 Back
BackCH 2 - The Chemistry of the Cell: Principles and Molecular Foundations
Study Guide - Smart Notes

The Chemistry of the Cell
Overview of Key Principles
This chapter introduces five foundational principles essential to cell biology: the characteristics of carbon, the importance of water, selectively permeable membranes, synthesis by polymerization, and self-assembly. Understanding these principles provides insight into the molecular basis of cellular structure and function.
Characteristics of carbon: Carbon's unique bonding properties underpin the diversity and stability of biological molecules.
Characteristics of water: Water's polarity and hydrogen bonding make it the universal solvent and temperature regulator in cells.
Selectively permeable membranes: Membranes control the movement of substances, maintaining cellular integrity.
Synthesis by polymerization: Macromolecules are constructed from monomers through stepwise polymerization.
Self-assembly: Biological structures form spontaneously through noncovalent interactions.
Characteristics of Carbon
Biological Importance and Bonding Properties
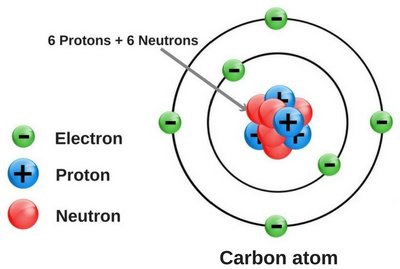
Carbon is the central atom in organic molecules, with a valence of four, allowing it to form four covalent bonds. This versatility enables the formation of complex and stable molecular structures essential for life.
Valence: Carbon (4), Hydrogen (1), Oxygen (2), Nitrogen (3)
Covalent bonds: Carbon forms single, double, or triple bonds, but always maintains four total bonds.
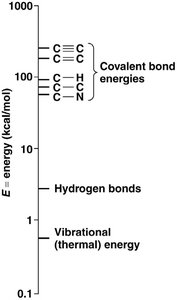
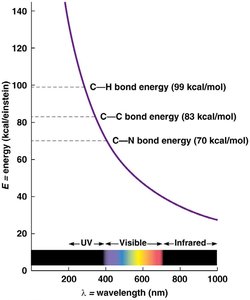
Stability: Multiple covalent bonds increase bond energy, making organic molecules resistant to disruption by visible light.

Covalent Bonding of Carbon Atoms
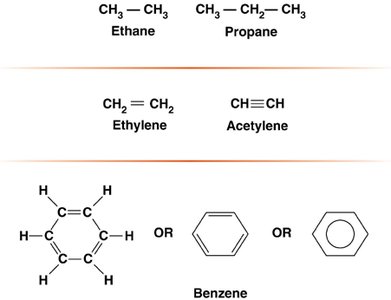
Covalent bonds involve the sharing of electron pairs between atoms. Carbon can form single, double, or triple bonds, contributing to molecular diversity and stability.
Single bond: Sharing one pair of electrons (e.g., methane, CH4)
Double bond: Sharing two pairs (e.g., ethylene, CH2=CH2)
Triple bond: Sharing three pairs (e.g., acetylene, CH≡CH)
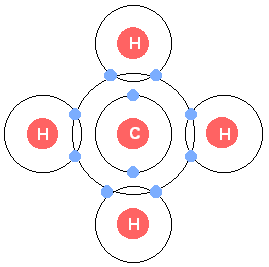
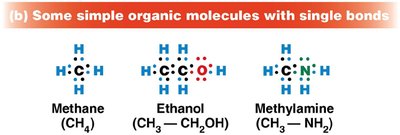
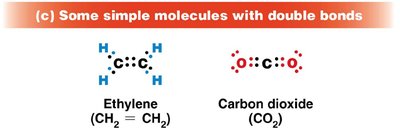

Stability and Diversity of Carbon-Containing Molecules
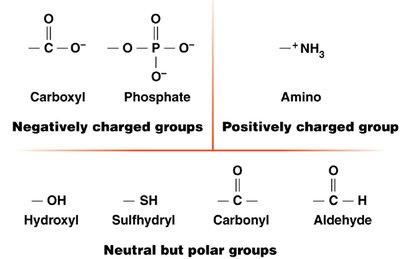
Bond energy reflects molecular stability. Carbon's covalent bonds are strong, making organic molecules stable under visible light but susceptible to higher-energy ultraviolet light. Hydrocarbons (chains/rings of C and H) are not water-soluble, while biological compounds include functional groups (e.g., carboxyl, phosphate, amino) that confer specific chemical properties.
