 Back
BackCh 8 - Transport Across Membranes: Overcoming the Permeability Barrier
Study Guide - Smart Notes

Transport Across Membranes: Overcoming the Permeability Barrier
Introduction
The selective transport of molecules and ions across cellular membranes is essential for maintaining homeostasis, acquiring nutrients, and removing waste products. Biological membranes act as barriers, but cells have evolved specialized mechanisms to regulate the movement of substances, ensuring that transport occurs efficiently and selectively.
Major Transport Processes
Overview of Membrane Transport
Transport across membranes can be classified into three main categories: simple diffusion, facilitated diffusion, and active transport. Each process differs in its energy requirements, directionality, and the types of molecules transported.
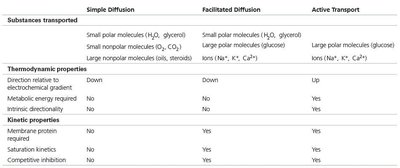
Simple Diffusion | Facilitated Diffusion | Active Transport | |
|---|---|---|---|
Substances transported | Small polar molecules (H2O, glycerol) Small nonpolar molecules (O2, CO2) Large nonpolar molecules (oils, steroids) | Small polar molecules (H2O, glycerol) Large polar molecules (glucose) Ions (Na+, K+, Ca2+) | Large polar molecules (glucose) Ions (Na+, K+, Ca2+) |
Direction relative to electrochemical gradient | Down | Down | Up |
Metabolic energy required | No | No | Yes |
Intrinsic directionality | No | Yes | Yes |
Membrane protein required | No | Yes | Yes |
Saturation kinetics | No | Yes | Yes |
Competitive inhibition | No | Yes | Yes |
Simple Diffusion
Unassisted Movement Down the Gradient
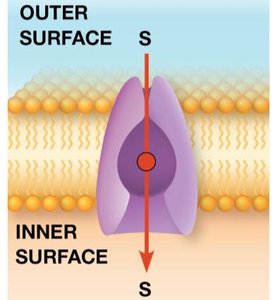
Simple diffusion is the passive movement of small or nonpolar molecules across the lipid bilayer, driven by the concentration gradient. No energy input or transport protein is required.
Key Point 1: Only small nonpolar molecules (e.g., O2, CO2, lipids) and some small polar molecules (e.g., H2O) can diffuse directly through the membrane.
Key Point 2: The rate of diffusion is proportional to the molecule's partition coefficient, which reflects its solubility in oil versus water.
Key Point 3: The direction of diffusion is always down the concentration gradient, moving toward equilibrium.
Key Point 4: If the membrane is impermeable to a solute, water will move by osmosis to balance solute concentrations.
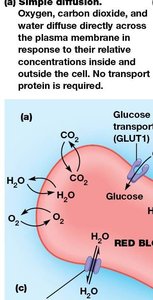
Example: Simple Diffusion in Red Blood Cells
Oxygen, carbon dioxide, and water diffuse directly across the plasma membrane of erythrocytes in response to their relative concentrations inside and outside the cell.

Facilitated Diffusion
Protein-Mediated Movement Down the Gradient
Facilitated diffusion enables the passive movement of large, polar molecules and ions across membranes via specific transport proteins. This process does not require metabolic energy but depends on the presence of carrier or channel proteins.
Key Point 1: Carrier proteins alternate between conformational states to transport molecules such as glucose (GLUT1 transporter) and ions (anion exchange protein).
Key Point 2: Channel proteins form hydrophilic pores for rapid movement of ions (ion channels), water (aquaporins), or other solutes (porins).
Key Point 3: Transport can be classified as uniport (single solute), symport (two solutes in the same direction), or antiport (two solutes in opposite directions).
Examples of Facilitated Diffusion in Erythrocytes
GLUT1 transporter: Moves glucose into the cell where its concentration is lower.
Anion exchange protein: Exchanges chloride (Cl-) and bicarbonate (HCO3-) ions in opposite directions (antiport).
Aquaporins: Facilitate rapid water movement depending on solute concentration differences.

Active Transport
Protein-Mediated Movement Up the Gradient
Active transport moves molecules and ions against their electrochemical gradients, requiring energy input. This process is essential for maintaining concentration differences across membranes, which are critical for cell function.
Key Point 1: Primary active transport uses ATP hydrolysis to drive transport (e.g., Na+/K+ pump).
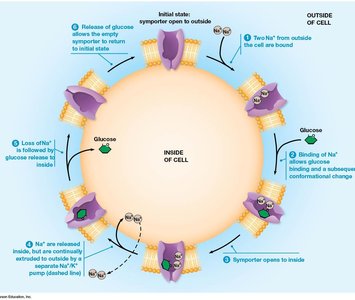
Key Point 2: Secondary (indirect) active transport uses the energy stored in ion gradients established by primary active transport (e.g., Na+/glucose symporter).
Key Point 3: Some active transport processes are powered by light energy (e.g., bacteriorhodopsin in Halobacterium).
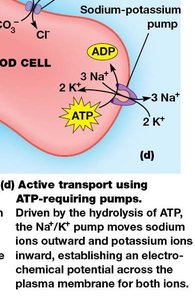
Examples of Active Transport
Na+/K+ Pump: A P-type ATPase that moves 3 Na+ ions out and 2 K+ ions into animal cells, maintaining electrochemical gradients.
Na+/Glucose Symporter: Uses the Na+ gradient to drive glucose uptake into cells (secondary active transport).
Bacteriorhodopsin: In Halobacterium, light energy drives proton pumping, which is used to synthesize ATP.
The Energetics of Transport
Free Energy Changes in Transport
The spontaneity of transport processes is determined by the change in free energy (ΔG). For uncharged solutes, ΔG depends only on the concentration gradient. For charged solutes, both the concentration gradient and the membrane potential must be considered.
Key Point 1: If ΔG < 0, transport is spontaneous; if ΔG > 0, energy input is required; if ΔG = 0, there is no net movement.
Key Point 2: For uncharged solutes, the equilibrium constant (Keq) is 1 and ΔGo is 0.
Equations:
For uncharged solutes:
For charged solutes:
Where R is the gas constant (1.987 cal/mol-K), T is the absolute temperature, [S] is the solute concentration, z is the charge on the solute, F is the Faraday constant (23,062 cal/mol-V), and Vm is the membrane potential.
Summary Table: Comparison of Transport Mechanisms
Property | Simple Diffusion | Facilitated Diffusion | Active Transport |
|---|---|---|---|
Energy Requirement | No | No | Yes |
Direction | Down gradient | Down gradient | Against gradient |
Transport Protein | No | Yes | Yes |
Saturation | No | Yes | Yes |
Examples | O2, CO2 | GLUT1, aquaporins | Na+/K+ pump |
Additional info: The above notes integrate textbook-level explanations, relevant equations, and examples from erythrocyte transport to provide a comprehensive overview of membrane transport processes in cell biology.
