Back
Back16: Energy Conversion in Mitochondria: The Electron Transport Chain and Oxidative Phosphorylation
Study Guide - Smart Notes

Energy Conversion in Cells
Introduction to Biological Energy Conversion
Cells convert energy from nutrients into usable forms through highly regulated biochemical pathways. The process of oxidative phosphorylation in mitochondria is central to this conversion, efficiently capturing energy in the form of ATP.
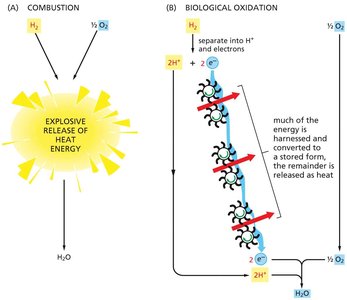
Biological oxidation stores about 50% of released energy in a useful form, with the remainder lost as heat.
In contrast, combustion of hydrogen releases nearly all energy as heat, with little to no energy captured for cellular work.
The Mitochondrial Electron Transport Chain (ETC)
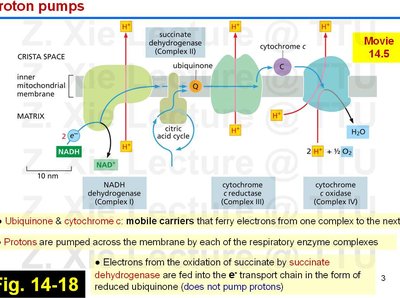
Overview of the Electron Transport Chain
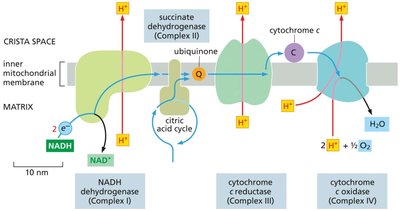
The ETC is a series of protein complexes and mobile carriers embedded in the inner mitochondrial membrane. It transfers electrons from NADH and FADH2 to oxygen, pumping protons to generate a proton gradient used for ATP synthesis.
Major complexes: NADH dehydrogenase (Complex I), succinate dehydrogenase (Complex II), cytochrome c reductase (Complex III), and cytochrome c oxidase (Complex IV).
Mobile carriers: Ubiquinone (Q) and cytochrome c shuttle electrons between complexes.
Protons (H+) are pumped from the matrix to the intermembrane space, creating an electrochemical gradient.
Mobile Electron Carriers and Proton Pumps
Electron flow through the ETC is coupled to proton pumping, which is essential for ATP synthesis.
Ubiquinone and cytochrome c are mobile carriers that transfer electrons between complexes.
Electrons from succinate (via succinate dehydrogenase) enter the chain as reduced ubiquinone but do not contribute to proton pumping at Complex II.
Each respiratory enzyme complex pumps protons across the membrane, except Complex II.
Electron Carriers in the ETC
Iron-Sulfur (Fe-S) Clusters
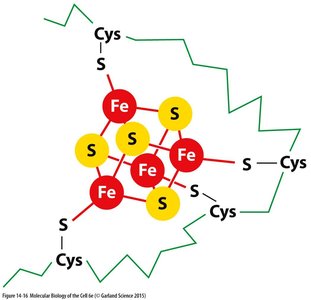
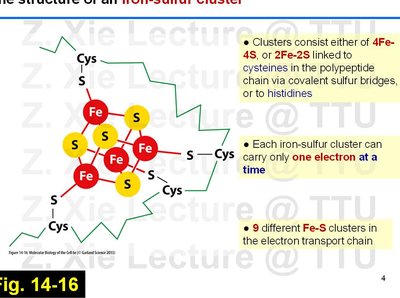
Iron-sulfur clusters are prosthetic groups found in several ETC complexes, facilitating single-electron transfers.
Types: 4Fe-4S and 2Fe-2S clusters, coordinated by cysteine residues or histidines in proteins.
Each cluster can carry only one electron at a time.
There are nine different Fe-S clusters in the ETC.