Bond energy: Higher for multiple bonds; visible light cannot break these bonds.
Functional groups: Determine solubility and reactivity.
Bond Polarity
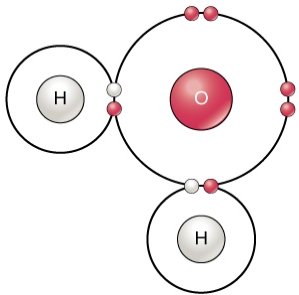
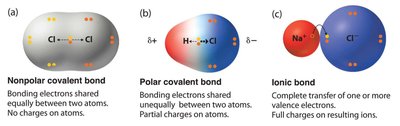
Bond polarity describes the distribution of electrons between atoms. Nonpolar bonds (e.g., C—C, C—H) share electrons equally, while polar bonds (e.g., C—O, C—S) result from differences in electronegativity, increasing water solubility.
Nonpolar bonds: Equal sharing, low solubility.
Polar bonds: Unequal sharing, high solubility.
Stereoisomers and Asymmetric Carbon Atoms
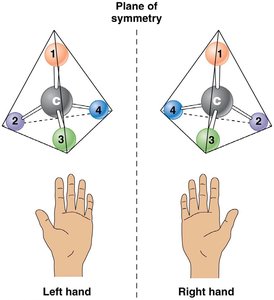
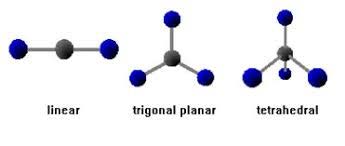
Carbon's tetrahedral geometry allows for stereoisomers—molecules with the same formula but different spatial arrangements. Asymmetric carbon atoms (attached to four different groups) can form two stereoisomers, and a molecule with n asymmetric carbons has 2n possible stereoisomers.
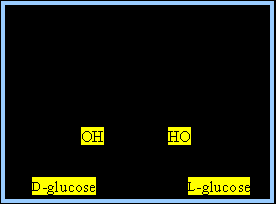
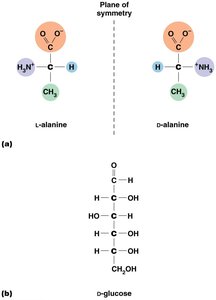
Stereoisomers: Mirror-image molecules (e.g., D- and L-glucose)
Biological significance: Only one stereoisomer is usually biologically active.
The Importance of Water
Water as the Universal Solvent
Water is the most abundant component of cells, comprising 75-85% of cell weight. Its polarity and hydrogen bonding make it essential for biological processes, including solvation, temperature regulation, and molecular interactions.
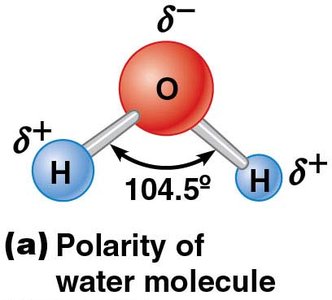
Polarity: Unequal electron distribution creates partial charges.
Cohesiveness: Hydrogen bonds between water molecules.
Temperature-stabilizing capacity: High specific heat and heat of vaporization.
Solvent properties: Hydration spheres around ions and polar molecules.

Polarity and Cohesion of Water Molecules
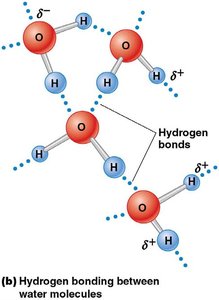
Water's polarity results in strong hydrogen bonding, leading to high surface tension and cohesiveness. This property is critical for cellular structure and function.
Hydrogen bonds: Electrostatic attraction between oxygen and hydrogen atoms.
Surface tension: Allows small objects and organisms to rest on water's surface.

