 Back
BackCytoskeletal Systems: Structure, Function, and Regulation
Study Guide - Smart Notes

Cytoskeletal Systems
Overview of Cytoskeletal Elements
The cytoskeleton is a complex network of protein filaments that provides structural support, organization, and dynamic movement within eukaryotic cells. It consists of three main types of filaments: microtubules, microfilaments (actin filaments), and intermediate filaments. These elements are interconnected, highly structured, and dynamic, enabling cells to maintain shape, move, and transport materials.
Microtubules (MTs): Largest and thickest cytoskeletal filaments, composed of tubulin heterodimers.
Microfilaments (MFs): Smallest and thinnest, primarily composed of actin.
Intermediate Filaments (IFs): Intermediate in size, provide mechanical strength and are tissue-specific.
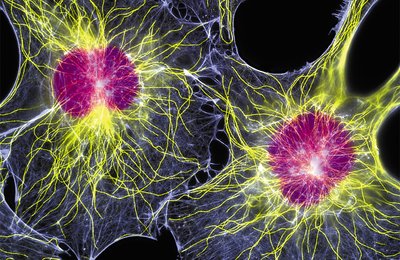
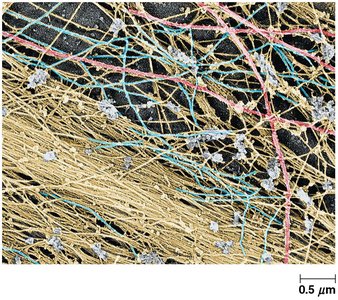
Microtubules
Structure and Assembly
Microtubules are straight, hollow cylinders typically composed of 13 longitudinal arrays of polymers called protofilaments. The basic subunit is a heterodimer of α-tubulin and β-tubulin, which bind noncovalently to form stable αβ-heterodimers.
Microtubules can exist as singlets (13 protofilaments), doublets (cilia and flagella), or triplets (basal bodies and centrioles).
Doublets and triplets contain one complete (A) tubule and one or two incomplete (B and C) tubules.

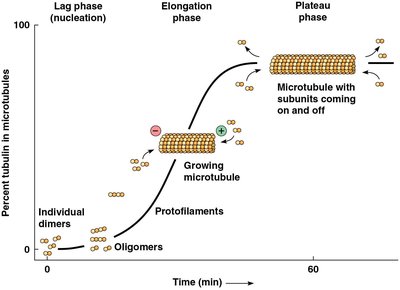
Kinetics of Microtubule Assembly
Microtubule assembly follows three phases: lag (nucleation), elongation, and plateau (equilibrium). The critical concentration is the tubulin heterodimer concentration at which assembly and disassembly are balanced.
Lag phase: Slow assembly of dimers into oligomers and protofilaments.
Elongation phase: Rapid growth of established microtubules.
Plateau phase: Structural equilibrium as free tubulin concentration falls.
Equation:
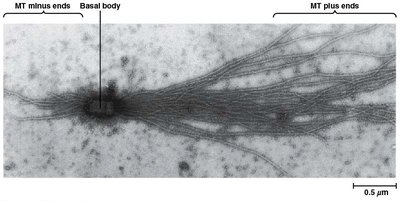
Microtubule Polarity and Growth
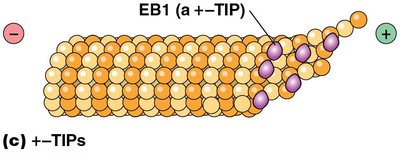
Microtubules have distinct plus and minus ends. Addition of tubulin dimers occurs more rapidly at the plus end, which is often visualized in vitro using basal bodies.