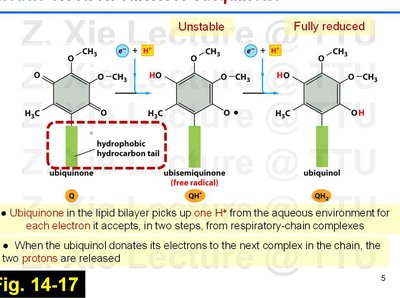
Ubiquinone (Coenzyme Q)
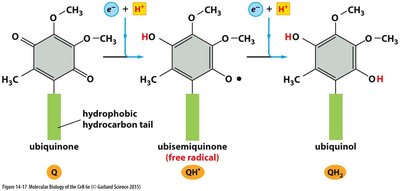
Ubiquinone is a lipid-soluble electron carrier that shuttles electrons and protons within the inner mitochondrial membrane.
Accepts electrons in two steps, forming a semiquinone intermediate before becoming fully reduced to ubiquinol (QH2).
For each electron accepted, one proton is taken up from the aqueous environment; two protons are released when electrons are donated to the next complex.
Cytochromes
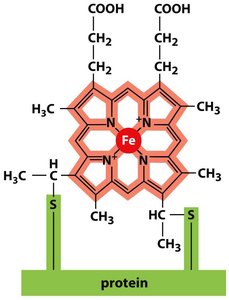
Cytochromes are heme-containing proteins that transfer electrons via reversible oxidation and reduction of their iron atom.
There are six different cytochromes in the respiratory chain, each with a unique electron affinity.
The heme group consists of a porphyrin ring with a central iron atom, covalently attached to the protein.
Redox Potential and Proton Pumping
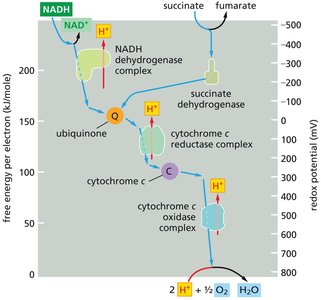
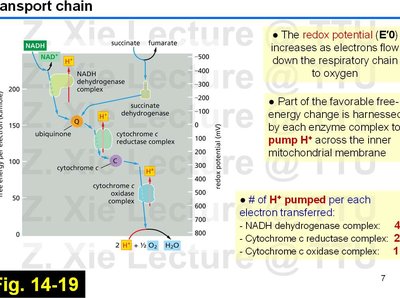
Redox Potential Changes Along the ETC
As electrons move through the ETC, the redox potential (E′0) increases, driving the transfer of electrons to oxygen, the terminal electron acceptor.
Each enzyme complex harnesses part of the free-energy change to pump protons across the inner mitochondrial membrane.
Number of protons pumped per electron transferred:
NADH dehydrogenase complex: 4 H+
Cytochrome c reductase complex: 2 H+
Cytochrome c oxidase complex: 1 H+
Structure and Function of ETC Complexes
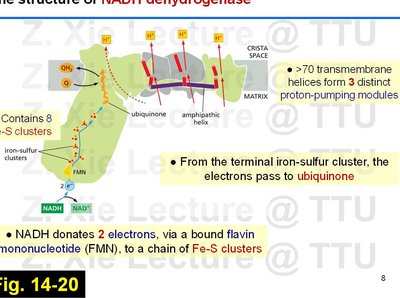
NADH Dehydrogenase (Complex I)
Complex I is the largest ETC complex, catalyzing electron transfer from NADH to ubiquinone and pumping protons across the membrane.
Contains 8 Fe-S clusters and over 70 transmembrane helices forming three proton-pumping modules.
NADH donates two electrons via flavin mononucleotide (FMN) to the Fe-S clusters, which then pass electrons to ubiquinone.
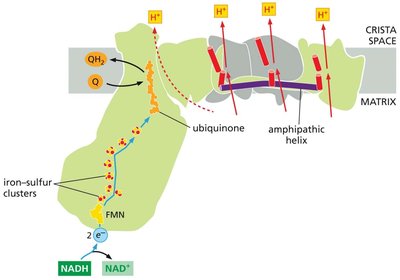
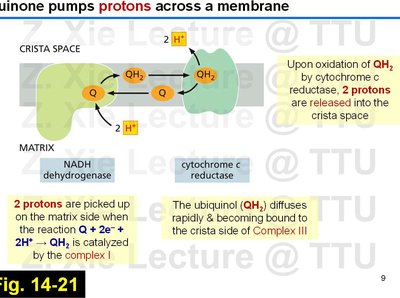
Quinone-Mediated Proton Pumping
Quinones play a key role in coupling electron transfer to proton translocation across the membrane.
