 Back
BackMacromolecules of the Cell: Structure, Properties, and Functions
Study Guide - Smart Notes

The Macromolecules of the Cell
Overview of Biological Macromolecules
Biological macromolecules are essential for cellular structure and function. They are synthesized from a limited set of small molecules, primarily amino acids, aromatic bases, sugars, and lipids. These macromolecules include proteins, nucleic acids, polysaccharides, and lipids, each with distinct roles in the cell.
Proteins: Catalysts, structural support, motility, regulation, transport, signaling, receptors, defense, and storage.
Nucleic Acids: Store, transmit, and express genetic information.
Sugars: Energy metabolism and structural components.
Lipids: Membrane structure and energy storage.
Protein Structure and Function
Diversity of Protein Function
Proteins are the most versatile macromolecules in cells, performing a wide range of functions:
Enzymes: Catalyze biochemical reactions.
Structural Proteins: Provide physical support and shape.
Motility Proteins: Enable contraction and movement.
Regulatory Proteins: Control and coordinate cellular activities.
Transport Proteins: Move substances across membranes.
Signaling Proteins: Facilitate communication between cells.
Receptor Proteins: Allow cells to respond to external stimuli.
Defensive Proteins: Protect against disease.
Storage Proteins: Serve as reservoirs of amino acids.
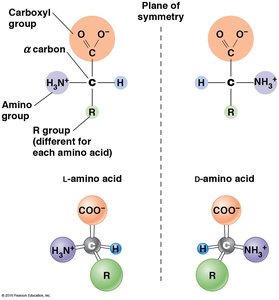
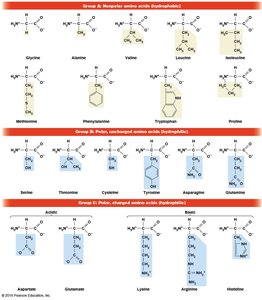
Amino Acids: The Monomers of Proteins
Proteins are polymers composed of 20 different amino acids, each with a common structure but unique side chain (R group). Amino acids exist as L and D isomers, but only L-amino acids are used in protein synthesis. Glycine is unique among non-polar amino acids as it does not have separate L and D isomers due to its symmetric structure.
Formation of Polypeptides
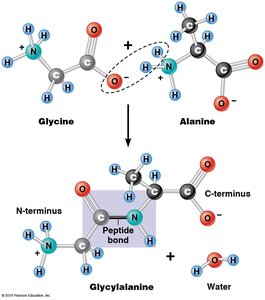
Amino acids are linked by dehydration (condensation) reactions, forming peptide bonds between the carboxyl group of one amino acid and the amino group of another. This results in a linear, unbranched polymer with an N-terminus and a C-terminus.
Polypeptide: Single, unbranched polymer of amino acids.
Protein: Functional molecule made of one or more polypeptides.
Monomeric and Multimeric Proteins
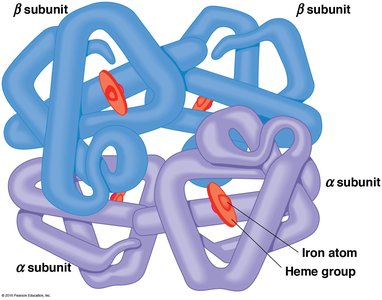
Proteins can be monomeric (single polypeptide) or multimeric (multiple polypeptides). Multimeric proteins are classified by the number and type of subunits, such as dimers, trimers, and tetramers. Hemoglobin is a classic example of a tetramer, consisting of two α and two β subunits.
Levels of Protein Structure
Primary Structure
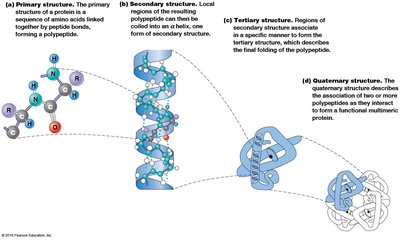
The primary structure is the linear sequence of amino acids in a polypeptide, determined by the nucleotide sequence of mRNA. It is read from the N-terminus to the C-terminus and dictates the higher-order structures.
Bonds: Covalent peptide bonds.
Secondary Structure
Secondary structure refers to local regions of structure stabilized by hydrogen bonding between NH and CO groups of the backbone. The two main patterns are the α helix and β sheet.
α Helix: Spiral shape with R groups projecting outward; stabilized by hydrogen bonds every fourth amino acid.