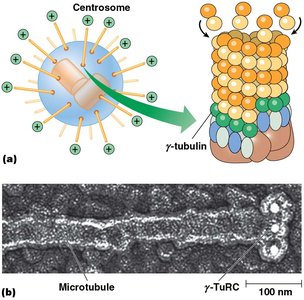
Microtubule-Organizing Centers (MTOCs) and Centrosomes
Microtubules originate from MTOCs, with the centrosome being the primary MTOC in animal cells. Centrosomes contain centrioles and pericentriolar material, including γ-tubulin ring complexes (γ-TuRCs) that nucleate microtubule assembly.
Centrioles: Walls formed by nine pairs of triplet microtubules, oriented at right angles.
γ-Tubulin: Essential for nucleation; loss prevents microtubule formation.
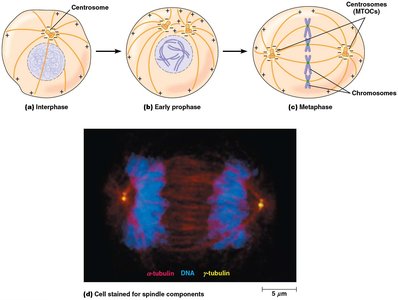
Microtubules in Cell Division
During cell division, MTOCs organize the mitotic spindle, which is essential for chromosome segregation. Microtubule dynamics are tightly regulated during the cell cycle.
Regulation of Microtubule Stability
Microtubule stability is regulated by various proteins and drugs:
Antimitotic drugs: Colchicine and nocodazole inhibit assembly; taxol stabilizes microtubules.
Microtubule-associated proteins (MAPs): Stabilize and bundle microtubules (e.g., Tau, MAP2).
+–TIP proteins: Stabilize growing plus ends (e.g., EB1).
Catastrophins: Promote depolymerization and regulate spindle shortening.
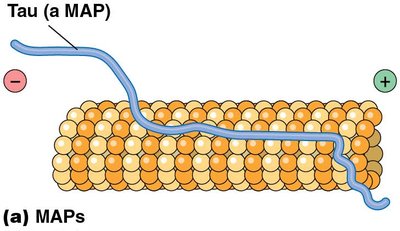
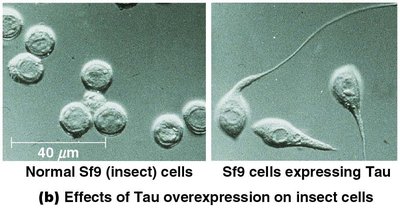
Microfilaments (Actin Filaments)
Structure and Function
Microfilaments are the smallest cytoskeletal filaments, composed of actin. They play key roles in muscle contraction, cell migration, and maintenance of cell shape, especially at the cell cortex.
Actin exists as G-actin (globular) and polymerizes into F-actin (filamentous).
F-actin filaments are double helical strands with intrinsic polarity.
Specialized Actin Structures
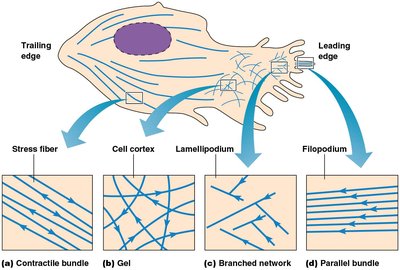
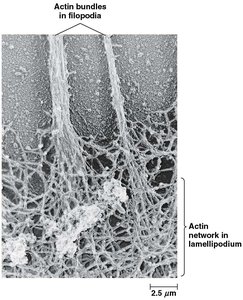
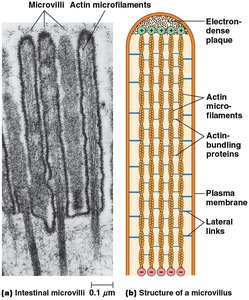
Actin forms specialized structures such as microvilli, lamellipodia, and filopodia, which are important for cell movement and increasing surface area.
Microvilli: Tight bundles of actin filaments, crosslinked by proteins like fimbrin and villin.
Lamellipodia: Branched actin networks at the leading edge of crawling cells.
Filopodia: Parallel actin bundles oriented toward the tip of protrusions.
