 Back
BackThe Eukaryotic Cell Cycle: Regulation, Control, and Key Molecular Players
Study Guide - Smart Notes

Chapter 17 – The Cell Cycle
Overview of the Cell Cycle
The cell cycle is a highly regulated series of events that leads to cell growth, chromosome duplication, and cell division. It ensures that genetic material is accurately replicated and distributed to daughter cells. The cycle is divided into distinct phases, each with specific molecular and structural events.
Cell Growth and Chromosome Duplication: The cell increases in size and duplicates its chromosomes to prepare for division.
Chromosome Segregation: Duplicated chromosomes are separated into two sets.
Cell Division: The cell physically divides to form two daughter cells.
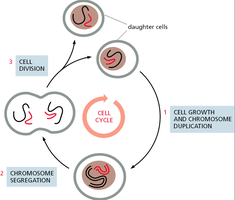
Major Events and Phases of the Cell Cycle
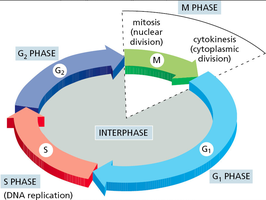
The cell cycle consists of four main phases: G1, S, G2, and M. Interphase (G1, S, G2) is the period of cell growth and DNA replication, while M phase includes mitosis and cytokinesis.
G1 phase: Cell grows and prepares for DNA synthesis.
S phase: DNA replication occurs, resulting in duplicated chromosomes.
G2 phase: Further growth and preparation for mitosis.
M phase: Mitosis (nuclear division) and cytokinesis (cytoplasmic division) occur.
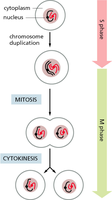
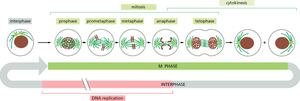
Live Cell Imaging and Staining of the Cell Cycle
Live cell imaging techniques allow visualization of cell cycle progression in real time. Fluorescent markers can distinguish cells in different phases based on DNA content or specific protein expression.
Fluorescent labeling: Used to track DNA replication and mitosis.
Time-lapse microscopy: Captures dynamic changes in cell morphology and nuclear events.
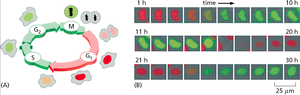
Analysis of Cell Cycle Phases by DNA Content
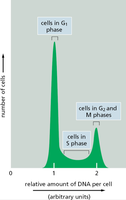
Flow cytometry can be used to analyze the DNA content of cells, distinguishing between G1, S, and G2/M phases based on the amount of DNA per cell.
G1 phase: Cells have a single set of DNA (2N).
S phase: DNA content increases as replication proceeds.
G2/M phase: Cells have double the DNA content (4N).
Cell Cycle Checkpoints and Control
Cell Cycle Checkpoints
Checkpoints are surveillance mechanisms that ensure the fidelity of cell division. They prevent progression to the next phase until critical processes are completed.
G1/S checkpoint: Checks for DNA damage and adequate cell size before DNA replication.
G2/M checkpoint: Ensures all DNA is replicated and undamaged before mitosis.
Metaphase-to-anaphase transition: Verifies that all chromosomes are properly attached to the spindle apparatus before segregation.
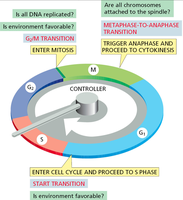
Cell Cycle Control System
The cell cycle is regulated by a complex network of proteins, primarily cyclins and cyclin-dependent kinases (Cdks). These proteins form complexes that drive the cell through different phases by phosphorylating target substrates.
Cyclins: Regulatory proteins whose levels fluctuate throughout the cell cycle.
Cyclin-dependent kinases (Cdks): Serine/threonine kinases activated by binding to cyclins.

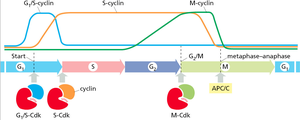
Sequential Activation of Cyclin-Cdk Complexes
Different cyclin-Cdk complexes are activated at specific points in the cell cycle, ensuring orderly progression through the phases.
G1/S-Cdk: Triggers entry into S phase.
S-Cdk: Initiates DNA replication.
M-Cdk: Promotes entry into mitosis.
