 Back
BackThe Chemical Components of Cells: Atoms, Elements, and Bonding
Study Guide - Smart Notes

The Chemical Context of Life
Matter, Elements, and Atoms
Understanding the chemical basis of life is fundamental in cell biology, as all living organisms are composed of atoms and molecules governed by chemical and physical laws. The organization of life begins with atoms, which form molecules, and these molecules assemble into cells—the basic unit of life.
Matter: Anything that occupies space and has mass. Exists as solids, liquids, or gases.
Elements: Pure substances consisting of only one type of atom. Cannot be broken down by chemical means.
States of Matter: Solid, liquid, gas.
Six essential elements for life: Carbon (C), Hydrogen (H), Nitrogen (N), Oxygen (O), Phosphorus (P), Sulfur (S) (CHNOPS).
Other important elements: Sodium (Na), Potassium (K), Calcium (Ca), Iron (Fe), Magnesium (Mg).
Trace elements: Iron (Fe), iodine (I), magnesium (Mg), zinc (Zn) are required in minute quantities for proper biological function.
Element Percentage of Body Mass:
Oxygen (O): 65%
Carbon (C): 18.5%
Hydrogen (H): 9.5%
Nitrogen (N): 3.3%
Calcium (Ca): 1.5%
Phosphorus (P): 1.0%
Potassium (K): 0.4%
Sulfur (S): 0.3%
Sodium (Na): 0.2%
Chlorine (Cl): 0.2%
Magnesium (Mg): 0.1%
The Periodic Table of Elements organizes elements by atomic number, electron structure, and chemical properties. Each element is represented by its atomic number, symbol, name, and atomic mass.
Atoms and Subatomic Particles
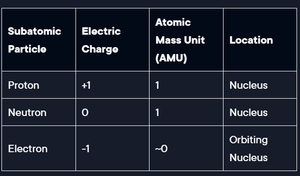
An atom is the smallest unit of an element that retains its chemical properties. Atoms are composed of subatomic particles: protons, neutrons, and electrons.
Atomic symbol: Shorthand for element name.
Atomic number: Number of protons in the nucleus.
Atomic mass: Number of protons plus number of neutrons.
Subatomic Particle | Electric Charge | Atomic Mass Unit (AMU) | Location |
|---|---|---|---|
Proton | +1 | 1 | Nucleus |
Neutron | 0 | 1 | Nucleus |
Electron | -1 | ~0 | Orbiting Nucleus |
Atomic Mass Calculation:
Example Calculations for Common Elements
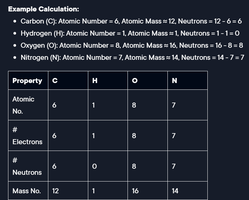
Atomic number, atomic mass, and number of neutrons for key elements:
Carbon (C): Atomic Number = 6, Atomic Mass = 12, Neutrons = 12 - 6 = 6
Hydrogen (H): Atomic Number = 1, Atomic Mass = 1, Neutrons = 1 - 1 = 0
Oxygen (O): Atomic Number = 8, Atomic Mass = 16, Neutrons = 16 - 8 = 8
Nitrogen (N): Atomic Number = 7, Atomic Mass = 14, Neutrons = 14 - 7 = 7
Property | C | H | O | N |
|---|---|---|---|---|
Atomic No. | 6 | 1 | 8 | 7 |
# Electrons | 6 | 1 | 8 | 7 |
# Neutrons | 6 | 0 | 8 | 7 |
Mass No. | 12 | 1 | 16 | 14 |
Isotopes
Isotopes are atoms of the same element with different numbers of neutrons, resulting in different atomic masses. Some isotopes are radioactive and emit particles and energy, which is useful in carbon dating and medical diagnostics.
Example: Carbon-14 (14C) is a radioactive isotope used in dating fossils.
Electron Arrangement and Chemical Reactivity
Electrons occupy energy levels or shells around the nucleus. Their arrangement determines chemical reactivity and bonding.
1st energy shell: 2 electrons
2nd energy shell: 8 electrons
3rd energy shell: 8 electrons
Valence shell: Outermost shell; atoms with incomplete valence shells are reactive.
Valence electrons: Electrons in the valence shell.
Octet rule: Atoms tend to complete their valence shell with 8 electrons.
Atoms and Molecules
Molecules & Types of Bonding
Molecules are formed when two or more atoms join together by chemical bonds. These can be diatomic (same element) or compound molecules (different elements).
Diatomic molecules: O2, H2
Compound molecules: H2O, CO2
Types of Chemical Bonds
Ionic bonds: Formed when one atom donates or gains electrons, resulting in charged ions. The attraction between oppositely charged ions forms the bond.
Covalent bonds: Formed when two atoms share a pair of valence electrons. The strength and nature of the bond depend on the atoms' electronegativity.
Hydrogen bonds: Weak attractions important in biological molecules. Occur when a hydrogen atom covalently bonded to one electronegative atom is attracted to another electronegative atom.
Electronegativity: The tendency of an atom to attract shared electrons.
Non-polar covalent bond: Electrons shared equally (e.g., H2).
Polar covalent bond: Electrons shared unequally (e.g., H2O).
Single, double, and triple bonds: Indicate the number of shared electron pairs.
Structural formula: H–H (single bond), O=O (double bond).
Molecular formula: Indicates the number and type of atoms (e.g., H2O, CH4).
Valence: The number of covalent bonds an atom can form, determined by the number of unpaired electrons in its outer shell.
Hydrogen: 1
Oxygen: 2
Nitrogen: 3
Carbon: 4
Phosphorus: 5
Ionic Bonds
Ionic bonds are formed when one atom donates or gains electrons, resulting in charged ions. The attraction between oppositely charged ions forms the bond.
Cation: Positively charged ion (e.g., Na+).
Anion: Negatively charged ion (e.g., Cl-).
Example: NaCl (table salt) is formed from sodium and chlorine ions.
Hydrogen Bonds
Hydrogen bonds are weak attractions important in biological molecules. They occur when a hydrogen atom covalently bonded to one electronegative atom is attracted to another electronegative atom. Although not a true bond, hydrogen bonds are significant forces in molecules like water and are responsible for many properties of water and the structure of DNA and proteins.
Example: The polarity of the O–H bond in water leads to hydrogen bonding between water molecules.
Additional info: These foundational chemical principles are essential for understanding biological processes such as metabolism, cell structure, and genetic inheritance.
