | Tissue | Function |
|---|---|---|---|
I | Acidic keratins | Epithelial cells | Mechanical strength |
II | Basic keratins | Epithelial cells | Mechanical strength |
III | Vimentin, desmin, GFA protein | Connective tissue, muscle, glial cells | Maintenance of cell shape, contractile machinery, structural support |
IV | Neurofilament proteins | Nerve cells | Axon strength, diameter |
V | Lamin A, B, C | All cell types | Forms nuclear scaffold |
VI | Nestin | Neuronal stem cells | Unknown |
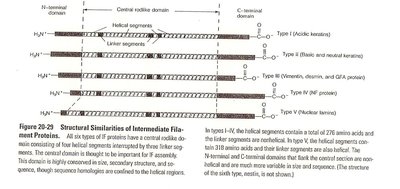
Structural Similarities of Intermediate Filament Proteins
All IF proteins have a central rod-like domain consisting of four helical segments, flanked by N- and C-terminal domains. This structure is conserved across IF types, though sequence homologies are confined to the helical regions.
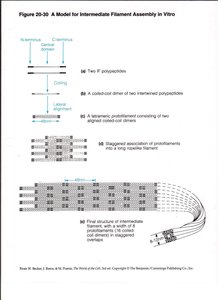
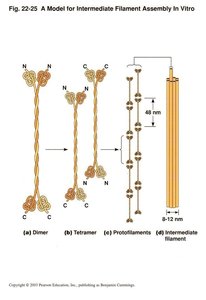
Assembly Model of Intermediate Filaments
IF assembly involves the formation of dimers, tetramers, protofilaments, and mature filaments. The staggered association of protofilaments results in a strong, rope-like structure.
Functional Integration of Cytoskeletal Elements
The cytoskeleton is a mechanically integrated structure. Microtubules resist compression, microfilaments generate tension, and intermediate filaments withstand tensile forces. Linker proteins such as plakins (e.g., plectin) connect these elements, ensuring structural integrity and dynamic remodeling.
Example: The nuclear lamina, composed of intermediate filaments, disassembles during mitosis and reassembles afterward, demonstrating the dynamic nature of the cytoskeleton.
----------------------------------------
