 Back
BackThe Endomembrane System and Protein Sorting: Structure, Function, and Trafficking
Study Guide - Smart Notes

The Endomembrane System: Structure and Function
Overview of the Endomembrane System
The endomembrane system is a network of interconnected organelles in eukaryotic cells that work together to modify, package, and transport proteins and lipids. This system enables compartmentalization, allowing multiple cellular processes to occur simultaneously without interference.
Key Components: Endoplasmic Reticulum (ER), Golgi Apparatus, Endosomes, Lysosomes, and Vesicles.
Some organelles, such as the nucleolus, are membraneless, but most are membrane-bound to organize intracellular functions.
Intracellular trafficking of proteins and lipids is tightly regulated to maintain cellular function.

Advantages of Compartmentalization
Allows for simultaneous, potentially conflicting processes (e.g., glucose synthesis and breakdown in different compartments).
Specialization of organelles for distinct cellular functions.
Endoplasmic Reticulum (ER)
Structure and Types of ER
The endoplasmic reticulum (ER) is a continuous network of flattened sacs, tubules, and vesicles. The ER is continuous with the outer nuclear membrane and is divided into two main types:
Rough ER (RER): Studded with ribosomes; involved in protein synthesis and processing.
Smooth ER (SER): Lacks ribosomes; involved in lipid synthesis, detoxification, and calcium storage.
Functions of the Rough ER
Protein Synthesis: Ribosomes on the RER synthesize proteins destined for membranes, organelles, or secretion.
Protein Folding: Newly synthesized proteins are folded with the help of chaperone proteins.
Post-Translational Modifications: Proteins undergo modifications such as glycosylation.
Quality Control: Misfolded proteins are recognized and exported for degradation in cytosolic proteasomes.
Functions of the Smooth ER
Lipid Synthesis: Synthesizes phospholipids and steroids.
Detoxification: Hydroxylation of drugs (catalyzed by cytochrome P-450 enzymes) increases solubility for excretion.
Calcium Storage: Especially in muscle cells (sarcoplasmic reticulum).
Carbohydrate Metabolism: Contains glucose-6-phosphatase for glycogen breakdown in liver cells.
Membrane Biosynthesis and Lipid Movement
The ER is the primary source of membrane lipids in eukaryotic cells. Fatty acids are synthesized in the cytoplasm and incorporated into the ER membrane, then transferred to the lumenal side by phospholipid translocators (flippases), creating membrane asymmetry.
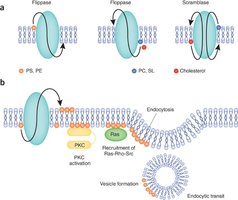
The Golgi Apparatus
Structure and Functional Regions
The Golgi apparatus consists of stacks of flattened membrane-bound sacs called cisternae. It is functionally and physically linked to the ER and is central to protein and membrane trafficking.
Cis-Golgi Network (CGN): Entry side; receives vesicles from the ER.
Medial Golgi: Middle region; further protein modification.
Trans-Golgi Network (TGN): Exit side; proteins are sorted and packaged into vesicles.
Functions of the Golgi Apparatus
Protein Modification: Further glycosylation, sulfation, and phosphorylation of proteins.
Protein Sorting: Directs proteins to their final destinations (e.g., plasma membrane, lysosomes, secretion).
Vesicle Formation: Packages proteins into vesicles for transport.
Models of Protein Movement Through the Golgi
Stationary Cisternae Model: Each cisterna is stable; shuttle vesicles mediate transport between cisternae.
Cisternal Maturation Model: Cisternae are transient and mature from CGN to TGN, carrying proteins forward.
Both models involve vesicle formation and are supported by experimental evidence.
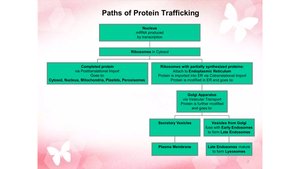
Protein Trafficking and Vesicle Transport
Overview of Protein Trafficking
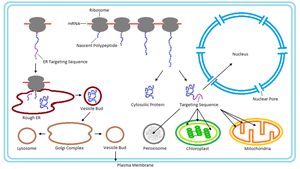
Proteins synthesized in the ER are trafficked to various destinations via vesicles. Each protein contains a specific "tag" (amino acid sequence, hydrophobic domain, or oligosaccharide) that directs it to the correct location.
Exocytosis: Vesicles fuse with the plasma membrane, releasing contents outside the cell.
Endocytosis: The cell engulfs external substances in vesicles formed from the plasma membrane.
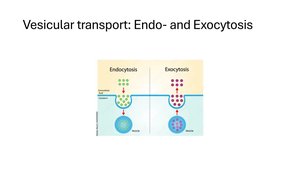
Types of Endocytosis
Phagocytosis: Engulfing large particles (e.g., bacteria).
Pinocytosis: Engulfing fluids and dissolved substances.
Receptor-Mediated Endocytosis: Specific molecules bind to receptors before being internalized.
Secretory Pathways
Constitutive Secretion: Continuous, unregulated release of vesicle contents (e.g., mucus secretion).
Regulated Secretion: Vesicles fuse with the plasma membrane in response to specific signals (e.g., neurotransmitter release).
Polarized Secretion: Exocytosis limited to a specific cell surface (e.g., digestive enzyme secretion in intestinal cells).
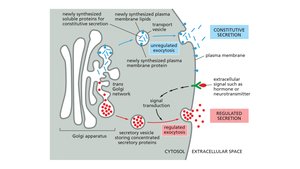
Protein Sorting and Targeting Mechanisms
Sorting Signals and Protein Import
Sorting signals are typically amino acid sequences that determine the destination of a protein. These signals are recognized by receptors and protein translocators, which guide the protein through organelle membranes. Chaperones often assist in the translocation process.

