 Back
BackCytoskeletal Systems: Microfilaments, Intermediate Filaments, and Muscle Contraction
Study Guide - Smart Notes

Microfilaments
Structure and Function of Microfilaments
Microfilaments are the smallest cytoskeletal filaments, primarily composed of the protein actin. They are essential for various cellular processes, including muscle contraction, cell migration, amoeboid movement, and cytoplasmic streaming.
Cell Shape and Structure: Microfilaments help maintain and develop cell shape, especially at the cell cortex just beneath the plasma membrane.
Microvilli: They form the structural core of microvilli in epithelial cells.
Actin: The protein building block of microfilaments, actin exists as globular G-actin (binds ATP/ADP) and polymerizes to form filamentous F-actin.
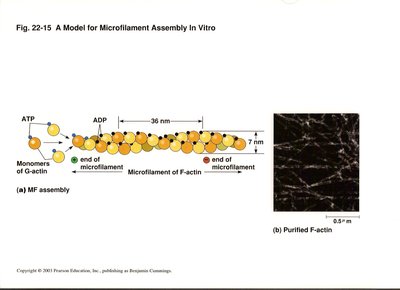
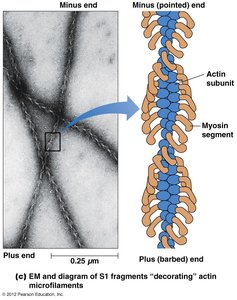
Polarity of Microfilaments
Microfilaments are polarized structures with distinct plus (barbed) and minus (pointed) ends. This polarity is revealed by the binding of myosin S1 fragments, which decorate actin filaments in a characteristic arrowhead pattern.
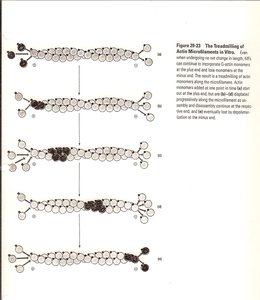
Assembly and Dynamics of Microfilaments
Microfilament assembly involves the polymerization of G-actin monomers into F-actin filaments. The process is regulated by ATP binding and hydrolysis, and the filaments exhibit dynamic behavior such as treadmilling, where subunits add at one end and dissociate from the other.
Treadmilling: A steady-state process where actin monomers add to the barbed end and dissociate from the pointed end, maintaining filament length but allowing turnover of subunits.
Drugs Affecting Microfilament Dynamics
Several compounds can modulate actin polymerization:
Cytochalasins: Fungal metabolites that prevent the addition of new actin monomers to filaments.
Latrunculin A: Sequesters actin monomers, preventing their incorporation into filaments.
Phalloidin: Stabilizes actin filaments and prevents their depolymerization.
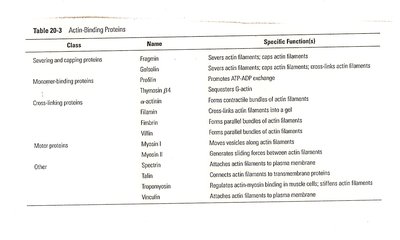
Actin-Binding Proteins
Cells regulate actin filament assembly, length, and organization using a variety of actin-binding proteins. These proteins control nucleation, elongation, severing, and cross-linking of filaments, as well as their association into networks.
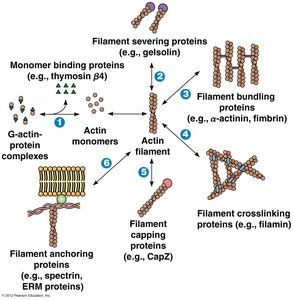
Class | Name | Specific Functions |
|---|---|---|
Severing and capping proteins | Fragmin, Gelsolin | Severs actin filaments; caps actin filaments |
Monomer-binding proteins | Profilin, Thymosin β4 | Promotes ATP-ADP exchange; sequesters G-actin |
Cross-linking proteins | α-actinin, Filamin, Fimbrin | Forms contractile bundles, gels, and parallel bundles of actin filaments |
Motor proteins | Myosin I, Myosin II | Generates sliding forces between actin filaments |
Other | Spectrin, Dystrophin, Talin, Vinculin | Anchors actin filaments to membrane or other structures |
Intermediate Filaments
Structure and Properties
Intermediate filaments (IFs) are the most stable and least soluble cytoskeletal components. Unlike microfilaments, they are not polarized. IFs provide mechanical strength to cells and tissues and are composed of various proteins, such as keratins, vimentin, desmin, and nuclear lamins.
