 Back
BackThe Cell Cycle: Regulation, Phases, and Control Mechanisms
Study Guide - Smart Notes

The Cell Cycle
Overview of the Cell Cycle
The cell cycle is the fundamental process by which cells duplicate their contents and divide to produce two genetically identical daughter cells. This process is essential for growth, development, and tissue repair in multicellular organisms.
Cell Cycle Definition: The ordered sequence of events that a cell goes through to duplicate its genome and divide.
Main Functions: DNA replication, cell growth, and division into daughter cells.
Phases: The cell cycle is divided into interphase (G1, S, G2) and M phase (mitosis and cytokinesis).
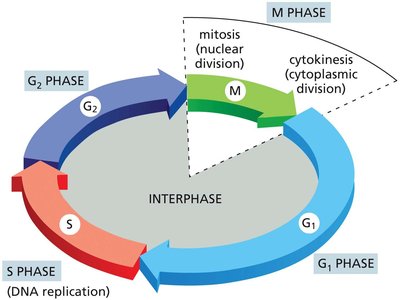
Interphase: Period of cell growth and DNA replication (G1, S, G2 phases).
M Phase: Includes mitosis (nuclear division) and cytokinesis (cytoplasmic division).
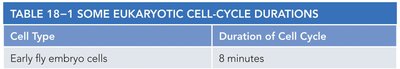
Cell Cycle Duration: Varies by cell type (e.g., early fly embryo cells: 8 min; mammalian fibroblasts: ~20 hours).
Example: Mammalian intestinal epithelial cells divide approximately every 12 hours.
Cell Type | Duration of Cell Cycle |
|---|---|
Early fly embryo cells | 8 minutes |
Early frog embryo cells | 30 minutes |
Mammalian intestinal epithelial cells | ~12 hours |
Mammalian fibroblasts in culture | ~20 hours |
Phases of the Cell Cycle
The eukaryotic cell cycle is divided into four main phases: G1, S, G2, and M. Interphase (G1, S, G2) is the period between cell divisions, while M phase is when the cell actually divides.
G1 Phase: Cell grows and prepares for DNA replication.
S Phase: DNA synthesis and chromosome duplication occur.
G2 Phase: Further growth and preparation for mitosis.
M Phase: Mitosis (nuclear division) and cytokinesis (cytoplasmic division).
Analysis of Cell Cycle Stages
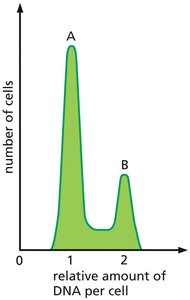
Flow cytometry can be used to analyze the distribution of cells in different phases of the cell cycle by measuring DNA content.
Peak A: Represents cells in G1 phase (one set of DNA).
Peak B: Represents cells in G2/M phase (two sets of DNA).
Longest Phase: Most cells are found in G1, indicating it is the longest phase.
Regulation of the Cell Cycle
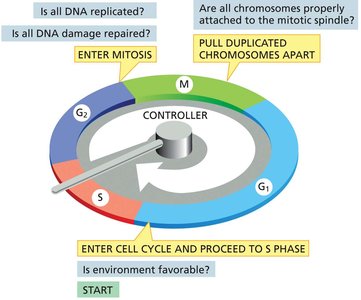
Cell Cycle Control System
The cell cycle is tightly regulated by a control system that ensures each phase is completed correctly before the next begins. This system can halt the cycle at specific checkpoints if conditions are unfavorable.
Checkpoints: G1/S, G2/M, and metaphase-anaphase transitions.
Control Mechanisms: Cyclic activation/inactivation of protein complexes (cyclin-Cdk complexes).
Checkpoint Questions: Is the environment favorable? Is all DNA replicated? Is all DNA damage repaired? Are all chromosomes attached to the spindle?
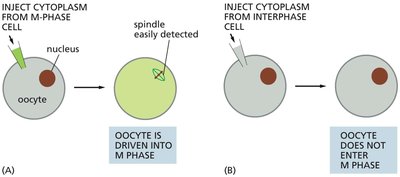
Discovery of Cell Cycle Regulators
Experiments with Xenopus eggs demonstrated that cytoplasm from M-phase cells can induce mitosis in interphase cells, suggesting the presence of regulatory proteins.
Key Finding: Cytoplasmic factors (later identified as cyclin-Cdk complexes) trigger entry into M phase.
