 Back
BackThe Macromolecules of the Cell: Structure, Function, and Organization
Study Guide - Smart Notes

The Macromolecules of the Cell
Introduction to Cellular Macromolecules
Cells are composed of a variety of macromolecules, each with distinct structures and functions. These macromolecules are primarily synthesized from a limited set of small molecules and are essential for the structure, function, and regulation of the cell.
Proteins: Polymers of amino acids with diverse roles in catalysis, structure, transport, and regulation.
Nucleic acids: DNA and RNA, responsible for storage, transmission, and expression of genetic information.
Polysaccharides: Long chains of sugars, serving as energy storage and structural components.
Lipids: Hydrophobic molecules involved in membrane structure, energy storage, and signaling.
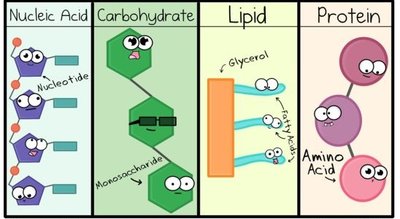
Common Small Molecules in Cells
Most biological macromolecules are synthesized from about 30 common small molecules, including amino acids, aromatic bases, sugars, and lipids. These serve as the building blocks for larger polymers.
Kind of Molecules | Number Present | Names of Molecules | Role in Cell |
|---|---|---|---|
Amino acids | 20 | See Table 3-2 | Monomeric units of all proteins |
Aromatic bases | 5 | Adenine, Cytosine, Guanine, Thymine, Uracil | Components of nucleic acids |
Sugars | varies | Ribose, Deoxyribose, Glucose | Components of RNA, DNA, energy metabolism |
Lipids | varies | Fatty acids, Cholesterol | Components of membranes, energy storage |
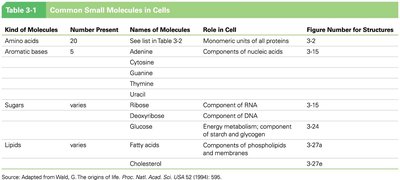
Polymerization and Macromolecule Synthesis
Dehydration (Condensation) Reactions
Polymers are synthesized by dehydration reactions, in which activated monomers are linked together by the removal of water. This process is fundamental to the formation of proteins, nucleic acids, and polysaccharides.
Dehydration reaction: Joins monomers by removing a water molecule, forming a covalent bond.
Hydrolysis: The reverse process, breaking polymers into monomers by adding water.
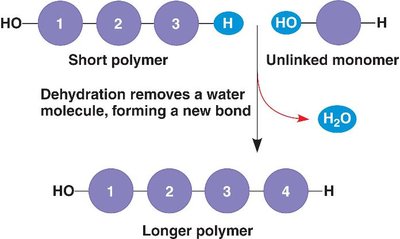
Proteins
Overview and Functions
Proteins are the most versatile macromolecules in the cell, performing a wide range of functions. They are polymers of amino acids, each with a unique sequence and structure.
Enzymes: Catalyze biochemical reactions.
Structural proteins: Provide support and shape (e.g., keratin, collagen).
Motility proteins: Involved in movement (e.g., myosin).
Regulatory proteins: Control cellular processes.
Transport proteins: Move substances across membranes (e.g., hemoglobin).
Signaling proteins: Mediate communication between cells.
Receptor proteins: Receive and transmit signals.
Defensive proteins: Protect against disease (e.g., antibodies).
Storage proteins: Store amino acids or ions.
Amino Acids: The Monomers of Proteins
Proteins are composed of 20 standard amino acids, each with a central (α) carbon, an amino group, a carboxyl group, a hydrogen atom, and a unique side chain (R group). The properties of the R group determine the characteristics of each amino acid.
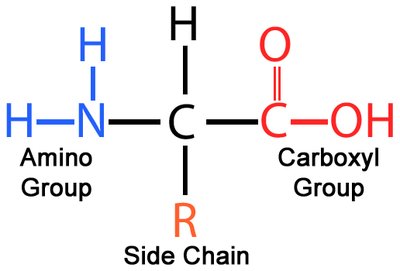
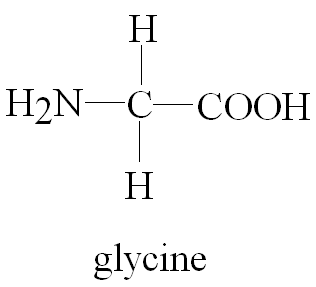
Glycine is the simplest amino acid, with a hydrogen as its R group.
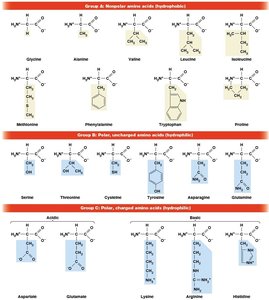
Classification of Amino Acids
Amino acids are classified based on the properties of their R groups:
Nonpolar (hydrophobic)
Polar, uncharged (hydrophilic)
Polar, charged (hydrophilic)
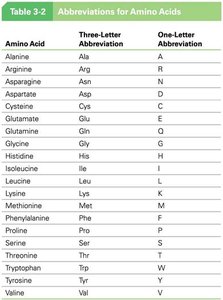
Abbreviations for Amino Acids
Amino Acid | Three-Letter Abbreviation | One-Letter Abbreviation |
|---|---|---|
Alanine | Ala | A |
Arginine | Arg | R |
Asparagine | Asn | N |
Aspartate | Asp | D |
Cysteine | Cys | C |
Glutamate | Glu | E |
Glutamine | Gln | Q |
Glycine | Gly | G |
Histidine | His | H |
Isoleucine | Ile | I |
Leucine | Leu | L |
Lysine | Lys | K |
Methionine | Met | M |
Phenylalanine | Phe | F |
Proline | Pro | P |
Serine | Ser | S |
Threonine | Thr | T |
Tryptophan | Trp | W |
Tyrosine | Tyr | Y |
Valine | Val | V |
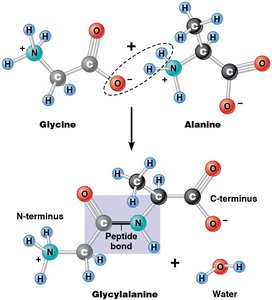
Peptide Bond Formation and Protein Structure
Amino acids are linked by peptide bonds through condensation reactions, forming polypeptides with directionality (N-terminus to C-terminus).
