 Back
BackStudy Guide: The Macromolecules of the Cell (Cell Biology, Ch. 3)
Study Guide - Smart Notes

The Macromolecules of the Cell
Overview of Cellular Macromolecules
Cellular macromolecules are essential for structure, function, and information storage in all living organisms. The four major classes are proteins, nucleic acids, polysaccharides, and lipids. Each class is composed of specific monomers and assembled through distinct biochemical processes.
Proteins: Polymers of amino acids, perform diverse cellular functions.
Nucleic Acids: Polymers of nucleotides, store and transmit genetic information.
Polysaccharides: Polymers of monosaccharides, serve structural and storage roles.
Lipids: Diverse group, not true polymers, important for membranes and energy storage.
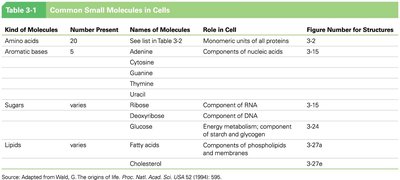
Common Small Molecules in Cells
Cells utilize a limited set of small molecules as building blocks for macromolecules. These include amino acids, aromatic bases, sugars, and lipids, each with specific roles in cellular structure and metabolism.
Kind of Molecule | Number Present | Names of Molecules | Role in Cell |
|---|---|---|---|
Amino acids | 20 | See Table 3-2 | Monomeric units of all proteins |
Aromatic bases | 5 | Adenine, Cytosine, Guanine, Thymine, Uracil | Components of nucleic acids |
Sugars | Varies | Ribose, Deoxyribose, Glucose | Components of RNA, DNA, energy metabolism |
Lipids | Varies | Fatty acids, Cholesterol | Membrane structure, energy storage |
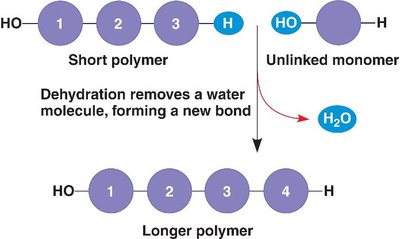
Polymerization and Macromolecule Synthesis
Condensation (Dehydration) Reactions
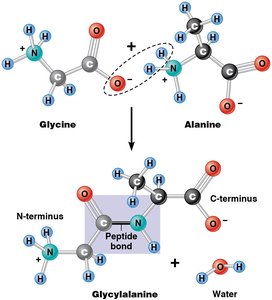
Macromolecules are synthesized by condensation reactions, also known as dehydration synthesis. In these reactions, monomers are joined together by the removal of a water molecule, forming covalent bonds and resulting in polymers with directionality.
Condensation Reaction: Joins monomers, releases H2O.
Directionality: Polymers have distinct ends (e.g., N-terminus and C-terminus in proteins).
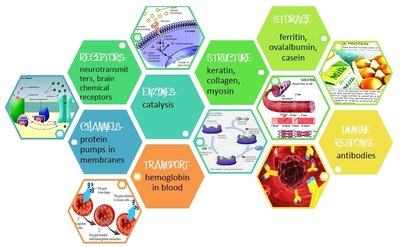
Proteins
Functions and Classes of Proteins
Proteins are the most versatile macromolecules in cells, with nine major functional classes. Their roles range from catalysis to structural support, transport, signaling, and defense.
Enzymes: Catalyze biochemical reactions.
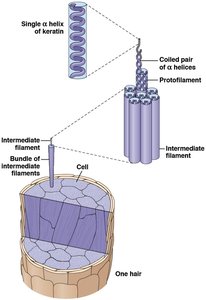
Structural proteins: Provide support and shape (e.g., keratin, collagen).
Motility proteins: Enable movement (e.g., myosin).
Regulatory proteins: Control cellular processes.
Transport proteins: Move substances across membranes.
Signaling proteins: Mediate communication.
Receptor proteins: Receive signals.
Defensive proteins: Protect against disease (e.g., antibodies).
Storage proteins: Store amino acids (e.g., ferritin).
Amino Acids: The Monomers of Proteins
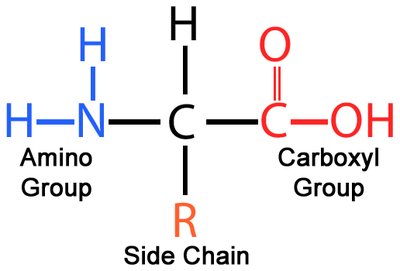
Proteins are polymers of 20 standard amino acids, each with a central (α) carbon, an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group) that determines its properties.
Structure: All amino acids share a common backbone; the R group varies.
Glycine: The only amino acid without an asymmetric α carbon.
Abbreviations for Amino Acids
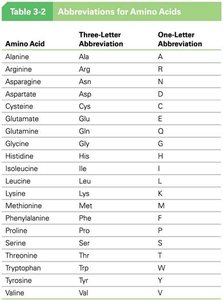
Amino acids are often represented by three-letter and one-letter abbreviations for convenience in sequence notation.