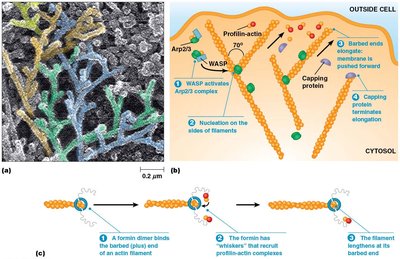
Regulation of Actin Assembly
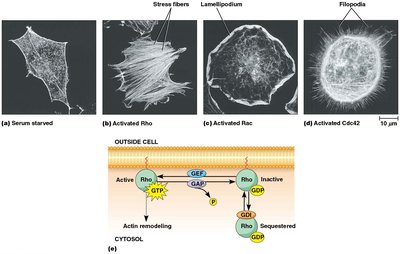
Actin assembly is regulated by plasma membrane phospholipids (e.g., PIP2) and Rho family GTPases (Rho, Rac, Cdc42), which control the formation of stress fibers, lamellipodia, and filopodia.
Rho: Promotes stress fiber formation.
Rac: Promotes lamellipodia extension.
Cdc42: Promotes filopodia formation.
Regulation involves GEFs (activate), GAPs (inactivate), and GDIs (sequester).
Intermediate Filaments
Structure and Function
Intermediate filaments are the most stable and least soluble cytoskeletal components, providing mechanical strength and supporting the cytoskeleton. They are abundant in animal cells and tissue-specific.
Keratin is a major IF in epithelial cells.
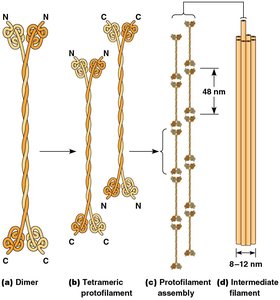
IFs are composed of fibrous proteins with a central rodlike domain.
Assembly involves dimer formation, lateral association into tetramers, and stacking into filaments.
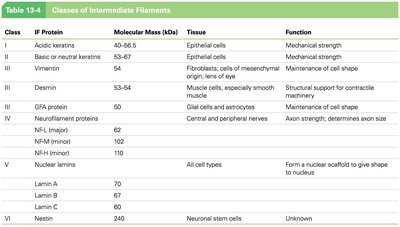
Classes of Intermediate Filaments
IF proteins are grouped into six classes based on their tissue distribution and function.
Class | IF Protein | Tissue | Function |
|---|---|---|---|
I | Acidic keratins | Epithelial cells | Mechanical strength |
II | Basic or neutral keratins | Epithelial cells | Mechanical strength |
III | Vimentin, Desmin, GFAP | Fibroblasts, muscle cells, glial cells | Maintenance of cell shape, structural support |
IV | Neurofilament proteins | Nerves | Axon strength |
V | Nuclear lamins | All cell types | Nuclear scaffold |
VI | Nestin | Neuronal stem cells | Unknown |
Integration of Cytoskeletal Components
Mechanical Cooperation and Linker Proteins
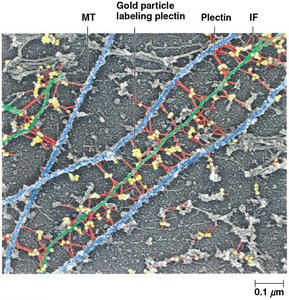
The cytoskeleton is mechanically integrated, with microtubules resisting compression, microfilaments generating tension, and intermediate filaments withstanding tensile forces. Linker proteins such as spectraplakins (e.g., plectin) connect all three filament types, facilitating coordinated cellular responses.
Example: Cancer diagnosis can utilize tissue-specific intermediate filament proteins to trace the origin of metastasizing tumors.
Additional info: The cytoskeleton is essential for cell division, motility, intracellular transport, and maintaining cell integrity. Disruption of cytoskeletal elements can lead to disease states, including cancer and neurodegenerative disorders.