Two protons are picked up on the matrix side during the reduction of Q to QH2 by Complex I.
Upon oxidation of QH2 by cytochrome c reductase (Complex III), two protons are released into the crista space.
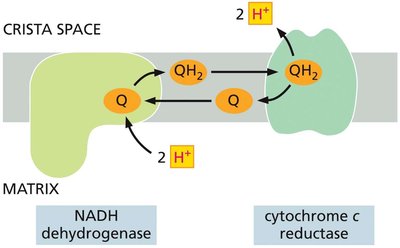
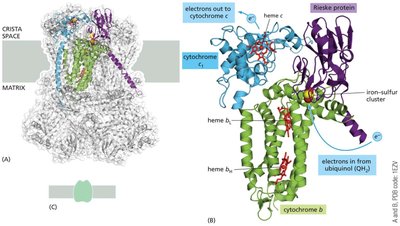
Cytochrome c Reductase (Complex III)
Complex III transfers electrons from ubiquinol to cytochrome c and contributes to the proton gradient via the Q-cycle mechanism.
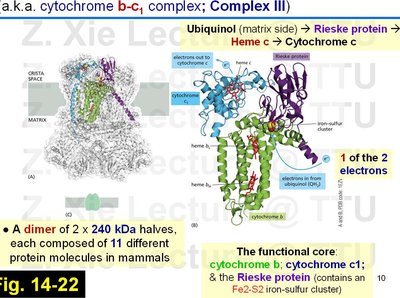
Composed of two identical halves, each with 11 different protein molecules in mammals.
Functional core includes cytochrome b, cytochrome c1, and the Rieske protein (with a Fe2-S2 cluster).
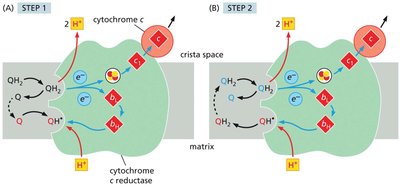
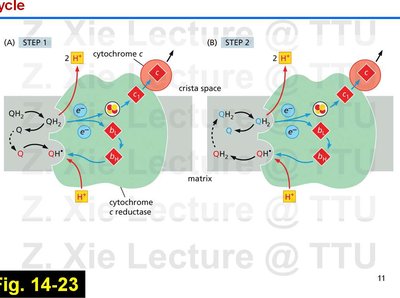
The Q-Cycle Mechanism
The Q-cycle describes the two-step process by which electrons from QH2 are transferred through Complex III, facilitating proton translocation.
Involves sequential oxidation and reduction of ubiquinone and ubiquinol, with protons released into the intermembrane space.
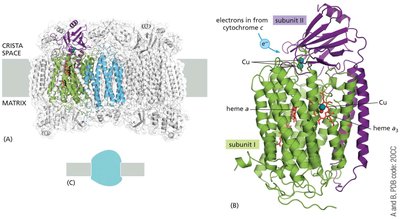
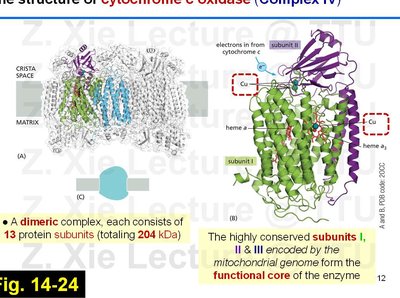
Cytochrome c Oxidase (Complex IV)
Complex IV is the terminal enzyme of the ETC, transferring electrons from cytochrome c to molecular oxygen, reducing it to water.
Dimeric complex, each half with 13 protein subunits (204 kDa total).
Subunits I, II, and III (encoded by mitochondrial DNA) form the functional core.