 Back
BackCh 10b - Electron Transport Chain and Oxidative Phosphorylation
Study Guide - Smart Notes

Electron Transport Chain (ETC) and Oxidative Phosphorylation
Overview of Electron Transport and Energy Yield
The electron transport chain (ETC) is a series of protein complexes and electron carriers located in the inner mitochondrial membrane. It is the final stage of aerobic respiration, responsible for the majority of ATP production in cells. While glycolysis and the citric acid cycle generate a small amount of ATP directly, most of the energy from glucose is stored in the reduced coenzymes NADH and FADH2, which are oxidized in the ETC to drive ATP synthesis.
Electron transport refers to the transfer of electrons from NADH and FADH2 to oxygen, the terminal electron acceptor, forming water.
This process is highly exergonic, releasing free energy that is used to pump protons and generate an electrochemical gradient.
ATP synthesis is tightly coupled to electron transport via this proton gradient, a process known as oxidative phosphorylation.
Major Electron Carriers in the ETC
The ETC consists of a series of electron carriers, each with distinct properties and roles:
Flavoproteins: Contain FAD or FMN as prosthetic groups; transfer both electrons and protons (e.g., NADH dehydrogenase, succinate dehydrogenase).
Iron-sulfur proteins: Contain Fe-S centers; transfer one electron at a time, no protons.
Cytochromes: Contain heme groups with iron; transfer one electron at a time, no protons. Types include b, c, c1, a, and a3.
Copper-containing cytochromes: Cytochromes a and a3 also contain copper, forming a bimetallic Fe-Cu center crucial for oxygen binding and reduction.
Coenzyme Q (ubiquinone): The only non-protein carrier; mobile within the membrane, accepts and donates both electrons and protons, essential for proton translocation.
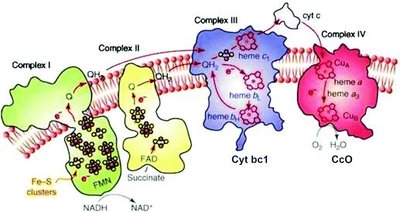
Organization of the Electron Transport Chain
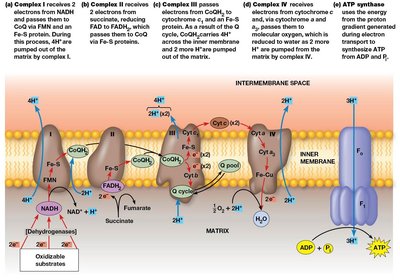
The ETC is organized into four large multiprotein complexes embedded in the inner mitochondrial membrane, with most electron carriers grouped into these complexes:
Complex I (NADH–CoQ oxidoreductase): Transfers electrons from NADH to CoQ, pumps 4 protons per 2 electrons.
Complex II (Succinate–CoQ oxidoreductase): Transfers electrons from succinate (via FADH2) to CoQ, does not pump protons.
Complex III (CoQ–cytochrome c oxidoreductase): Transfers electrons from CoQH2 to cytochrome c, pumps 4 protons per 2 electrons.
Complex IV (Cytochrome c oxidase): Transfers electrons from cytochrome c to O2, reducing it to water, pumps 2 protons per 2 electrons.
Detailed Function of Each Complex
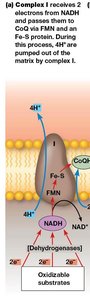
Complex I: NADH–CoQ Oxidoreductase
Complex I receives electrons from NADH, transferring them to FMN and then through a series of Fe-S centers to CoQ. This process is coupled to the translocation of 4 protons from the mitochondrial matrix to the intermembrane space.
Reaction:
Proton pumping: 4 H+ per 2 electrons
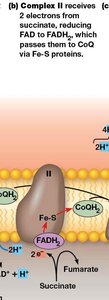
Complex II: Succinate–CoQ Oxidoreductase
Complex II receives electrons from succinate via FAD, forming FADH2. Electrons are passed through Fe-S centers to CoQ. Unlike other complexes, Complex II does not pump protons.
Reaction:
Proton pumping: None
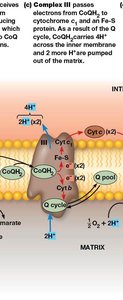
Complex III: CoQ–Cytochrome c Oxidoreductase
Complex III transfers electrons from CoQH2 to cytochrome c via the Q cycle, involving cytochromes b and c1 and Fe-S proteins. This process pumps 4 protons per 2 electrons.
Reaction:
Proton pumping: 4 H+ per 2 electrons
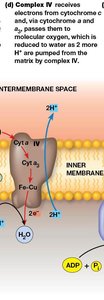
Complex IV: Cytochrome c Oxidase
Complex IV receives electrons from cytochrome c and transfers them to molecular oxygen, reducing it to water. This complex contains both heme and copper centers, and pumps 2 protons per 2 electrons.
Reaction:
Proton pumping: 2 H+ per 2 electrons
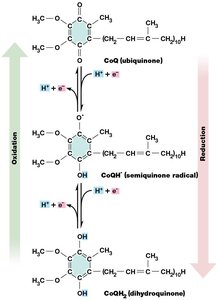
Coenzyme Q (Ubiquinone) and Its Redox States
Coenzyme Q (CoQ) is a lipid-soluble electron carrier that shuttles electrons between complexes I/II and III. It can exist in three redox states: oxidized (ubiquinone), semiquinone (radical), and fully reduced (ubiquinol). CoQ also plays a vital role in proton translocation across the membrane.
Summary Table: Electron Transport Chain Complexes
Complex | Main Function | Electron Donor | Electron Acceptor | Proton Pumping |
|---|---|---|---|---|
I | NADH to CoQ | NADH | CoQ | Yes (4 H+) |
II | Succinate to CoQ | FADH2 | CoQ | No |
III | CoQH2 to cytochrome c | CoQH2 | cytochrome c | Yes (4 H+) |
IV | cytochrome c to O2 | cytochrome c | O2 | Yes (2 H+) |
Electrochemical Proton Gradient and ATP Synthesis
The energy released during electron transport is used to pump protons from the mitochondrial matrix to the intermembrane space, creating an electrochemical proton gradient (proton-motive force). This gradient drives the synthesis of ATP by ATP synthase as protons flow back into the matrix.
Oxidative phosphorylation refers to ATP synthesis powered by the transfer of electrons to oxygen.
ATP synthesis and electron transport are tightly coupled; inhibitors or uncouplers can disrupt this relationship.
Regulation and Inhibition of the ETC
Certain chemicals, such as cyanide and azide, inhibit the ETC by binding to the Fe-Cu center of cytochrome c oxidase (Complex IV), blocking electron transfer to oxygen. Incomplete reduction of oxygen at complexes I and III can generate reactive oxygen species (ROS) like superoxide anion (O2–) and hydrogen peroxide (H2O2), which contribute to cellular aging and oxidative stress.
Key Equations
Overall reaction for NADH oxidation:
Overall reaction for FADH2 oxidation:
Summary
The ETC is essential for aerobic energy metabolism, maximizing ATP yield from glucose.
Electron flow through the ETC is coupled to proton pumping, generating a gradient used for ATP synthesis.
Disruption of the ETC can have severe cellular consequences, including energy failure and oxidative damage.
