 Back
BackTransport Across Membranes: Overcoming the Permeability Barrier
Study Guide - Smart Notes

Transport Across Membranes: Overcoming the Permeability Barrier
Introduction
The permeability barrier of cell membranes is essential for maintaining cellular homeostasis. Selective transport of molecules and ions is achieved through various mechanisms, allowing cells to regulate their internal environment and respond to external changes. Membranes are selectively permeable, permitting certain substances to cross while restricting others.
Mechanisms of Solute Transport
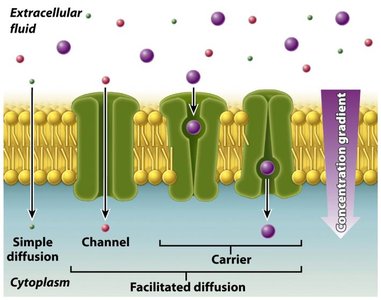
Solutes such as dissolved gases, ions, and small organic molecules cross membranes via three primary mechanisms:
Simple Diffusion: Direct, unaided movement of solutes down their concentration gradient.
Facilitated Diffusion: Transport proteins assist solutes to move down their concentration gradient without energy input.
Active Transport: Transport proteins move solutes against their concentration gradient, requiring energy.
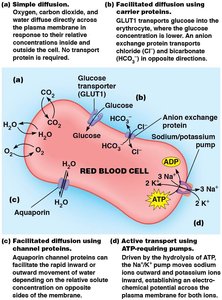
Simple Diffusion
Simple diffusion is the movement of molecules such as oxygen, carbon dioxide, and water directly across the lipid bilayer, driven by concentration differences. No transport protein or energy is required.
Example: Oxygen gas traverses the erythrocyte plasma membrane by simple diffusion, moving from areas of high concentration (lungs) to low concentration (body tissues).

Facilitated Diffusion
Facilitated diffusion involves transport proteins that help solutes move down their concentration gradient. This process does not require energy and is essential for molecules that cannot cross the membrane unaided.
Carrier Proteins: Bind solute molecules, undergo conformational changes, and release solutes on the opposite side.
Channel Proteins: Form hydrophilic channels for solute passage.

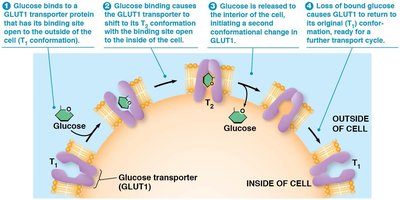
Carrier Protein Mechanism
Carrier proteins alternate between two conformational states to transport solutes:
Solute binds to the carrier protein on one side.
Protein changes conformation, exposing the solute to the other side.
Solute is released, and the protein returns to its original state.
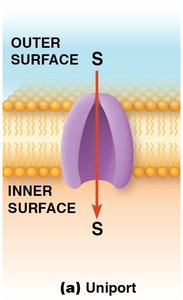
Types of Carrier Protein Transport
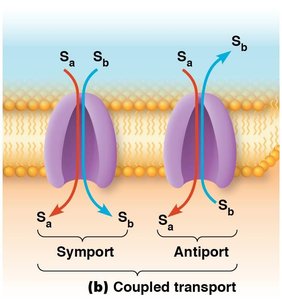
Uniport: Transports a single solute across the membrane. The protein is called a uniporter.
Coupled Transport: Transports two solutes simultaneously. If both move in the same direction, it is symport (cotransport); if in opposite directions, it is antiport (countertransport).
Active Transport
Active transport moves solutes against their concentration gradient and requires energy. It is categorized as:
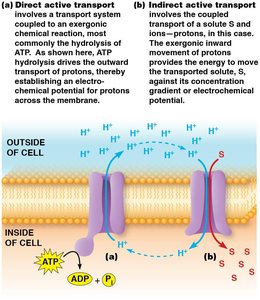
Direct Active Transport (Primary): Coupled directly to an exergonic chemical reaction, usually ATP hydrolysis. Transport proteins driven by ATP are called ATPase pumps.
Indirect Active Transport (Secondary): Depends on the simultaneous transport of two solutes. Favorable movement of one solute down its gradient drives the unfavorable movement of another up its gradient. Can be symport or antiport.
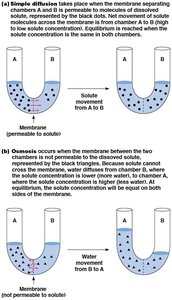
Osmosis
Osmosis is the diffusion of water across a selectively permeable membrane. Water moves toward the region of higher solute concentration. Osmotic movement depends on the relative osmolarity (total solute concentration) inside versus outside the cell.
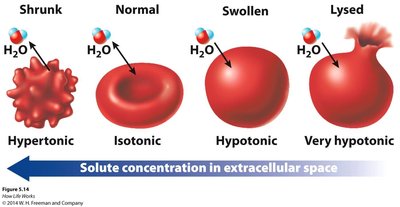
Hypertonic: Higher solute concentration outside the cell; water moves out, cell shrinks.
Hypotonic: Lower solute concentration outside the cell; water moves in, cell swells.
Isotonic: Equal solute concentration; no net water movement.
Effects of Osmosis on Cells
Cells maintain their size and shape using active transport to regulate osmotic balance. Human red blood cells avoid shrinking or bursting by keeping their intracellular environment isotonic with the blood.
Example: In a hypertonic solution, cells lose water and shrink; in a hypotonic solution, cells gain water and may burst.

Summary Table: Types of Membrane Transport
Transport Type | Energy Requirement | Direction | Example |
|---|---|---|---|
Simple Diffusion | No | Down gradient | O2, CO2 |
Facilitated Diffusion | No | Down gradient | Glucose via GLUT1 |
Active Transport (Direct) | Yes (ATP) | Against gradient | Na+/K+ pump |
Active Transport (Indirect) | Yes (gradient) | Against gradient | Na+/glucose symport |
Osmosis | No | Water toward higher solute | Red blood cell in hypotonic solution |
Key Equations
Fick's Law of Diffusion:
Osmotic Pressure:
ATP Hydrolysis (for active transport):
Additional info: Academic context was added to clarify mechanisms, provide definitions, and summarize key points for exam preparation.
