Back
BackMacromolecules of the Cell: Nucleic Acids, Polysaccharides, and Lipids
Study Guide - Smart Notes

Macromolecules of the Cell
Nucleic Acids: DNA and RNA
Nucleic acids are essential informational macromolecules in cells, responsible for the storage, transmission, and expression of genetic information. The two main types are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
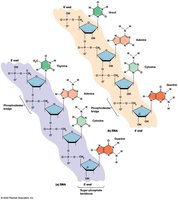
Structure: Nucleic acids are linear polymers of nucleotides linked by 3′,5′ phosphodiester bridges. Each nucleotide consists of a phosphate group, a five-carbon sugar (ribose in RNA, deoxyribose in DNA), and a nitrogenous base.
Directionality: Polynucleotides have a 5′ phosphate group at one end and a 3′ hydroxyl group at the other. Sequences are written 5′ to 3′.
Nucleic Acid Synthesis and Base Pairing
Nucleic acid synthesis relies on a template strand to ensure correct nucleotide addition. Complementary base pairing is fundamental to nucleic acid structure and function.
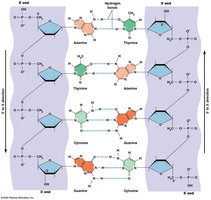
Base Pairing: Adenine (A) pairs with Thymine (T) in DNA (or Uracil (U) in RNA) via two hydrogen bonds; Guanine (G) pairs with Cytosine (C) via three hydrogen bonds.
Template-Directed Synthesis: New nucleotides are added according to the sequence of the template strand, ensuring fidelity in replication and transcription.
The DNA Double Helix
DNA is typically found as a double-stranded helix, a structure first described by Watson and Crick in 1953. This model explained DNA's physical and chemical properties and suggested a mechanism for replication.

Structure of Double-Stranded DNA
Antiparallel Strands: The two DNA strands run in opposite directions (5′ to 3′ and 3′ to 5′).
Complementarity: Each strand serves as a template for the other, with specific base pairing.
Backbone: Composed of alternating sugar and phosphate groups.
Forms: B-DNA is the main cellular form; A-DNA and Z-DNA are alternative conformations.
RNA Structure and Base Pairing
RNA is usually single-stranded but can form complex secondary structures through intramolecular base pairing. These pairings are less extensive than in DNA and often occur between different regions of the same molecule.
Polysaccharides
Overview of Polysaccharides
Polysaccharides are long-chain polymers of sugars and sugar derivatives. Unlike nucleic acids, they are not informational molecules but serve primarily structural and storage roles in cells.
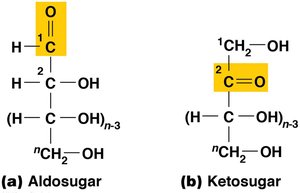
Monomers: The repeating units are monosaccharides, which can be aldosugars (terminal carbonyl group) or ketosugars (internal carbonyl group).
Classification by Carbon Number: Trioses (3C), Tetroses (4C), Pentoses (5C), Hexoses (6C), Heptoses (7C).
Glucose: The Central Monosaccharide
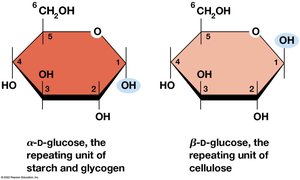
D-glucose (C6H12O6) is the most common monosaccharide. It exists in both linear (Fischer projection) and ring (Haworth projection) forms, with the ring form predominating in cells.
Ring Forms: The ring closure at carbon 1 creates two anomers: α (hydroxyl down) and β (hydroxyl up).
Disaccharides
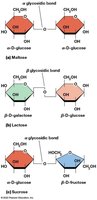
Disaccharides are formed by covalent linkage of two monosaccharides via a glycosidic bond (elimination of water). The type of glycosidic bond (α or β) depends on the configuration of the anomeric carbon involved.
Maltose: Two glucose units, α glycosidic bond.
Lactose: Glucose + galactose, β glycosidic bond.
Sucrose: Glucose + fructose, α glycosidic bond.
Storage Polysaccharides: Starch and Glycogen
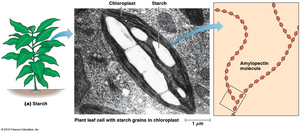
Starch (plants) and glycogen (animals, bacteria) are storage polysaccharides composed of α-D-glucose units linked by α(1→4) glycosidic bonds, with branching via α(1→6) bonds.
