Back
BackStem Cells in Tissue Homeostasis and Regeneration: Study Notes
Study Guide - Smart Notes

Stem Cells in Tissue Homeostasis and Regeneration
Tissue Homeostasis
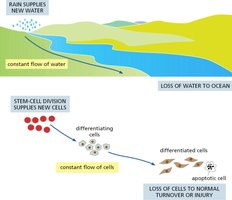
Tissue homeostasis refers to the maintenance of stable cell numbers and tissue function through a balance of cell proliferation, differentiation, and cell loss. Stem cells play a central role in this process by continuously supplying new cells to replace those lost to normal turnover or injury.
Key Point 1: Stem cell division supplies new cells, which differentiate and replace lost or apoptotic cells.
Key Point 2: The process is analogous to a constant flow, similar to water supplied by rain and lost to the ocean.
Example: Gut epithelium and skin epidermis are maintained by stem cell activity.
Stem Cell Division and Self-Renewal
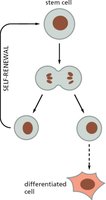
Stem cells are unique in their ability to both self-renew and produce differentiated progeny. Self-renewal ensures the persistence of the stem cell population, while differentiation allows for tissue maintenance and repair.
Key Point 1: Stem cells divide to produce one stem cell (self-renewal) and one differentiated cell.
Key Point 2: This process is tightly regulated to prevent depletion or over-proliferation.
Example: Hematopoietic stem cells in bone marrow.
Cell Hierarchy in Tissues
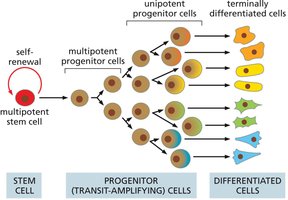
Many tissues are organized in a hierarchical manner, with multipotent stem cells at the top, followed by progenitor cells, and finally terminally differentiated cells. This hierarchy allows for efficient tissue regeneration and specialization.
Key Point 1: Multipotent stem cells give rise to progenitor cells, which further differentiate into specialized cell types.
Key Point 2: Progenitor cells often undergo several rounds of division before terminal differentiation.
Example: Hematopoietic system, where stem cells produce various blood cell types.
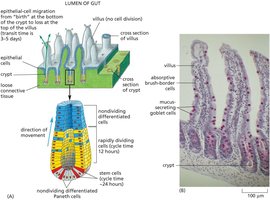
Gut Epithelial Lining Renewal
The gut epithelium is a rapidly renewing tissue, maintained by stem cells located at the base of crypts. These stem cells divide and their progeny migrate upwards, differentiating into various cell types as they move toward the villus tip.
Key Point 1: Stem cells reside in the crypts and produce progeny that migrate and differentiate.
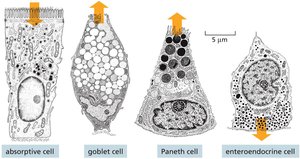
Key Point 2: Four main differentiated cell types are found in the gut lining: absorptive cells, goblet cells, Paneth cells, and enteroendocrine cells.
Example: Migration from crypt to villus takes 3-5 days.
Stem Cell Tracking and Fate Mapping
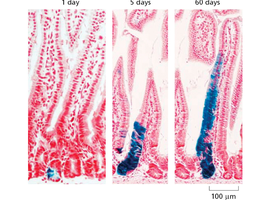
Labeling stem cells and tracking their progeny is a key method for studying cell fate and tissue renewal. This technique reveals the dynamics of cell migration and differentiation over time.
Key Point 1: Fate mapping allows visualization of stem cell contribution to tissue maintenance.
Key Point 2: Quiescent (slow-cycling) stem cells can be tracked to understand their role in regeneration.
Example: Tracking labeled cells in gut epithelium over days.
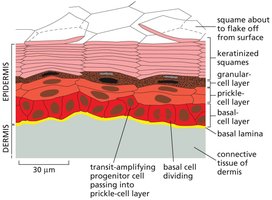
Epidermal Stem Cells and Skin Renewal
The epidermis is a multilayered tissue maintained by basal stem cells. These cells divide and their progeny move upward, differentiating into keratinized squames that eventually flake off.