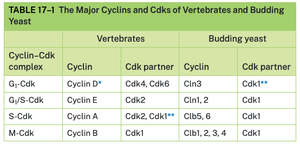
Major Cyclins and Cdks in Vertebrates and Yeast
The table below summarizes the main cyclin-Cdk complexes in vertebrates and budding yeast, highlighting the conservation and specialization of cell cycle regulators.
Cyclin–Cdk complex | Cyclin (Vertebrates) | Cdk partner (Vertebrates) | Cyclin (Budding yeast) | Cdk partner (Budding yeast) |
|---|---|---|---|---|
G1-Cdk | Cyclin D | Cdk4, Cdk6 | Cln3 | Cdk1 |
G1/S-Cdk | Cyclin E | Cdk2 | Cln1, 2 | Cdk1 |
S-Cdk | Cyclin A | Cdk2, Cdk1 | Clb5, 6 | Cdk1 |
M-Cdk | Cyclin B | Cdk1 | Clb1, 2, 3, 4 | Cdk1 |
Regulation of Cyclin-Cdk Activity
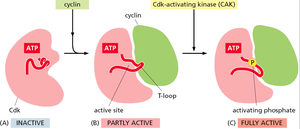
Cdk Activation
Cdk activity is tightly regulated by cyclin binding and phosphorylation. Cyclin binding partially activates the Cdk, while full activation requires phosphorylation by a Cdk-activating kinase (CAK).
Inactive Cdk: No cyclin bound, T-loop blocks active site.
Partly active: Cyclin binding exposes the active site.
Fully active: CAK phosphorylates the T-loop, enabling full activity.
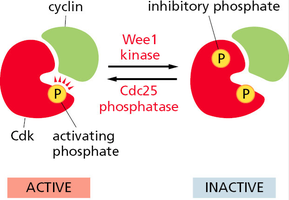
Cdk Inactivation
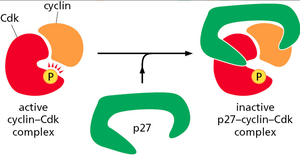
Cdk activity can be inhibited by phosphorylation at specific inhibitory sites (by Wee1 kinase) or by binding of Cdk inhibitor proteins (CKIs) such as p27. Removal of inhibitory phosphates by Cdc25 phosphatase reactivates Cdks.
Wee1 kinase: Adds inhibitory phosphate, inactivating Cdk.
Cdc25 phosphatase: Removes inhibitory phosphate, activating Cdk.
CKIs (e.g., p27): Bind and inactivate cyclin-Cdk complexes.
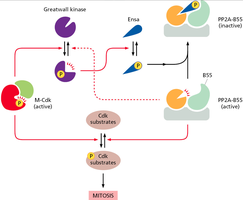
Regulation by Protein Phosphatases
Protein phosphatases such as PP2A-B55 counteract Cdk activity by removing phosphates from Cdk substrates. Their activity is regulated by kinases and inhibitory proteins, contributing to the precise timing of mitotic events.
PP2A-B55: Dephosphorylates Cdk substrates, opposing mitotic entry.
Greatwall kinase: Inhibits PP2A-B55 via phosphorylation of regulatory proteins.
Positive Feedback in M-Cdk Activation
M-Cdk activation is reinforced by positive feedback loops, ensuring a rapid and irreversible transition into mitosis. Active M-Cdk promotes further activation of Cdc25 and inhibition of Wee1, amplifying its own activation.
Positive feedback: Ensures switch-like entry into mitosis.
Coordination: Synchronizes mitotic events across the cell.
Summary Table: Key Regulatory Proteins in the Cell Cycle
Protein/Complex | Function |
|---|---|
Cyclin | Regulates Cdk activity; levels fluctuate during the cell cycle |
Cdk | Phosphorylates target proteins to drive cell cycle transitions |
CAK | Activates Cdk by phosphorylation |
Wee1 kinase | Inhibits Cdk by phosphorylation |
Cdc25 phosphatase | Activates Cdk by removing inhibitory phosphate |
CKIs (e.g., p27) | Inhibit cyclin-Cdk complexes |
PP2A-B55 | Dephosphorylates Cdk substrates, opposing mitosis |
Greatwall kinase | Inhibits PP2A-B55, promoting mitosis |
Additional info: The cell cycle is fundamental to development, tissue repair, and cancer biology. Disruption of cell cycle regulation can lead to uncontrolled cell proliferation and tumorigenesis.