Glycogen: Highly branched, stored in liver and muscle cells.
Starch: Mixture of unbranched amylose and branched amylopectin, stored in plastids (chloroplasts and amyloplasts).
Structural Polysaccharides: Cellulose and Others
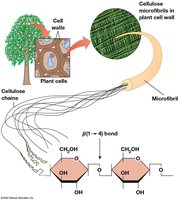
Cellulose is the main structural polysaccharide in plant cell walls, composed of β-D-glucose units linked by β(1→4) bonds, forming rigid microfibrils. Other structural polysaccharides include chitin (in fungi and arthropods) and peptidoglycan (in bacteria).
Cellulose: Not digestible by mammals due to β(1→4) linkage.
Chitin: Composed of N-acetylglucosamine units, β(1→4) bonds.
Peptidoglycan: Alternating GlcNAc and MurNAc units in bacterial cell walls.
Polysaccharide Structure and Glycosidic Bonds
The type of glycosidic bond (α or β) determines the three-dimensional structure and biological function of the polysaccharide. α-linked polysaccharides (starch, glycogen) form helices, while β-linked polysaccharides (cellulose) form rigid rods.
Lipids
Overview and Features of Lipids
Lipids are a diverse group of hydrophobic or amphipathic molecules, not formed by linear polymerization. They play key roles in energy storage, membrane structure, and signaling.
Solubility: Insoluble in water, soluble in nonpolar solvents.
Amphipathic Nature: Some lipids have both polar and nonpolar regions.
Main Classes of Lipids
Lipids are classified into six main groups: fatty acids, triacylglycerols, phospholipids, glycolipids, steroids, and terpenes.
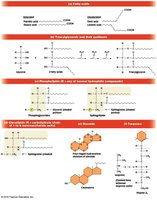
Fatty Acids
Fatty acids are long, unbranched hydrocarbon chains with a terminal carboxyl group. They are building blocks for several lipid classes.
Saturated Fatty Acids: No double bonds; straight chains pack tightly.
Unsaturated Fatty Acids: One or more double bonds; kinks prevent tight packing.
Trans Fats: Unsaturated fats with trans double bonds; less bend, associated with health risks.
Triacylglycerols (Triglycerides)
Triacylglycerols are storage lipids composed of glycerol esterified with three fatty acids. They serve as major energy reserves in animals and plants.
Fats: Solid at room temperature, mostly saturated fatty acids.
Oils: Liquid at room temperature, mostly unsaturated fatty acids.
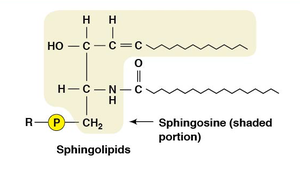
Phospholipids
Phospholipids are major components of cellular membranes due to their amphipathic nature. They are divided into phosphoglycerides and sphingolipids.
Phosphoglycerides: Glycerol backbone, two fatty acids, phosphate group, and a small alcohol (e.g., serine, choline).
Sphingolipids: Sphingosine backbone, fatty acid, and phosphate or carbohydrate group.
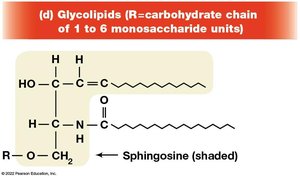
Glycolipids
Glycolipids are membrane lipids with carbohydrate groups attached. They are important for cell recognition and are mainly found on the outer leaflet of the plasma membrane.
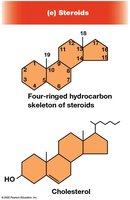
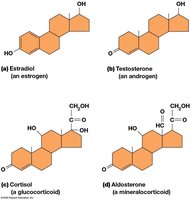
Steroids
Steroids are lipids with a characteristic four-ring hydrocarbon skeleton. Cholesterol is the most common steroid in animal cells and is a precursor for steroid hormones.
Steroid Hormones
Steroid hormones are derived from cholesterol and include sex hormones (estrogens, androgens), glucocorticoids, and mineralocorticoids. They regulate diverse physiological processes such as metabolism, inflammation, and ion balance.
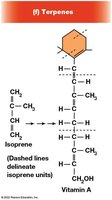
Terpenes
Terpenes are synthesized from isoprene units and include important biological molecules such as vitamin A, carotenoids, dolichols, and electron carriers (coenzyme Q, plastoquinone). Polyisoprenoids are found in archaeal membranes.