Key Point 1: Basal cells in the epidermis are stem cells responsible for skin renewal.
Key Point 2: Transit-amplifying progenitor cells pass through several layers before terminal differentiation.
Example: Skin turnover is essential for barrier function and wound healing.
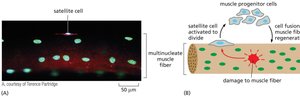
Muscle Regeneration and Satellite Cells
Muscle regeneration is mediated by satellite cells, a type of stem cell located adjacent to muscle fibers. Upon injury, satellite cells activate, proliferate, and fuse to repair damaged muscle.
Key Point 1: Satellite cells are quiescent under normal conditions but activate in response to injury.
Key Point 2: Muscle progenitor cells contribute to muscle fiber regeneration.
Example: Regeneration after exercise or trauma.
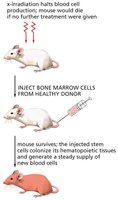
Hematopoietic Stem Cell Transplantation
Hematopoietic stem cell transplantation is a clinical procedure used to restore blood cell production in patients with damaged or diseased bone marrow. Donor stem cells colonize the recipient's marrow and generate new blood cells.
Key Point 1: Transplantation can rescue patients from bone marrow failure.
Key Point 2: Hematopoietic stem cells are multipotent and give rise to all blood cell types.
Example: Treatment for leukemia and other blood disorders.
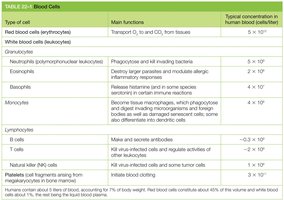
Blood Cell Types and Functions
Blood contains a variety of cell types, each with specialized functions. These cells are derived from hematopoietic stem cells through a hierarchical differentiation process.
Key Point 1: Red blood cells transport oxygen; white blood cells are involved in immunity.
Key Point 2: Granulocytes, monocytes, and lymphocytes are major classes of white blood cells.
Example: Neutrophils phagocytose bacteria; B cells produce antibodies.
Type of cell | Main functions | Typical concentration in human blood (cells/liter) |
|---|---|---|
Red blood cells (erythrocytes) | Transport O2 to, and CO2 from, tissues | 5 × 1012 |
White blood cells (leukocytes) | Immunity | 4–11 × 109 |
Neutrophils | Phagocytose and kill invading bacteria | 2–7 × 109 |
Eosinophils | Destroy larger parasites and modulate allergic inflammatory responses | 0.05–0.5 × 109 |
Basophils | Release histamine in some species; important in certain immune reactions | 0.01–0.1 × 109 |
Monocytes | Phagocytose and kill microorganisms; differentiate into dendritic cells | 0.2–0.8 × 109 |
B cells | Make and secrete antibodies | ~0.3–3 × 109 |
T cells | Kill virus-infected cells and regulate activities of other immune cells | ~0.3–3 × 109 |
Natural killer (NK) cells | Kill virus-infected cells and some tumor cells | ~0.1–1 × 109 |
Platelets | Stop bleeding from injured blood vessels | 1.5–4 × 1011 |
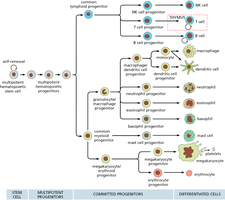
Hematopoietic Cell Differentiation Pathways
Hematopoietic stem cells differentiate through a series of progenitor stages to produce all blood cell types. This process is regulated by growth factors and environmental cues.
Key Point 1: Multipotent stem cells give rise to committed progenitors, which then differentiate into mature blood cells.
Key Point 2: Lineage commitment is influenced by signaling molecules and niche interactions.
Example: Lymphoid and myeloid lineages produce distinct cell types.
Stem Cell Niche and Regulation
The stem cell niche is a specialized microenvironment that regulates stem cell behavior through physical and chemical signals. The size and composition of the niche can limit the number of stem cells and influence their fate.
Key Point 1: Niche-supporting cells and extracellular matrix concentrate signaling molecules.
Key Point 2: Asymmetric division and position relative to the niche determine cell fate.
Example: Intestinal crypt and bone marrow niches.