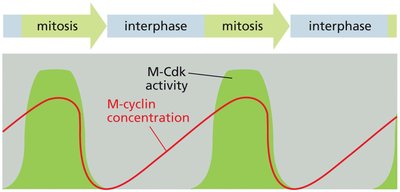
Cyclin-Dependent Kinases (Cdks) and Cyclins
Cyclin-dependent kinases (Cdks) are central to cell cycle regulation. They require binding to cyclins for activation and control progression through the cell cycle by phosphorylating target proteins.
Cdk: Protein kinase that is only active when bound to a cyclin.
Cyclin: Regulatory protein whose levels fluctuate during the cell cycle.
Major Cyclin-Cdk Complexes: G1-Cdk (Cyclin D/Cdk4,6), G1/S-Cdk (Cyclin E/Cdk2), S-Cdk (Cyclin A/Cdk2), M-Cdk (Cyclin B/Cdk1).
Function: Different cyclin-Cdk complexes trigger different cell cycle events.
Cyclin–Cdk Complex | Cyclin | Cdk Partner |
|---|---|---|
G1-Cdk | Cyclin D | Cdk4, Cdk6 |
G1/S-Cdk | Cyclin E | Cdk2 |
S-Cdk | Cyclin A | Cdk2 |
M-Cdk | Cyclin B | Cdk1 |

Regulation of Cyclin-Cdk Activity
Cyclin-Cdk complexes are regulated by synthesis and degradation of cyclins, phosphorylation/dephosphorylation, and Cdk inhibitor proteins (CKIs).
Cyclin Degradation: Cyclins are marked for destruction by the Anaphase Promoting Complex (APC/C), leading to inactivation of Cdks.
Phosphorylation: Inhibitory kinases (e.g., Wee1) add phosphates to Cdks, keeping them inactive; activating phosphatases (e.g., Cdc25) remove these phosphates to activate Cdks.
Cdk Inhibitor Proteins (CKIs): Proteins like p27 bind to cyclin-Cdk complexes and inhibit their activity, especially after mitosis.
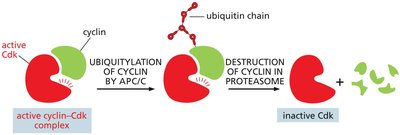
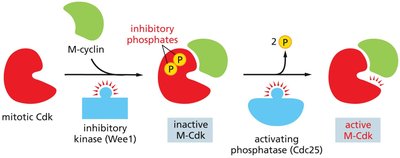
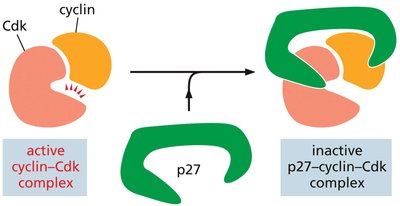
Cell Cycle Checkpoints and Arrest
The cell cycle can be paused at various checkpoints to ensure proper completion of each phase and to prevent the propagation of errors.
G1 Checkpoint: Checks for DNA damage and sufficient resources for DNA replication.
G2/M Checkpoint: Ensures all DNA is replicated and undamaged before mitosis.
Spindle Assembly Checkpoint: Ensures all chromosomes are properly attached to the spindle before anaphase.
Mechanisms of Arrest: Inhibition of Cdc25, activation of CKIs, and inhibition of APC/C can block cell cycle progression.
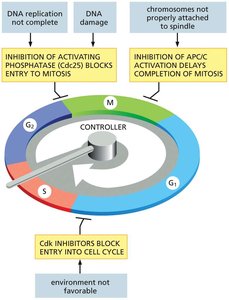
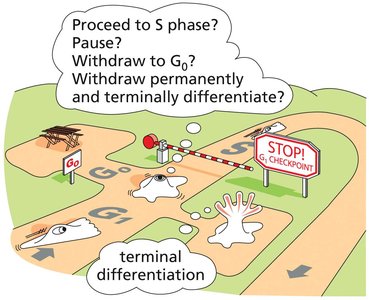
G1 Phase: Decision Point
G1 to S Phase Transition
The transition from G1 to S phase is a critical decision point where cells can either proceed to DNA replication, enter a quiescent state (G0), or differentiate terminally.
