 Back
BackStem Cells, Cellular Reprogramming, and Viruses: Mechanisms and Applications
Study Guide - Smart Notes

Stem Cells and Cellular Reprogramming
Embryonic Stem Cells (ESCs) and Cloning
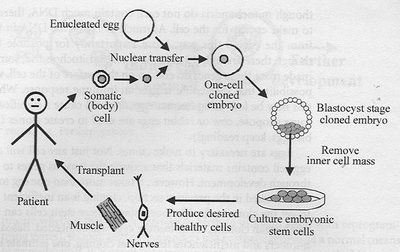
Embryonic stem cells (ESCs) are pluripotent cells derived from the inner cell mass of the blastocyst. They have the ability to differentiate into any cell type in the body. Cloning by somatic cell nuclear transfer (SCNT) involves transferring the nucleus of a somatic cell into an enucleated egg, resulting in a cloned embryo from which ESCs can be derived. These ESCs can be cultured and differentiated into specific cell types for therapeutic purposes.
Somatic Cell Nuclear Transfer (SCNT): A technique where the nucleus of a somatic cell is transferred into an enucleated egg, leading to the formation of a cloned embryo.
Blastocyst Stage: The cloned embryo develops to the blastocyst stage, from which the inner cell mass is isolated to derive ESCs.
Therapeutic Cloning: ESCs can be differentiated into desired cell types (e.g., muscle, nerves) for transplantation back into the patient, potentially treating degenerative diseases.
Example: ESC-derived neurons or muscle cells can be used to replace damaged tissues in patients with neurodegenerative or muscular disorders.
Induced Pluripotent Stem Cells (iPSCs)
Induced pluripotent stem cells (iPSCs) are generated by reprogramming adult somatic cells to a pluripotent state using defined factors. This process, pioneered by Shinya Yamanaka and James Thomson, allows for the creation of patient-specific pluripotent cells without the ethical concerns associated with ESCs.
Reprogramming Factors: Key transcription factors (e.g., Oct4, Sox2, Klf4, c-Myc) are introduced to somatic cells to induce pluripotency.
Alternative Methods: Safer and more efficient reprogramming methods include using microRNAs, mRNA, chemical compounds, or even acid wash, avoiding viral integration.
Applications: iPSCs are used in disease modeling, drug screening, and regenerative medicine.
Example: Patient-derived iPSCs can be differentiated into cardiomyocytes to study heart disease mechanisms.
Adult Stem Cells
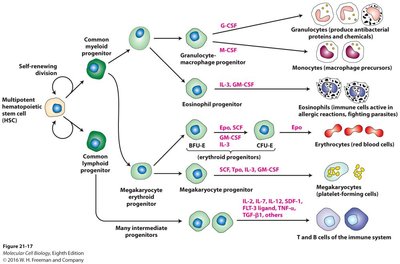
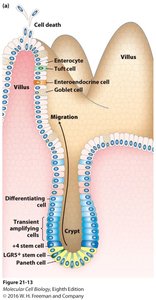
Adult stem cells are multipotent or unipotent cells found in various tissues, responsible for tissue maintenance and repair. Examples include hematopoietic stem cells (HSCs) in the bone marrow, intestinal stem cells (ISCs) in the gut, and skin stem cells.
Hematopoietic Stem Cells (HSCs): Give rise to all blood cell types through a hierarchy of progenitors.
Intestinal Stem Cells (ISCs): LGR5+ cells located in the crypts generate all epithelial cell types of the intestine.
Skin Stem Cells: Responsible for the regeneration of the epidermis and hair follicles.
Example: HSC transplantation is used to treat leukemia and other blood disorders.
Organoids from Adult and Pluripotent Stem Cells
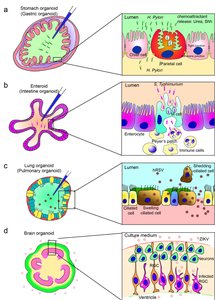
Organoids are three-dimensional, stem cell-derived tissue cultures that mimic the architecture and function of real organs. They are valuable for studying development, disease mechanisms, and drug responses.
ASC-derived Organoids: Derived from adult stem cells, such as intestinal or lung organoids.
PSC-derived Organoids: Generated from pluripotent stem cells, including brain and kidney organoids.
Applications: Used to model infectious diseases (e.g., Zika virus in brain organoids, SARS-CoV-2 in lung organoids) and for personalized medicine.
Example: Brain organoids have been used to study the effects of Zika virus on neural development.

Trans-differentiation
Trans-differentiation refers to the direct conversion of one differentiated cell type into another without passing through a pluripotent state. This process can be induced by specific transcription factors or chemical treatments.