Amino Acid | Three-Letter Abbreviation | One-Letter Abbreviation |
|---|---|---|
Alanine | Ala | A |
Arginine | Arg | R |
Asparagine | Asn | N |
Aspartate | Asp | D |
Cysteine | Cys | C |
Glutamate | Glu | E |
Glutamine | Gln | Q |
Glycine | Gly | G |
Histidine | His | H |
Isoleucine | Ile | I |
Leucine | Leu | L |
Lysine | Lys | K |
Methionine | Met | M |
Phenylalanine | Phe | F |
Proline | Pro | P |
Serine | Ser | S |
Threonine | Thr | T |
Tryptophan | Trp | W |
Tyrosine | Tyr | Y |
Valine | Val | V |
Classification of Amino Acids by R Groups
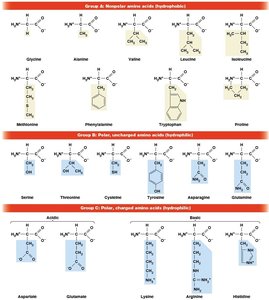
Amino acids are classified based on the chemical nature of their R groups: nonpolar (hydrophobic), polar (hydrophilic), and charged (acidic or basic).
Nonpolar: Glycine, Alanine, Valine, Leucine, Isoleucine, Methionine, Phenylalanine, Tryptophan, Proline
Polar, uncharged: Serine, Threonine, Cysteine, Tyrosine, Asparagine, Glutamine
Polar, charged: Aspartate, Glutamate, Lysine, Arginine, Histidine
Polypeptides and Protein Structure
Peptide Bond Formation
Amino acids are linked by peptide bonds formed through condensation reactions. The resulting polypeptide has directionality, with an N-terminus (amino end) and a C-terminus (carboxyl end).
Levels of Protein Structure
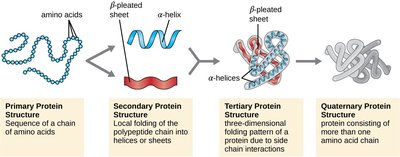
Protein structure is described in four hierarchical levels:
Primary: Linear sequence of amino acids.
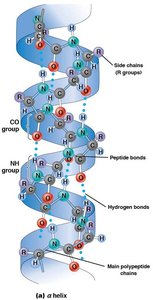
Secondary: Local folding into α helices and β sheets, stabilized by hydrogen bonds.
Tertiary: Overall three-dimensional shape, determined by interactions among R groups.
Quaternary: Association of multiple polypeptide chains (subunits).
Monomeric vs. Multimeric Proteins
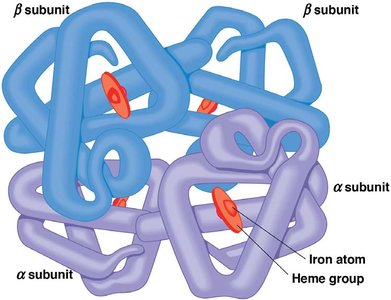
Proteins may consist of a single polypeptide (monomeric) or multiple polypeptides (multimeric), such as dimers, trimers, or tetramers.
Protein Folding and Stability
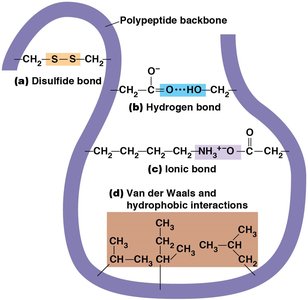
Protein folding is stabilized by covalent and noncovalent interactions:
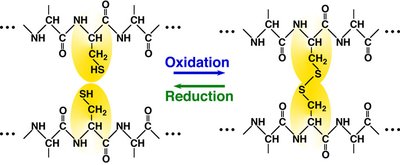
Covalent disulfide bonds: Formed between cysteine residues.
Noncovalent interactions: Hydrogen bonds, ionic bonds, van der Waals forces, hydrophobic interactions.
Primary Structure
The primary structure is the unique sequence of amino acids in a polypeptide, written from N-terminus to C-terminus. This sequence determines all higher levels of structure.
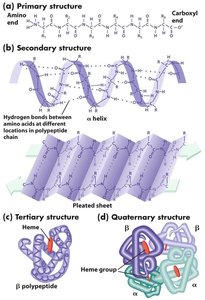
Secondary Structure: α Helix and β Sheet
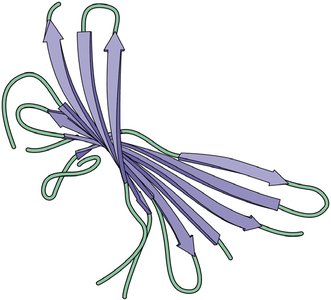
Secondary structure arises from hydrogen bonding along the polypeptide backbone, forming α helices (spiral) and β sheets (extended, pleated).
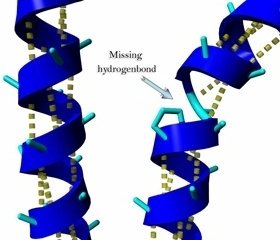
α Helix: Stabilized by hydrogen bonds every fourth amino acid.
