 Back
BackGlycolysis, Fermentation, and Gluconeogenesis: Pathways and Regulation
Study Guide - Smart Notes

Pyruvate as a Branchpoint in Metabolism
Fate of Pyruvate: Aerobic vs. Anaerobic Conditions
Pyruvate, the end product of glycolysis, serves as a critical metabolic branchpoint. Its fate depends on the presence or absence of oxygen:
Aerobic Respiration: In the presence of oxygen, pyruvate enters the mitochondria, where it is converted to acetyl-CoA and further oxidized in the tricarboxylic acid (TCA) cycle and electron transport chain (ETC), generating large amounts of ATP.
Anaerobic Respiration (Fermentation): In the absence of oxygen, pyruvate undergoes fermentation to regenerate NAD+, allowing glycolysis to continue. This process yields less ATP.
Fermentation Pathways
Purpose and Types of Fermentation
The main goal of fermentation is to oxidize NADH back to NAD+ for glycolysis. No additional ATP is produced beyond that generated in glycolysis. There are several types of fermentation:
Lactate Fermentation: Pyruvate is reduced to lactate by lactate dehydrogenase, regenerating NAD+. This occurs in muscle cells and certain bacteria.
Ethanol Fermentation: Pyruvate is first decarboxylated to acetaldehyde, then reduced to ethanol, regenerating NAD+. This is common in yeast and some plant cells.
Other Types: Propionate (used in Swiss cheese production) and butylene glycol (associated with food spoilage).
The Warburg Effect and Cancer Metabolism
Some cancer cells preferentially use glycolysis and fermentation even in the presence of oxygen (aerobic glycolysis), a phenomenon known as the Warburg effect. These cells increase glucose uptake and use glycolytic intermediates for anabolic processes, supporting rapid growth and proliferation.
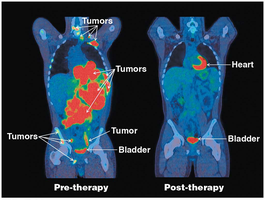
Example: PET scans use radiolabeled glucose analogues to detect areas of high glycolytic activity, such as tumors.
Glycolytic Substrates and Entry Points
Monosaccharides and Polysaccharides
Glycolysis can utilize a variety of substrates beyond glucose:
Hexoses (e.g., fructose, galactose): These sugars are phosphorylated and isomerized to enter glycolysis as intermediates.
Pentoses: Enter glycolysis via the pentose phosphate pathway, which converts them into glycolytic intermediates.
Disaccharides and Polysaccharides: Disaccharides are cleaved into monosaccharides, while polysaccharides like starch and glycogen are broken down by phosphorolysis.
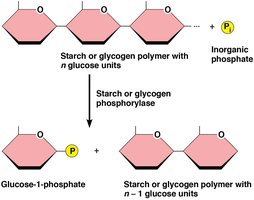
Phosphorolysis of Storage Carbohydrates
Phosphorolysis is the cleavage of a bond using inorganic phosphate instead of water, releasing glucose-1-phosphate from glycogen or starch. This is catalyzed by glycogen phosphorylase or starch phosphorylase.
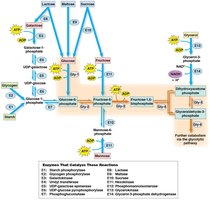
Entry of Various Sugars into Glycolysis
Different sugars are converted into glycolytic intermediates by specific enzymes, allowing them to feed into the pathway at various points.
Gluconeogenesis
Overview and Importance
Gluconeogenesis is the anabolic process of synthesizing glucose from non-carbohydrate precursors such as pyruvate, lactate, and certain amino acids. This pathway is essential in animals (mainly in the liver and kidneys) for maintaining blood glucose levels during fasting or intense exercise.
It reverses most steps of glycolysis but bypasses the highly exergonic, irreversible steps using unique enzymes.
Gluconeogenesis consumes 4 ATP and 2 GTP per glucose molecule synthesized.
Key Steps of Gluconeogenesis
Pyruvate to Phosphoenolpyruvate (PEP): A two-step reaction involving pyruvate carboxylase and PEP carboxykinase. CO2 is added and then removed, and ATP/GTP are consumed.
Fructose-1,6-bisphosphate (F1,6BP) to Fructose-6-phosphate (F6P): Catalyzed by fructose-1,6-bisphosphatase, this step involves hydrolysis to release inorganic phosphate.
Glucose-6-phosphate (G6P) to Glucose: Catalyzed by glucose-6-phosphatase, another hydrolysis step releases inorganic phosphate.
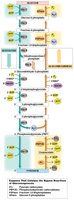
The Cori Cycle
Lactate produced by anaerobic glycolysis in muscle is transported to the liver, where it is converted back to pyruvate and then to glucose via gluconeogenesis. This glucose can then return to muscle tissue, completing the Cori cycle.
Regulation of Glycolysis and Gluconeogenesis
Spatial and Enzymatic Regulation
Glycolysis and gluconeogenesis are reciprocally regulated to prevent futile cycling. Glycolysis primarily occurs in muscle and other tissues, while gluconeogenesis is mainly in the liver. Regulation focuses on unique enzymes in each pathway:
Glycolysis: Hexokinase, phosphofructokinase-1 (PFK-1), pyruvate kinase
Gluconeogenesis: Pyruvate carboxylase, PEP carboxykinase, fructose-1,6-bisphosphatase, glucose-6-phosphatase
Allosteric Regulation
Key metabolites exert opposite effects on glycolysis and gluconeogenesis:
AMP: Stimulates glycolysis, inhibits gluconeogenesis
Acetyl-CoA: Inhibits glycolysis, stimulates gluconeogenesis
Fructose-2,6-bisphosphate (F2,6BP): Stimulates glycolysis, inhibits gluconeogenesis
Regulation of Phosphofructokinase-1 (PFK-1) and ATP
ATP acts as both a substrate and an allosteric inhibitor of PFK-1. At high concentrations, ATP binds to an allosteric site, reducing the enzyme's activity and slowing glycolysis.
Phosphofructokinase-2 (PFK-2) and Fructose-2,6-bisphosphatase
PFK-2 is a bifunctional enzyme with both kinase and phosphatase activities:
Unphosphorylated: Kinase activity converts F6P to F2,6BP, stimulating glycolysis.
Phosphorylated: Phosphatase activity converts F2,6BP back to F6P, stimulating gluconeogenesis.
cAMP-dependent protein kinase regulates the phosphorylation state of PFK-2, integrating hormonal signals with metabolic control.
Summary Table: Key Regulatory Molecules
Molecule | Effect on Glycolysis | Effect on Gluconeogenesis |
|---|---|---|
AMP | Stimulates | Inhibits |
Acetyl-CoA | Inhibits | Stimulates |
F2,6BP | Stimulates | Inhibits |
ATP (on PFK-1) | Inhibits (allosteric) | — |
Key Takeaways
Fermentation regenerates NAD+ for glycolysis but does not produce additional ATP.
Cancer cells often favor glycolysis and fermentation (Warburg effect).
Various sugars can enter glycolysis as intermediates.
Gluconeogenesis bypasses irreversible glycolytic steps using unique enzymes and is energetically costly.
Fructose-2,6-bisphosphate is a central regulator of glycolysis and gluconeogenesis.
