 Back
BackThe Endomembrane System and Protein Sorting: Structure, Function, and Cellular Trafficking
Study Guide - Smart Notes

The Endomembrane System
Overview of the Endomembrane System
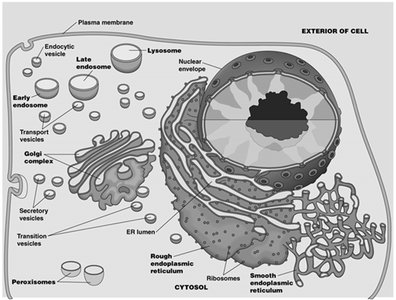
The endomembrane system is a collection of membranous organelles within eukaryotic cells that work together to modify, package, and transport proteins and lipids. It includes the nuclear envelope (NE), endoplasmic reticulum (ER), Golgi complex, endosomes, and lysosomes. These organelles are interconnected by vesicular transport and play central roles in cellular metabolism and protein sorting.
Nuclear Envelope (NE): Surrounds the nucleus and is continuous with the ER.
Endoplasmic Reticulum (ER): Divided into rough (with ribosomes) and smooth (without ribosomes) regions.
Golgi Complex: Processes and sorts proteins and lipids received from the ER.
Endosomes: Involved in sorting and recycling of internalized material.
Lysosomes: Digestive organelles containing hydrolytic enzymes.
Endoplasmic Reticulum (ER)
Structure and Functions of Rough and Smooth ER
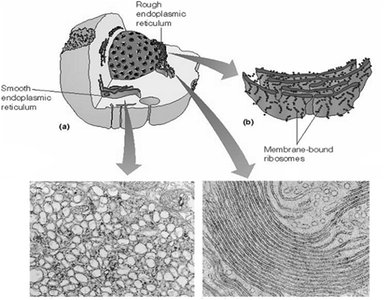
The ER is a network of membranous tubules and sacs. The rough ER is studded with ribosomes and is the site of protein synthesis, while the smooth ER lacks ribosomes and is involved in lipid metabolism and detoxification.
Rough ER:
Site for biosynthesis of membrane and secretory proteins
Initial glycosylation of glycoproteins
Polypeptide folding and regulation
Smooth ER:
Abundant in liver cells
Drug detoxification (cytochrome P450 catalyzed hydroxylation)
Glycogen catabolism
Lipid synthesis
Calcium sequestration (muscle cells)
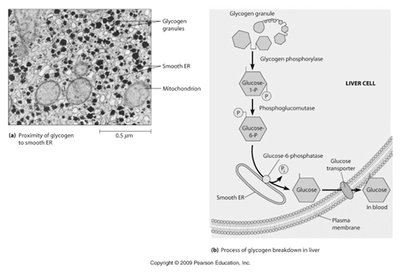
Smooth ER and Glycogen Catabolism in Liver Cells
The smooth ER in liver cells is closely associated with glycogen granules and mitochondria. It plays a key role in the breakdown of glycogen to glucose, which is then released into the bloodstream. The enzyme glucose-6-phosphatase, a marker for the smooth ER, is essential for this process.
Glucose-6-phosphatase: Converts glucose-6-phosphate to glucose, allowing its release from liver cells.
Muscle and brain cells: Lack this enzyme, so glucose cannot be released from these tissues.
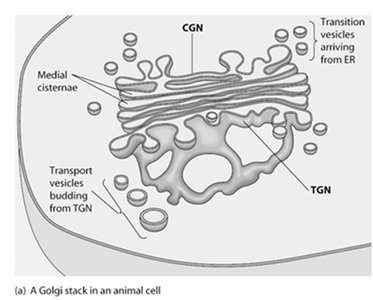
Golgi Complex
Structure and Functional Regions
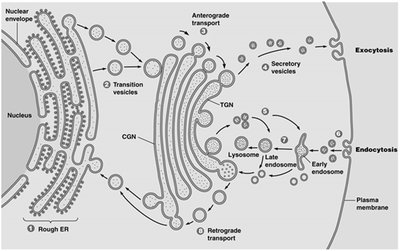
The Golgi complex is composed of stacks of flattened sacs called cisternae. It has three distinct regions: the cis-Golgi network (CGN), medial cisternae, and trans-Golgi network (TGN), each with specific roles in protein processing and sorting.
CGN: Receives transition vesicles from the ER.
Medial cisternae: Processing of proteins and lipids.