Mechanism: Involves reprogramming the gene expression profile of a cell to adopt a new identity.
Applications: Potential for regenerative medicine, such as converting fibroblasts directly into neurons or cardiomyocytes.
Example: "OKSM transdifferentiation" uses the same factors as iPSC generation but directs cells to a different lineage.
Viruses: Structure, Classification, and Life Cycle
What is a Virus?
A virus is an obligatory intracellular parasite composed of a nucleic acid genome (DNA or RNA) enclosed by a protein coat, and sometimes a lipid envelope. Viruses rely on host cell machinery for replication and expression of their genes.
Genome: Can be DNA or RNA, single- or double-stranded, continuous or segmented.
Protein Coat (Capsid): Protects the viral genome and facilitates entry into host cells.
Lipid Envelope: Present in some viruses, derived from host cell membranes.
Obligate Parasites: Cannot replicate independently; require host cell resources.
Viral Genome Structure and Baltimore Classification
Viral genomes vary widely in size and structure. The Baltimore classification system categorizes viruses based on their genome type and replication strategy.
Genome Types: DNA or RNA, single- or double-stranded, positive- or negative-sense.
Baltimore Classification: Groups viruses into seven classes based on genome and replication method.
Genome Size: Ranges from a few thousand to over a million base pairs (e.g., Mimivirus: 1,200,000 bp; Poliovirus: 7,500 bp).
Efficient Coding: Viral genomes often use overlapping open reading frames (ORFs) to maximize coding capacity.
Virus | Genome Size (bp/nt) |
|---|---|
Human | 3,000,000,000 |
Mimivirus | 1,200,000 |
Smallpox virus | 185,000 |
Herpes simplex virus | 152,000 |
SARS-CoV-2 | 30,000 |
Ebola virus | 18,000 |
Influenza A | 13,500 |
Hepatitis C virus | 9,600 |
HIV-1 | 9,000 |
Poliovirus | 7,500 |
Hepatitis B virus | 3,200 |
Viral Replication Cycle
The viral replication cycle consists of several key steps: attachment, entry, genome replication, gene expression, assembly, and release of progeny virions.
Attachment: Virions bind to specific cell surface receptors, determining host and tissue tropism.
Entry and Uncoating: Virus enters the cell via membrane fusion or endocytosis, releasing its genome.
Gene Expression: Early and late genes are expressed; structural proteins are typically produced late.
Genome Replication: Viral genome is replicated using host or viral enzymes.
Assembly and Release: New virions are assembled and released by cell lysis, budding, or exocytosis.
Virus-Host Interactions and Tropism
Viruses interact with host cells by hijacking cellular machinery and evading antiviral responses. Tropism refers to the specificity of a virus for certain cell types, often determined by the presence of appropriate receptors.
Receptor Usage: Some viruses require multiple receptors or co-receptors (e.g., HIV uses CD4 and CCR5/CXCR4).
Species Specificity: Differences in receptor expression contribute to species barriers.
Example: H5N1 avian influenza virus can acquire mutations that broaden its host range to infect humans.
Viral Genome Packaging and Re-assortment
Viral genomes are selectively packaged into new virions using unique packaging signals. Some viruses, like influenza, have segmented genomes that can undergo re-assortment during co-infection, leading to new viral strains.
Packaging Signals: Specific sequences in the viral genome ensure correct packaging.
Re-assortment: Exchange of genome segments between different viral strains can result in novel viruses with pandemic potential.
Biosafety Levels and Laboratory Containment
Working with viruses requires appropriate biosafety measures, classified into four biosafety levels (BSL-1 to BSL-4) based on the risk posed by the agent.
BSL-1: Minimal risk, standard precautions.
BSL-2: Moderate risk, lab coats and biosafety cabinets required.
BSL-3: Serious risk, restricted access, negative pressure rooms.
BSL-4: Highest risk, full isolation and containment (e.g., Ebola virus).
Example: SARS-CoV-2 is handled at BSL-3, while vaccine strains may be BSL-2.
Summary Table: Key Differences Between ESCs, iPSCs, and Adult Stem Cells
Property | ESCs | iPSCs | Adult Stem Cells |
|---|---|---|---|
Source | Blastocyst inner cell mass | Reprogrammed somatic cells | Adult tissues (e.g., bone marrow, intestine) |
Pluripotency | Yes | Yes | Limited (multipotent/unipotent) |
Ethical Issues | Yes | No | No |
Clinical Use | Potential, limited by ethics | Potential, patient-specific | Established (e.g., HSC transplantation) |