β Sheet: Extended sheet-like conformation; can be parallel or anti-parallel; stabilized by hydrogen bonds between adjacent strands.
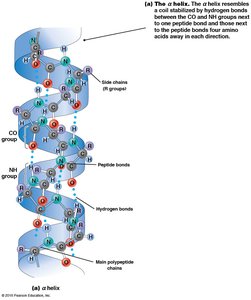
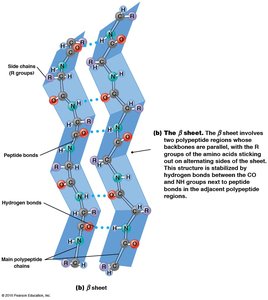
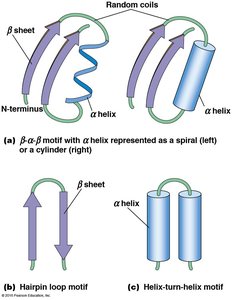
Tertiary Structure
Tertiary structure is the three-dimensional folding of a single polypeptide, determined by interactions among R groups. It is unique for each protein and involves hydrophobic, hydrophilic, ionic, and van der Waals interactions, as well as disulfide bonds.
Fibrous Proteins: Highly ordered, repetitive structure (e.g., fibroin, keratin).
Globular Proteins: Compact, unique tertiary structure; most enzymes are globular.
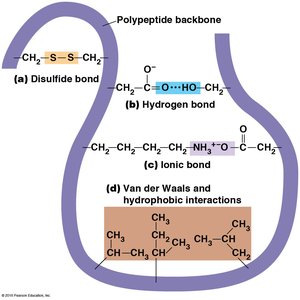
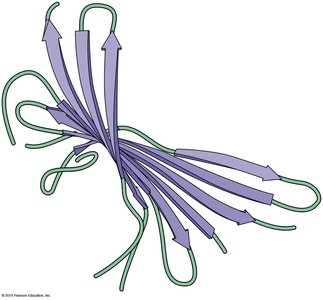
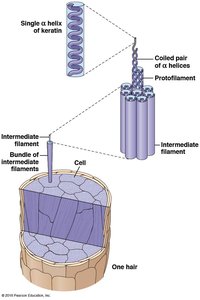
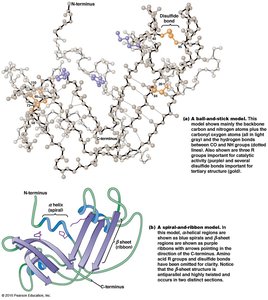
Quaternary Structure
Quaternary structure describes the association of multiple polypeptides to form a functional multimeric protein. The same types of bonds and interactions as in tertiary structure maintain quaternary structure.
Homodimer: Two identical subunits.
Heterodimer: Two different subunits.
Nucleic Acids: Structure and Function
Nucleic Acid Components
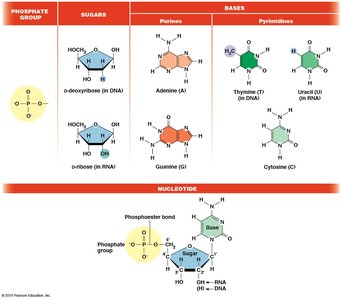
Nucleic acids are linear polymers of nucleotides, each composed of a phosphate group, a five-carbon sugar (ribose or deoxyribose), and a nitrogenous base (purine or pyrimidine). The phosphate group is responsible for the acidic nature of nucleic acids.

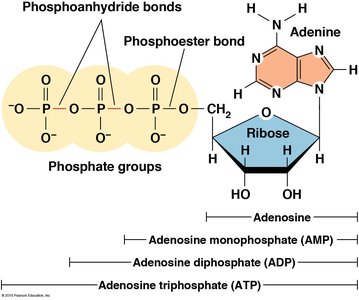
Nucleotide Nomenclature
Nucleotides are named based on their base, sugar, and number of phosphate groups. For example, adenosine monophosphate (AMP) has one phosphate, adenosine diphosphate (ADP) has two, and adenosine triphosphate (ATP) has three.