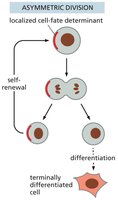
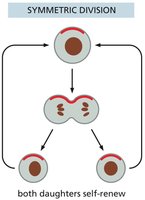
Stem Cell Division: Asymmetric vs. Symmetric
Stem cells can divide asymmetrically, producing one stem cell and one differentiated cell, or symmetrically, producing two stem cells or two differentiated cells. The mode of division affects tissue regeneration and stem cell pool size.
Key Point 1: Asymmetric division maintains stem cell numbers while generating differentiated cells.
Key Point 2: Symmetric division can expand or deplete the stem cell pool.
Example: Asymmetric division is common in tissues with constant renewal.
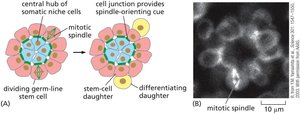
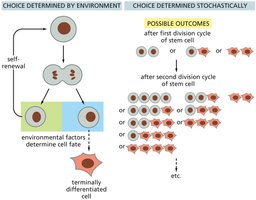
Stem Cell Fate Determination
Stem cell fate can be determined by environmental cues or by stochastic (random) mechanisms. Environmental factors such as niche signals can direct differentiation, while stochastic processes allow flexibility in cell fate outcomes.
Key Point 1: Environmental determination involves signals from niche or neighboring cells.
Key Point 2: Stochastic determination results in variable outcomes after each division.
Example: Germ-line stem cells in Drosophila are influenced by niche signals.
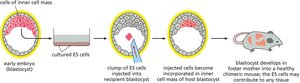
Stem Cell Potency: Totipotency and Pluripotency
Stem cells are classified by their potency, or ability to differentiate into various cell types. Totipotent cells can give rise to all cell types in an organism, while pluripotent cells can form most, but not all, cell types.
Key Point 1: Embryonic stem cells are totipotent; pluripotent cells are derived from embryonic stem cells.
Key Point 2: Pluripotency is essential for development and regenerative medicine.
Example: ES cells can contribute to all tissues in a chimeric mouse.
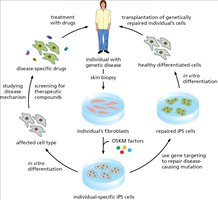
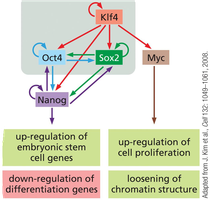
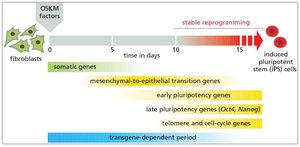
Induced Pluripotent Stem Cells (iPS Cells)
Induced pluripotent stem cells (iPS cells) are generated by reprogramming somatic cells using defined transcription factors (OKSM: Oct4, Klf4, Sox2, Myc). iPS cells exhibit pluripotency similar to embryonic stem cells and are valuable for research and therapy.
Key Point 1: iPS cells are created by introducing OKSM factors into fibroblasts.
Key Point 2: iPS technology enables patient-specific cell therapies and disease modeling.
Example: Reprogramming fibroblasts to iPS cells for regenerative medicine.

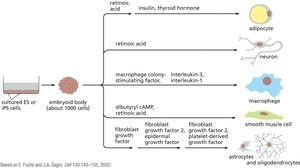
Differentiation of iPS Cells
iPS cells can be differentiated into various cell types by exposing them to specific growth factors and signaling molecules. This process is used to generate cells for research, drug screening, and potential therapies.
Key Point 1: Differentiation protocols guide iPS cells to become adipocytes, neurons, macrophages, smooth muscle cells, etc.
Key Point 2: Embryoid bodies are intermediate structures used in differentiation.
Example: Generation of neurons from iPS cells for studying neurodegenerative diseases.
Applications of iPS Technology in Research
iPS technology has revolutionized biomedical research by enabling the creation of patient-specific cell lines for disease modeling, drug screening, and gene therapy. It also allows for the transplantation of genetically repaired cells.
Key Point 1: iPS cells are used to study disease mechanisms and screen therapeutic compounds.
Key Point 2: Gene targeting can repair disease-causing mutations in patient-derived iPS cells.
Example: Transplantation of repaired cells to treat genetic diseases.