Temperature-Stabilizing Capacity of Water
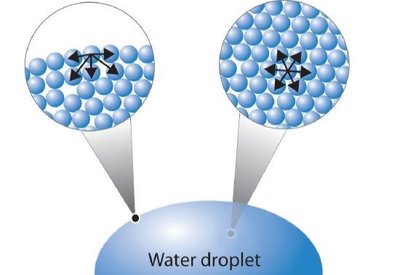
Water's extensive hydrogen bonding gives it a high specific heat and heat of vaporization, allowing it to absorb and release heat slowly. This property is vital for maintaining stable cellular and organismal temperatures.
Specific heat: Amount of heat required to raise temperature by 1ºC.
Heat of vaporization: Water acts as an effective coolant.

Water as an Excellent Solvent
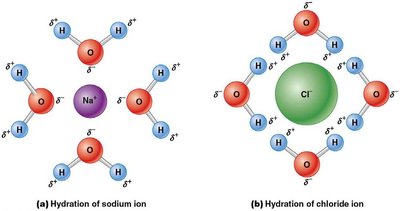
Water dissolves many cellular molecules by forming hydration spheres around ions and polar molecules, preventing their reassociation. Hydrophilic molecules (e.g., sugars, organic acids) dissolve readily, while hydrophobic molecules (e.g., lipids) do not.
Hydration spheres: Water molecules surround ions, stabilizing them in solution.
Hydrophilic vs. hydrophobic: Determines solubility and cellular distribution.
The Importance of Selectively Permeable Membranes
Membrane Structure and Function
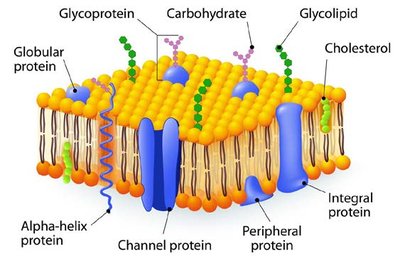
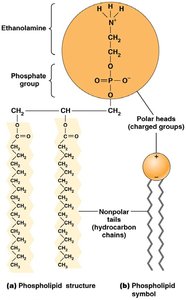
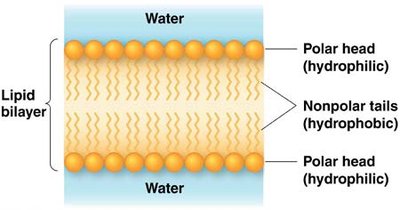
Cell membranes provide a physical barrier between cellular contents and the external environment. Membrane lipids (phospholipids, glycolipids, sterols) are amphipathic, forming a lipid bilayer that is selectively permeable.
Amphipathic lipids: Contain both hydrophobic and hydrophilic regions.
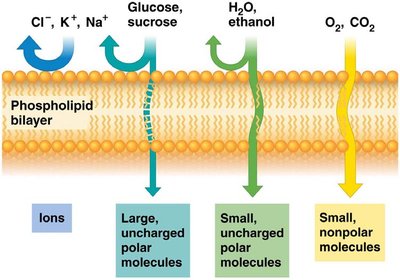
Lipid bilayer: Impermeable to most polar molecules, permeable to nonpolar molecules.
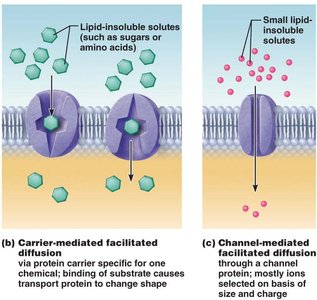
Transport: Protein channels and carriers facilitate movement of ions, sugars, and amino acids.
The Importance of Synthesis by Polymerization
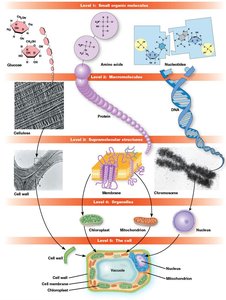
Macromolecules and Cellular Hierarchy
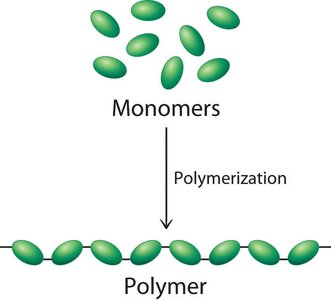
Most cellular structures are composed of macromolecules, which are linear polymers formed by the polymerization of small organic monomers. The cellular hierarchy progresses from small molecules to macromolecules, supramolecular structures, organelles, and the cell itself.