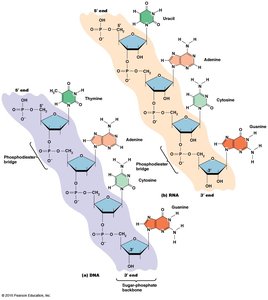
Polymerization of Nucleic Acids
Nucleic acids are synthesized by linking nucleotides via 3ʹ,5ʹ phosphodiester bonds, forming a sugar-phosphate backbone. The resulting polynucleotide has directionality, with a 5ʹ phosphate group at one end and a 3ʹ hydroxyl group at the other. Sequences are written from 5ʹ to 3ʹ.
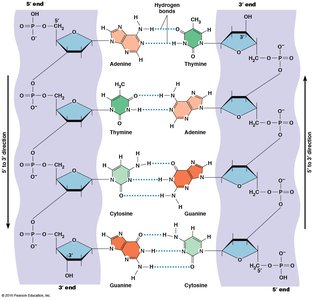
Complementary Base Pairing
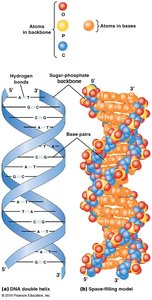
Complementary base pairing is fundamental to nucleic acid structure. In DNA, adenine pairs with thymine via two hydrogen bonds, and guanine pairs with cytosine via three hydrogen bonds. DNA is double-stranded and antiparallel, while RNA is usually single-stranded but can form local base-paired regions.
Base Pairing: A–T (or A–U in RNA), G–C
Antiparallel: Strands run in opposite directions.
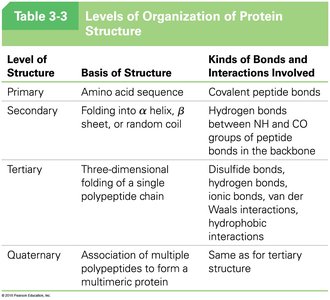
Summary Table: Levels of Protein Structure
Level of Structure | Basis of Structure | Kinds of Bonds and Interactions Involved |
|---|---|---|
Primary | Amino acid sequence | Covalent peptide bonds |
Secondary | Folding into α helix, β sheet, or random coil | Hydrogen bonds between NH and CO groups of peptide bonds in the backbone |
Tertiary | Three-dimensional folding of a single polypeptide chain | Disulfide bonds, hydrogen bonds, ionic bonds, van der Waals interactions, hydrophobic interactions |
Quaternary | Association of multiple polypeptides to form a multimeric protein | Same as for tertiary structure |
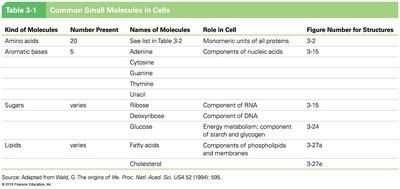
Summary Table: Common Small Molecules in Cells
Kind of Molecules | Number Present | Names of Molecules | Role in Cell |
|---|---|---|---|
Amino acids | 20 | See list in Table 3-2 | Monomeric units of all proteins |
Aromatic bases | 5 | Adenine, Cytosine, Guanine, Thymine, Uracil | Components of nucleic acids |
Sugars | varies | Ribose, Deoxyribose, Glucose | Components of RNA, DNA, energy metabolism |
Lipids | varies | Fatty acids, Cholesterol | Membrane structure, energy storage |
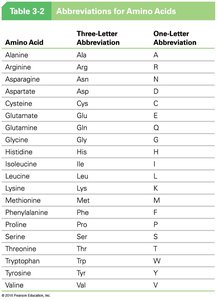
Summary Table: Abbreviations for Amino Acids
Amino Acid | Three-Letter Abbreviation | One-Letter Abbreviation |
|---|---|---|
Alanine | Ala | A |
Arginine | Arg | R |
Asparagine | Asn | N |
Aspartate | Asp | D |
Cysteine | Cys | C |
Glutamate | Glu | E |
Glutamine | Gln | Q |
Glycine | Gly | G |
Histidine | His | H |
Isoleucine | Ile | I |
Leucine | Leu | L |
Lysine | Lys | K |
Methionine | Met | M |
Phenylalanine | Phe | F |
Proline | Pro | P |
Serine | Ser | S |
Threonine | Thr | T |
Tryptophan | Trp | W |
Tyrosine | Tyr | Y |
Valine | Val | V |
Key Equations
Peptide Bond Formation:
Phosphodiester Bond Formation in Nucleic Acids:
Additional info: Academic context was added to clarify the structure, function, and synthesis of macromolecules, as well as the importance of primary, secondary, tertiary, and quaternary protein structures. Tables were recreated for clarity and completeness.