Keratin: An abundant IF protein in epithelial cells and structures derived from skin.
Elasticity: IFs can withstand tensile forces, contributing to cellular integrity under stress.
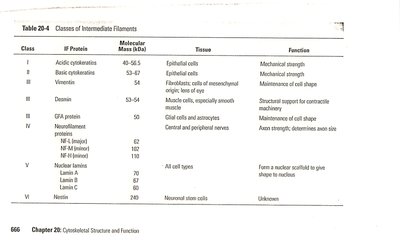
Classes of Intermediate Filament Proteins
IF proteins are tissue-specific and classified into six major classes:
Class | IF Proteins | Tissue | Function |
|---|---|---|---|
I | Acidic cytokeratins | Epithelial cells | Mechanical strength |
II | Basic cytokeratins | Epithelial cells | Mechanical strength |
III | Vimentin, Desmin | Connective tissue, muscle cells | Maintenance of cell shape, contractile machinery |
IV | Neurofilament proteins | Nerve cells | Axon strength, determines axon size |
V | Lamin A, B, C | All cell types | Form nuclear scaffold |
VI | Nestin | Neuronal stem cells | Unknown |
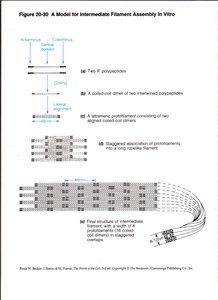
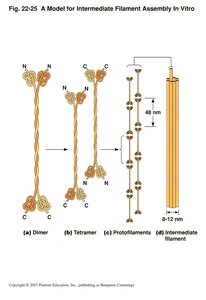
Assembly and Structure of Intermediate Filaments
IFs are composed of central α-helical rod domains flanked by variable N- and C-terminal domains. Assembly involves the formation of coiled-coil dimers, which associate into staggered tetramers, protofilaments, and finally mature IFs.
Functional Roles of Intermediate Filaments
Intermediate filaments confer mechanical strength on tissues and integrate with other cytoskeletal elements through linker proteins such as plakins (e.g., plectin). They are dynamically remodeled and play a role in nuclear structure (nuclear lamina).
Muscle Contraction and Thin Filaments
Thin Filaments: Structure and Regulatory Proteins
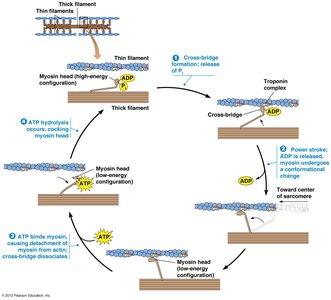
Thin filaments in muscle cells are composed of F-actin intertwined with regulatory proteins tropomyosin and troponin. Troponin is a complex of three polypeptides (TnT, TnC, TnI) and, together with tropomyosin, forms a calcium-sensitive switch that regulates muscle contraction.

Regulation of Muscle Contraction by Calcium
Muscle contraction is regulated by the concentration of calcium ions in the sarcoplasm. At low calcium levels, tropomyosin blocks myosin-binding sites on actin, preventing contraction. When calcium binds to TnC, tropomyosin shifts, exposing the binding sites and allowing contraction to occur.
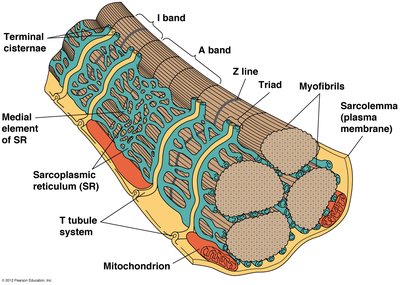
Neuromuscular Junction and Calcium Release
The neuromuscular junction is the site where a motor neuron communicates with a muscle cell, triggering contraction via an action potential. Acetylcholine released from the neuron depolarizes the muscle membrane, leading to calcium release from the sarcoplasmic reticulum (SR) and initiating contraction. Calcium is pumped back into the SR for muscle relaxation.
Summary Table: Cytoskeletal Elements and Functions
Element | Protein | Main Functions |
|---|---|---|
Microfilaments | Actin | Cell shape, movement, muscle contraction |
Intermediate Filaments | Keratin, vimentin, desmin, lamins | Mechanical strength, nuclear structure |
Additional info: The cytoskeleton is a dynamic and integrated network, with microfilaments, intermediate filaments, and microtubules working together to maintain cell structure, facilitate movement, and regulate intracellular transport.