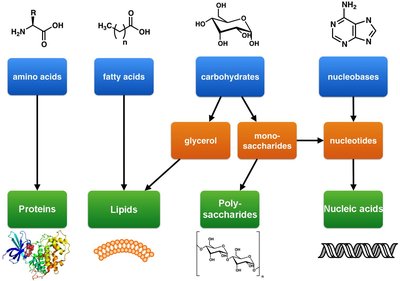
Monomers: Repeating units (e.g., amino acids, nucleotides, monosaccharides).
Polymerization: Stepwise assembly of monomers into polymers.
Hierarchical assembly: Efficient and quality-controlled construction of cellular structures.
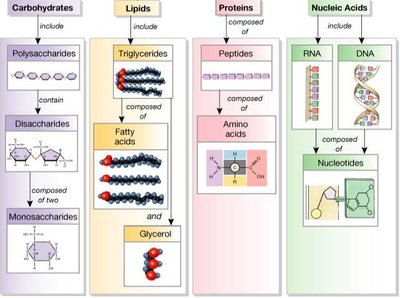
Major Macromolecular Polymers in the Cell
Cells contain three main types of macromolecular polymers: polysaccharides, proteins, and nucleic acids. Each type has distinct structural and functional roles.
Polysaccharides: Structural (cellulose, chitin) and storage (starch, glycogen) functions.
Proteins: Composed of 20 amino acids; functions include structure, defense, transport, catalysis, and signaling.
Nucleic acids: DNA and RNA; informational macromolecules encoding genetic information.
Polymerization Mechanisms
Macromolecules are synthesized by stepwise polymerization of monomers, often requiring carrier molecules. Condensation (dehydration) reactions elongate polymers, while hydrolysis degrades them.
Carrier molecules: ADP/UDP for sugars, tRNA for amino acids, ATP/GTP for nucleotides.
Condensation: Formation of covalent bonds by removing water.
Hydrolysis: Addition of water to break covalent bonds.
The Importance of Self-Assembly
Self-Assembly and Noncovalent Interactions
Macromolecules and cellular structures often self-assemble without external guidance, relying on noncovalent interactions such as hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.
Hydrogen bonds: Weak, but numerous and essential for structure.
Ionic bonds: Electrostatic attraction between oppositely charged ions.
Van der Waals interactions: Weak attractions between atoms in close proximity.
Hydrophobic interactions: Nonpolar groups cluster to minimize contact with water.
Protein Conformation and Molecular Chaperones
Proteins must fold into their native conformation to function. Denaturation disrupts this structure, while renaturation can restore function. Molecular chaperones assist in proper folding and prevent incorrect assembly.
Native conformation: Functional three-dimensional structure.
Denaturation: Loss of structure and function due to environmental changes.
Renaturation: Restoration of structure and function.
Molecular chaperones: Assist in folding, not part of final structure.
Hierarchical Assembly and Cellular Efficiency
Biological structures are assembled hierarchically, providing chemical simplicity, efficiency, and quality control. Defective components are discarded before incorporation, reducing waste.
Hierarchical assembly: From monomers to macromolecules to supramolecular structures.
Quality control: Ensures functional cellular structures.
Summary Table: Types of Macromolecules and Their Functions
Macromolecule | Monomer | Function |
|---|---|---|
Polysaccharides | Monosaccharides | Structure, energy storage |
Proteins | Amino acids | Structure, catalysis, signaling |
Nucleic acids | Nucleotides | Information storage, transmission |
Example: The self-assembly of the tobacco mosaic virus (TMV) demonstrates the limits and advantages of hierarchical assembly in biological systems.
Additional info: These notes expand on brief points with academic context, definitions, and examples to ensure completeness and clarity for cell biology students.