TGN: Distributes processed molecules via transport vesicles.
Directionality: Anterograde (forward) and retrograde (backward) transport.
Intracellular Trafficking: ER and Golgi
Proteins synthesized in the ER are transported to the Golgi for further processing. The Golgi sorts and packages proteins for delivery to their final destinations, including lysosomes, the plasma membrane, or secretion outside the cell.
Anterograde transport: ER → CGN → medial cisternae → TGN → plasma membrane or lysosome.
Retrograde transport: Retrieval of ER-resident proteins from Golgi back to ER.
Compartmentalization of Glycosylation and Protein Modification
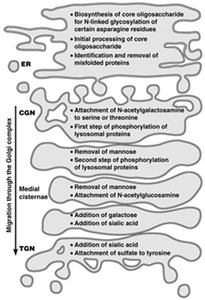
Glycosylation and other modifications of proteins occur in specific compartments of the ER and Golgi. The extent of oligosaccharide processing can be monitored by sensitivity to endoglycosidase H (Endo H).
High-mannose oligosaccharides: Endo H sensitive.
Complex carbohydrates: Endo H resistant.
Mannosidase II: Enzyme involved in glycan processing.
Retention and Retrieval of ER Resident Proteins
ER resident proteins are maintained in the ER by retention and retrieval mechanisms. A retrieval tag, such as KDEL in mammals, ensures that proteins are returned to the ER if they escape to the Golgi.
KDEL sequence: Necessary and sufficient for ER localization.
KDEL receptors: Located in the CGN, capture ER proteins and mediate retrograde transport.
Lysosomal Targeting and Disease
Mannose-6-Phosphate Tag and Lysosomal Enzyme Targeting
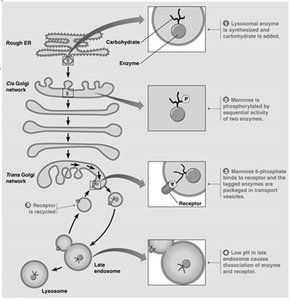
Lysosomal enzymes are targeted to lysosomes by a mannose-6-phosphate (M6P) tag, which acts as a zip code. Only lysosomal enzymes receive this tag, and the M6P receptor ensures delivery to lysosomes.
M6P tag: Added in the Golgi, recognized by M6P receptors.
I-cell disease: Genetic defect in phosphotransferase leads to secretion of hydrolases and accumulation of waste in lysosomes.
Secretory Pathway and Exocytosis
Pulsed-Chase Experiments and Secretory Pathway Evidence
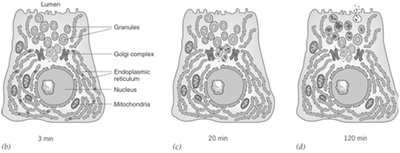
Pulsed-chase experiments, such as those by George Palade, provide evidence for the secretory pathway. Proteins are labeled in the ER, move to the Golgi, and then to secretory vesicles before being released by exocytosis.
Pulse: Label proteins in ER.
Chase: Track movement to Golgi and vesicles over time.
Exocytosis: Mechanism and Types
Exocytosis is the process by which secretory vesicles fuse with the plasma membrane to release their contents outside the cell. This process is essential for secretion of hormones, enzymes, and other molecules.
Constitutive exocytosis: Continuous secretion.
Regulated exocytosis: Triggered by specific signals.
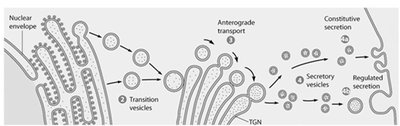
Endocytosis and Cellular Uptake
Endocytosis: Mechanism and Types
Endocytosis is the process by which cells internalize macromolecules and particles from the extracellular environment. It involves invagination, fission, and formation of vesicles.
Endocytosis: Uptake for digestion in lysosomes.
Transcytosis: Transport across the cell.
Autophagy: Digestion of internal organelles for recycling.
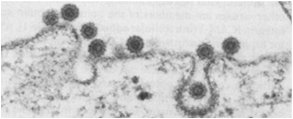
Receptor-Mediated Endocytosis (RME) and LDL Transport
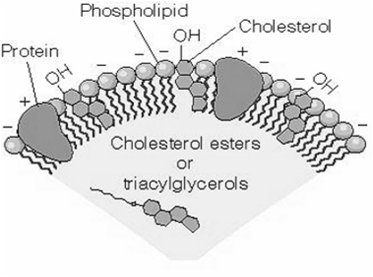
Structure and Function of LDL Particles
Low-density lipoprotein (LDL) particles are the main carriers of cholesterol in human plasma. They consist of a phospholipid monolayer, embedded apoprotein B-100, and internal cholesterol and cholesteryl esters.