β Sheet: Stabilized by hydrogen bonds between adjacent strands; can be parallel or antiparallel.
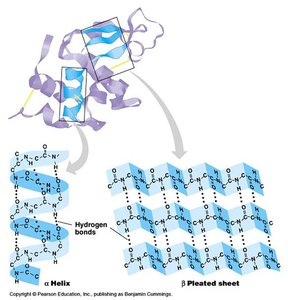
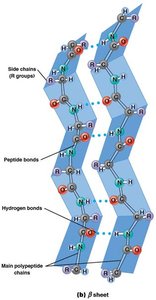
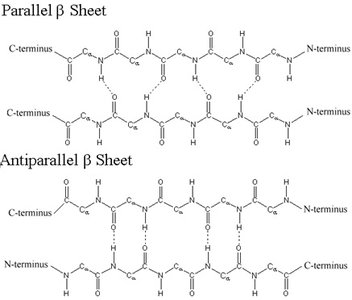
Motifs and Domains
Motifs are combinations of secondary structures (e.g., β–α–β, hairpin loop, helix-turn-helix) that recur in different proteins. Domains are discrete, functional units of tertiary structure, often with independent activity.
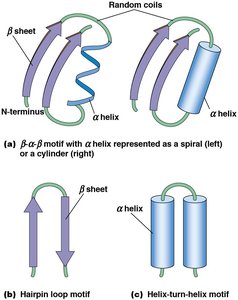
Tertiary Structure
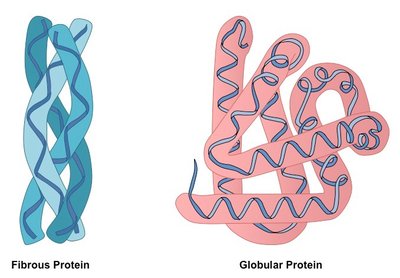
Tertiary structure is the overall three-dimensional shape of a polypeptide, resulting from interactions among R groups. Proteins are classified as fibrous (elongated, structural) or globular (compact, functional).
Fibrous proteins: Provide mechanical support (e.g., keratin, collagen).
Globular proteins: Perform diverse functions (e.g., enzymes, antibodies).
Quaternary Structure
Quaternary structure describes the assembly of multiple polypeptide subunits into a functional protein complex. The same types of bonds and forces as in tertiary structure maintain quaternary structure.
Nucleic Acids
Structure and Function
Nucleic acids are linear polymers of nucleotides. DNA stores genetic information, while RNA is involved in its expression.
DNA: Repository of genetic information.
RNA: Functions in transcription, translation, and regulation.
Nucleotides and Nucleosides
Nucleotides consist of a five-carbon sugar, a phosphate group, and a nitrogenous base. Nucleosides are sugar-base complexes without phosphate.
Pyrimidines: Cytosine, Thymine (DNA), Uracil (RNA).
Purines: Adenine, Guanine.
Polymerization and Directionality
Nucleic acids are formed by 3ʹ,5ʹ phosphodiester bonds, giving directionality (5ʹ to 3ʹ). DNA is double-stranded and forms a double helix; RNA is usually single-stranded.
Polysaccharides
Structure and Function
Polysaccharides are long chains of monosaccharides, serving structural (cellulose, chitin) and storage (starch, glycogen) functions.
Monosaccharides: Classified by number of carbons (triose, tetrose, pentose, hexose, heptose).
Glucose: Most common monosaccharide; exists in α and β ring forms.
Disaccharides: Two monosaccharides linked (e.g., maltose, lactose, sucrose).
Storage polysaccharides: Starch (plants), glycogen (animals).
Structural polysaccharides: Cellulose (plants), chitin (fungi, insects).
Lipids
Structure and Classes
Lipids are hydrophobic macromolecules, not true polymers, important for membranes, energy storage, and signaling. Six main classes: fatty acids, triacylglycerols, phospholipids, glycolipids, steroids, terpenes.
Fatty acids: Long hydrocarbon chains with carboxyl group; can be saturated or unsaturated.
Triacylglycerols: Glycerol with three fatty acids; energy storage.
Phospholipids: Major membrane component; amphipathic.
Glycolipids: Membrane stability and recognition.
Steroids: Four-ring structure; includes cholesterol and hormones.
Terpenes: Derived from isoprene; includes vitamins and pigments.
Fatty Acid Saturation
Saturated: No double bonds; straight chains, solid at room temperature.
Unsaturated: One or more double bonds; bent chains, liquid at room temperature.
Trans fats: Unsaturated with trans double bond; artificially produced, health risks.
Phospholipids and Membrane Structure
Phospholipids are amphipathic, forming bilayers in membranes. Sphingolipids and glycolipids contribute to membrane function and cell recognition.
Steroids and Terpenes
Steroids are hydrophobic molecules with diverse functions, including cholesterol and hormones. Terpenes are derived from isoprene and include important biomolecules like vitamin A and ubiquinone.
Additional info: This study guide expands on brief lecture notes with academic context, definitions, and examples to ensure completeness and clarity for exam preparation.