Experimental Evidence for Sorting Signals
Gene cloning and protein engineering experiments demonstrate that sorting signals are both necessary and sufficient for proper protein targeting. Chimeric proteins with relocated signals can be redirected to new destinations.
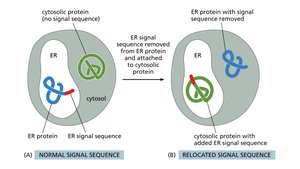
Pathways for Protein Sorting
Posttranslational Import: Proteins synthesized in the cytosol are imported into organelles after translation (e.g., mitochondria, chloroplasts, peroxisomes).
Cotranslational Import: Proteins destined for the endomembrane system are inserted into the ER membrane during synthesis.
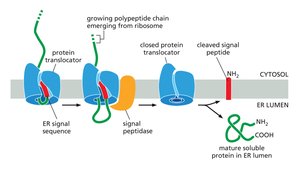
Mechanism of Cotranslational Import
The signal recognition particle (SRP) binds the ER signal sequence on the nascent polypeptide, halting translation.
The SRP-ribosome complex binds to the ER membrane via a translocon.
Translation resumes, and the polypeptide is translocated into the ER lumen or membrane.
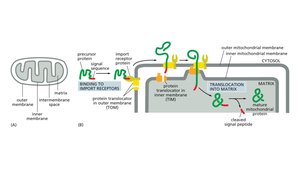
Protein Import into Mitochondria
Proteins destined for mitochondria contain specific signal sequences recognized by import receptors. The TOM and TIM complexes facilitate translocation across the outer and inner mitochondrial membranes, with chaperones assisting in protein unfolding and refolding.
Summary Table: Typical Signal Sequences
Function of Signal | Example of Signal Sequence |
|---|---|
Import into ER | H2N-Met-Met-Ser-Leu-Leu-Val-Gly-Ile-Val-Gly-Tyr-Lys-Lys-Leu-Phe-Gln-... |
Retention in lumen of ER | -Lys-Asp-Glu-Leu-COO- |
Import into mitochondria | H2N-Met-Leu-Ser-Leu-Arg-Gln-Ser-Leu-Tyr-Arg-Thr-Phe-Thr-Leu-Lys-Ser-Leu-... |
Export from nucleus | Met-Glu-Glu-Leu-Ala-Leu-Ala-Ser-Ser-Pro-... |
Import into peroxisomes | -Ser-Lys-Leu |
Additional info: Positively charged amino acids are shown in red, negatively charged in blue, and hydrophobic in green. H2N indicates the N-terminus, COO- the C-terminus.
Key Concepts and Applications
Compartmentalization allows for specialized and simultaneous cellular processes.
Protein sorting relies on signal sequences, receptors, and translocators.
Vesicular transport (exocytosis and endocytosis) is essential for material exchange and membrane composition balance.
Quality control mechanisms in the ER and Golgi ensure only properly folded and modified proteins proceed to their destinations.
