 Back
BackPolysaccharides and Lipids: Structure, Function, and Biological Significance
Study Guide - Smart Notes

Polysaccharides
Definition and General Properties
Polysaccharides are long-chain polymers composed of sugars and sugar derivatives. They play essential roles in cellular structure and energy storage, typically consisting of repeating units of a single sugar or an alternating pattern of two types. Shorter sugar chains, known as oligosaccharides, are often attached to cell surface proteins, contributing to cell recognition and signaling.
Structure: Polysaccharides are made up of monosaccharide units linked by glycosidic bonds.
Function: Serve as structural components (e.g., cellulose) and storage molecules (e.g., starch, glycogen).
Monomer Types: Most sugars have 3–7 carbons and are classified as trioses, tetroses, pentoses, hexoses, or heptoses.
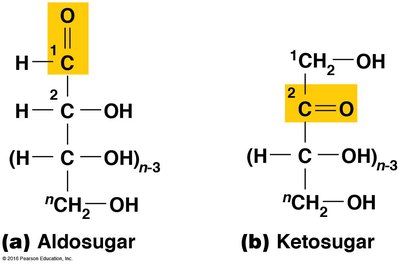
Monosaccharide Structure and Classification
Monosaccharides are classified based on the location of their carbonyl group and the number of carbon atoms.
Aldosugars: Contain a terminal carbonyl group (aldehyde).
Ketosugars: Contain an internal carbonyl group (ketone).
Examples: Ribose, deoxyribose, glucose, galactose, fructose.
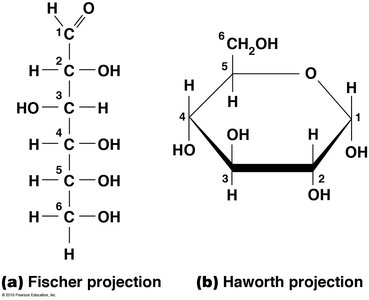
Glucose Structure
Glucose, a hexose, is a primary energy source in cells. It can be represented in two main forms:
Fischer projection: Linear form.
Haworth projection: Cyclic (ring) form, which predominates in solution.
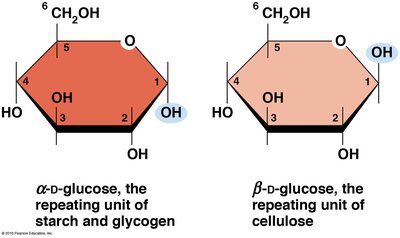
Ring Forms of Glucose
Glucose exists in two ring forms, α and β, which differ in the orientation of the hydroxyl group at carbon 1.
α-D-glucose: The repeating unit of starch and glycogen.
β-D-glucose: The repeating unit of cellulose.
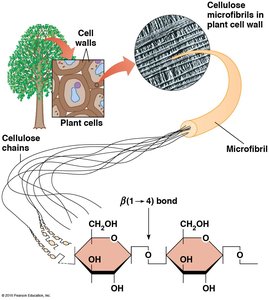
Glycosidic Bonds
Glycosidic bonds are formed between two monosaccharides by the elimination of water. The type of glycosidic bond (α or β) determines the structure and function of the resulting polysaccharide.
α glycosidic bonds: Found in starch and glycogen.
β glycosidic bonds: Found in cellulose and chitin.
Storage Polysaccharides
Storage polysaccharides are crucial for energy storage in cells. The most familiar examples are starch in plants and glycogen in animals and bacteria.
Starch: Composed of amylose (unbranched) and amylopectin (branched).
Glycogen: Highly branched, found in animals.
Structure: Both consist of α-D-glucose units linked by α(1→4) glycosidic bonds, with occasional α(1→6) bonds for branching.
Structural Polysaccharides
Structural polysaccharides provide rigidity and support to cells. Cellulose is the best-known example, forming plant cell walls.
Cellulose: Composed of β-D-glucose monomers linked by β(1→4) glycosidic bonds.
Microfibrils: Cellulose chains aggregate into microfibrils, which are embedded in a matrix of other polymers and proteins.
Chitin: Structural polysaccharide in insect exoskeletons and crustacean shells.
Bacterial polysaccharides: Components of gram-negative bacterial cell walls (e.g., LPS).
Polysaccharide Structure and Glycosidic Bonds
The type of glycosidic bond (α or β) leads to marked structural differences:
α polysaccharides: Form loose helices (starch, glycogen).
β polysaccharides: Form rigid linear rods (cellulose, chitin).
Plant and fungal cell walls: Contain cellulose microfibrils in a matrix of hemicellulose, pectin, and extensin.
Lipids
Definition and General Properties
Lipids are a diverse group of hydrophobic molecules not formed by linear polymerization. They have few polar groups, but some are amphipathic, possessing both polar and nonpolar regions. Lipids serve in energy storage, membrane structure, and signaling.
Hydrophobic nature: Lipids are insoluble in water.
Amphipathic lipids: Have both hydrophilic and hydrophobic regions (e.g., phospholipids).