Apoprotein B-100: Mediates binding to cell surface receptors.
LDL uptake: Occurs via receptor-mediated endocytosis.
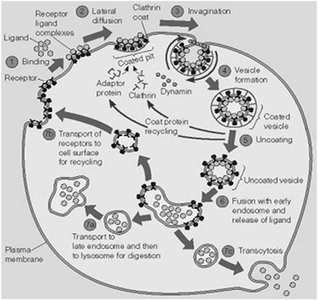
Mechanism of LDL Uptake: Clathrin-Coated Pits and Vesicles
LDL particles bind to LDL receptors, cluster in clathrin-coated pits, and are internalized by endocytosis. Clathrin-coated vesicles are formed, uncoated, and fuse with early endosomes. Acidic pH in late endosomes dissociates LDL from its receptor, allowing LDL to be delivered to lysosomes for breakdown.
Clathrin: Protein coat for vesicle formation.
Dynamin: GTPase required for vesicle pinching off.
Receptor recycling: LDL receptor returns to plasma membrane.
Familial Hypercholesterolemia (FH)
FH is a genetic disorder characterized by high blood cholesterol due to defects in LDL receptor-mediated endocytosis. This leads to impaired feedback regulation of cholesterol synthesis and early onset of cardiovascular disease.
Heterozygous FH: 2x increase in plasma cholesterol, heart attacks at age 30-40.
Homozygous FH: 6-10x increase, heart attacks in childhood.
Protein-Mediated Vesicle Targeting and Fusion
Coat Proteins, Rab Proteins, and SNAREs
Vesicle targeting and fusion are mediated by specific proteins. Coat proteins (clathrin, COPI, COPII) select cargo and routes. Rab proteins specify vesicle identity and destination. SNARE proteins (v-SNAREs and t-SNAREs) mediate membrane fusion, and GTP-bound G proteins provide energy.
Coat proteins: Clathrin (PM and TGN), COPI (CGN Golgi), COPII (ER to CGN).
Rab proteins: Vesicle/membrane targeting.
SNARE proteins: Fusion mediators.
GTP-bound G proteins: Energy source.
The SNARE Hypothesis
The SNARE hypothesis states that vesicle sorting and targeting involve v-SNAREs (on vesicles) and t-SNAREs (on target membranes). These complementary proteins ensure specificity in vesicle fusion.
v-SNAREs: Vesicle SNAREs.
t-SNAREs: Target membrane SNAREs.
Phagocytosis, Pinocytosis, and Autophagy
Phagocytosis and Pinocytosis
Phagocytosis is the ingestion of large particles or cells, mainly by specialized phagocytes such as neutrophils and macrophages. Pinocytosis is the uptake of fluids and small molecules.
Phagocytosis: Defense and debris clearance.
Pinocytosis: "Cellular drinking" of extracellular fluid.
Autophagy: Biological Recycling
Autophagy is the process by which cells degrade and recycle damaged or unneeded organelles. Defects in autophagy and lysosomal digestion can lead to various diseases, including glycogen storage diseases, lysosomal storage diseases, and possibly Crohn's disease.
Autophagic vacuole: Organelle wrapped in double membrane from ER.
Diseases: Type II glycogenesis, Hurler syndrome, Hunter syndrome, Tay-Sachs disease.
Peroxisomes: Structure and Function
Exclusion from the Endomembrane System
Peroxisomes are single-membrane-bound organelles not derived from the ER and are therefore excluded from the endomembrane system. They proliferate as cells grow and divide, a process called biogenesis, which requires peroxins.
Biogenesis: Increase in peroxisome number as cells grow.
Peroxins: Proteins required for peroxisome biogenesis.
Functions of Peroxisomes
Most peroxisomal functions are linked to hydrogen peroxide metabolism. They play essential roles in detoxification, fatty acid oxidation, metabolism of nitrogen-containing compounds, and catabolism of unusual substances.
Hydrogen peroxide metabolism
Detoxification of harmful compounds
β-oxidation of fatty acids
Metabolism of nitrogen-containing compounds
Catabolism of unusual substances