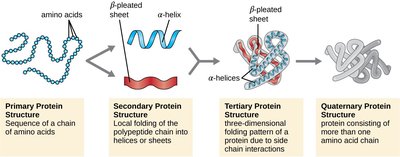
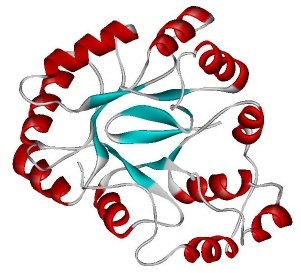
Levels of Protein Structure
Primary structure: Linear sequence of amino acids.
Secondary structure: Local folding into α helices and β sheets, stabilized by hydrogen bonds.
Tertiary structure: Overall 3D conformation, determined by interactions among R groups.
Quaternary structure: Association of multiple polypeptide subunits.
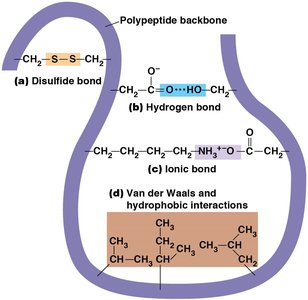
Stabilizing Interactions in Protein Structure
Covalent bonds: Disulfide bridges between cysteine residues.
Noncovalent interactions: Hydrogen bonds, ionic bonds, van der Waals forces, and hydrophobic interactions.
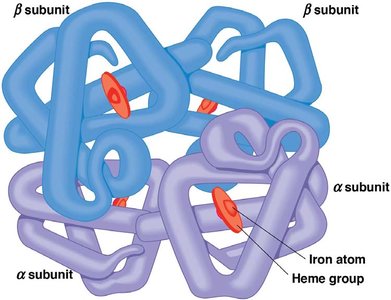
Examples of Protein Structure
Monomeric proteins: Single polypeptide chain.
Multimeric proteins: Multiple polypeptide chains (e.g., hemoglobin is a tetramer).
Fibrous vs. Globular Proteins
Fibrous proteins: Elongated, insoluble, provide structural support (e.g., keratin, collagen).
Globular proteins: Compact, soluble, functional roles (e.g., enzymes, antibodies).
Nucleic Acids
Structure and Function
Nucleic acids are linear polymers of nucleotides. DNA stores genetic information, while RNA is involved in its expression and regulation.
DNA: Deoxyribonucleic acid, double-stranded, repository of genetic information.
RNA: Ribonucleic acid, single-stranded, involved in protein synthesis and regulation.
Nucleotides and Nucleosides
Nucleotide: Composed of a five-carbon sugar, phosphate group, and nitrogenous base.
Nucleoside: Sugar and base, without phosphate.
Pyrimidines: Cytosine, thymine (DNA), uracil (RNA).
Purines: Adenine, guanine.
Phosphodiester Bonds and Directionality
Nucleotides are linked by 3',5' phosphodiester bonds, giving nucleic acids directionality (5' to 3').
Base Pairing
DNA: A pairs with T (2 hydrogen bonds), G pairs with C (3 hydrogen bonds).
RNA: A pairs with U, G pairs with C.
Polysaccharides
Structure and Function
Polysaccharides are long chains of monosaccharides, serving as energy storage (starch, glycogen) or structural components (cellulose, chitin).
Monosaccharides: Simple sugars, classified by carbon number (triose, tetrose, pentose, hexose, heptose).
Disaccharides: Two monosaccharides linked (e.g., maltose, lactose, sucrose).
Storage polysaccharides: Starch (plants), glycogen (animals).
Structural polysaccharides: Cellulose (plants), chitin (fungi, arthropods).
Lipids
Structure and Function
Lipids are hydrophobic molecules not formed by linear polymerization. They are important for membrane structure, energy storage, and signaling.
Fatty acids: Building blocks of many lipids, amphipathic with hydrophilic head and hydrophobic tail.
Triacylglycerols: Glycerol with three fatty acids, function in energy storage.
Phospholipids: Major membrane components, amphipathic.
Glycolipids: Membrane components with carbohydrate groups.
Steroids: Four-ring structure, includes cholesterol and hormones.
Terpenes: Derived from isoprene, includes vitamins and pigments.
Saturation of Fatty Acids
Saturated fatty acids: No double bonds, straight chains, solid at room temperature.
Unsaturated fatty acids: One or more double bonds, bent chains, liquid at room temperature.
Trans fats: Unsaturated with trans double bonds, associated with health risks.
Summary Table: Major Macromolecules and Their Monomers
Macromolecule | Monomer | Bond Type | Main Function |
|---|---|---|---|
Protein | Amino acid | Peptide bond | Catalysis, structure, transport, regulation |
Nucleic acid | Nucleotide | Phosphodiester bond | Genetic information storage and transfer |
Polysaccharide | Monosaccharide | Glycosidic bond | Energy storage, structure |
Lipid | Fatty acid, glycerol | Ester bond | Membranes, energy storage, signaling |