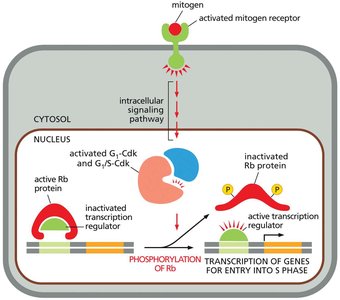
Mitogens: Extracellular signals that stimulate cell division by promoting cyclin synthesis.
Rb Protein: Retinoblastoma protein inhibits transcription factors required for S phase entry; phosphorylation by G1/S-Cdk inactivates Rb, allowing S phase gene expression.
G0 Phase: Non-dividing state; cells can re-enter the cycle or remain permanently differentiated.
DNA Damage Response in G1
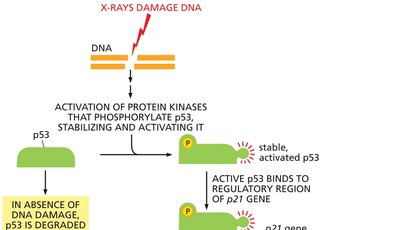
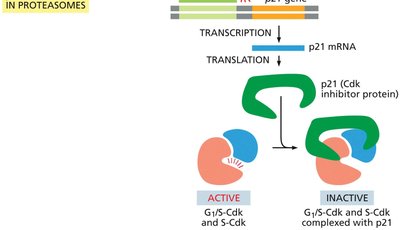
DNA damage in G1 activates the tumor suppressor protein p53, which induces the expression of the Cdk inhibitor p21, halting cell cycle progression to allow for DNA repair.
p53: Transcription factor stabilized by DNA damage; activates p21 gene expression.
p21: Inhibits G1/S-Cdk and S-Cdk, preventing entry into S phase.
Outcome: If DNA is not repaired, p53 can trigger apoptosis.
S Phase: DNA Replication
Initiation and Regulation of DNA Replication
During S phase, the cell duplicates its DNA and centrosomes. Replication is tightly regulated to ensure it occurs only once per cycle.
Origin Recognition Complex (ORC): Binds to replication origins; recruits Cdc6 and DNA helicases to form the prereplicative complex in G1.
S-Cdk: Triggers initiation of DNA replication and prevents re-replication by phosphorylating Cdc6.
Centrosome Duplication: Ensures proper spindle formation in mitosis.
G2 Phase and DNA Damage Checkpoint
During G2, the cell checks for completion of DNA replication and repairs any DNA damage before entering mitosis.
ATM and ATR Kinases: Detect DNA damage and activate Chk1/Chk2 kinases, leading to cell cycle arrest.
Checkpoint Arrest: Prevents entry into mitosis until DNA is fully replicated and undamaged.
M Phase: Mitosis and Cytokinesis
Entry into Mitosis
M-Cdk activation drives entry into mitosis by phosphorylating target proteins. Cdc25 removes inhibitory phosphates from M-Cdk, while Wee1 adds them.
Chromosome Condensation: Cohesins hold sister chromatids together; condensins compact chromosomes.
Mitotic Spindle Formation: Centrosomes nucleate microtubules; spindle apparatus segregates chromosomes.
Spindle Assembly Checkpoint: Ensures proper attachment of chromosomes before anaphase.
Stages of Mitosis
Prophase: Chromosomes condense, spindle begins to form.
Prometaphase: Nuclear envelope breaks down, chromosomes attach to spindle via kinetochores.
Metaphase: Chromosomes align at metaphase plate.
Anaphase: Cohesins are degraded, sister chromatids separate.
Telophase: Nuclear envelope reassembles, chromosomes decondense.
Cytokinesis
The contractile ring, composed of actin and myosin, pinches the cell into two daughter cells. The position of the cleavage furrow is determined by signals from the central spindle.
Control of Cell Number and Size
Growth Factors, Mitogens, and Apoptosis
Cell number and size are regulated by a balance of growth factors, mitogens, and programmed cell death (apoptosis).
Growth Factors: Stimulate cell growth by promoting protein synthesis and inhibiting degradation (e.g., via RTK-AKT-Tor pathway).
Mitogens: Stimulate cell division by promoting cyclin synthesis.
Apoptosis: Programmed cell death mediated by caspases; ensures removal of damaged or unnecessary cells.
Bcl2 Family: Regulates apoptosis; Bax and Bak promote cytochrome c release and caspase activation.
Additional info: Apoptosis is essential for development, tissue homeostasis, and prevention of cancer. Survival factors suppress apoptosis by regulating Bcl2 family proteins.