Fatty Acids
Fatty acids are the building blocks of several classes of lipids. They are long, unbranched hydrocarbon chains with a carboxyl group at one end.
Structure: Amphipathic, with a hydrophobic tail and a hydrophilic carboxyl head.
Function: Components of storage lipids and membrane lipids.
Number of Carbons | Number of Double Bonds | Common Name |
|---|---|---|
12 | 0 | Laurate |
14 | 0 | Myristate |
16 | 0 | Palmitate |
18 | 0 | Stearate |
20 | 0 | Arachidate |
16 | 1 | Palmitoleate |
18 | 1 | Oleate |
18 | 2 | Linoleate |
18 | 3 | Linolenate |
20 | 4 | Arachidonate |
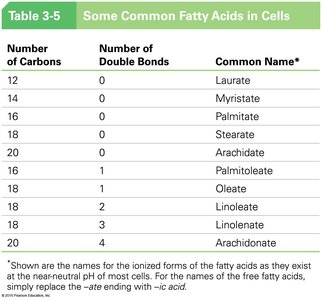
Saturated vs. Unsaturated Fatty Acids
Fatty acids can be saturated (no double bonds) or unsaturated (one or more double bonds).
Saturated fatty acids: Straight chains, solid at room temperature (e.g., palmitate).
Unsaturated fatty acids: Have kinks due to double bonds, liquid at room temperature (e.g., oleate).
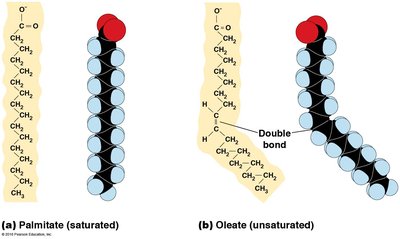
Cis and Trans Fatty Acids
Cis double bonds: Cause a bend in the fatty acid chain, found in natural unsaturated fats.
Trans double bonds: Do not cause a bend, found in some processed foods.
Triacylglycerols (Triglycerides)
Triacylglycerols are storage lipids formed by the esterification of three fatty acids to a glycerol molecule.
Function: Energy storage in animals and plants.
Physical state: Saturated triacylglycerols are solid (fats); unsaturated are liquid (oils).
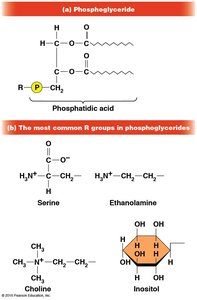
Phospholipids
Phospholipids are major components of cell membranes due to their amphipathic nature.
Structure: Composed of glycerol, two fatty acids, a phosphate group, and a variable R group.
Function: Form bilayers in membranes, providing structural integrity and fluidity.
Glycolipids
Glycolipids are specialized membrane components containing a carbohydrate group instead of a phosphate group.
Structure: Often derivatives of sphingosine and glycerol.
Function: Occur mainly on the outer monolayer of the plasma membrane, involved in cell recognition.
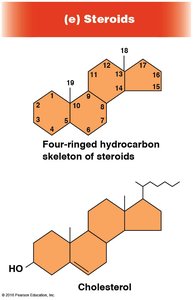
Steroids
Steroids are lipids characterized by a four-ringed hydrocarbon skeleton. Cholesterol is the precursor for other steroids.
Structure: Four fused rings, mostly non-polar.
Function: Membrane structure (cholesterol), signaling (steroid hormones).
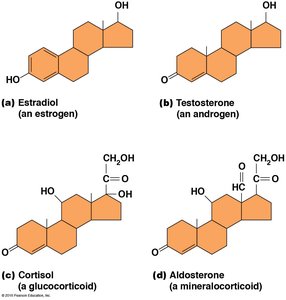
Steroid Hormones
Steroid hormones are signaling molecules derived from cholesterol. They regulate various physiological processes.
Examples: Estradiol (estrogen), testosterone (androgen), cortisol (glucocorticoid), aldosterone (mineralocorticoid).
Functions: Control metabolism, ion balance, and sexual differentiation.
General Trends in Biological Polymers
Structure and Function Relationship
The order and bonding of monomers in biological polymers determine their three-dimensional structure and function.
Key Point: The specific arrangement of monomers and the type of bonds formed are critical for the functional properties of the polymer.
Example: The difference between α and β glycosidic bonds in glucose polymers leads to distinct structures and functions (energy storage vs. structural support).
Summary Table: Comparison of Polysaccharides and Lipids
Polymer Type | Monomer | Bond Type | Function |
|---|---|---|---|
Starch | α-D-glucose | α(1→4), α(1→6) | Energy storage (plants) |
Glycogen | α-D-glucose | α(1→4), α(1→6) | Energy storage (animals) |
Cellulose | β-D-glucose | β(1→4) | Structural support (plants) |
Chitin | N-acetylglucosamine | β(1→4) | Structural support (insects, fungi) |
Triacylglycerol | Fatty acids, glycerol | Ester | Energy storage |
Phospholipid | Fatty acids, glycerol, phosphate | Ester, phosphodiester | Membrane structure |
Steroid | Cholesterol | None (rings) | Signaling, membrane structure |
