 Back
BackVesicular Traffic, Secretion, and Endocytosis: Structure and Function in Cell Biology
Study Guide - Smart Notes

Vesicular Traffic, Secretion, and Endocytosis
Basic Principles of Vesicle Trafficking
Vesicle trafficking is a fundamental process in eukaryotic cells, governing the transport of proteins and lipids between cellular compartments. This process is essential for maintaining the composition of the plasma membrane and for the secretion and uptake of molecules.
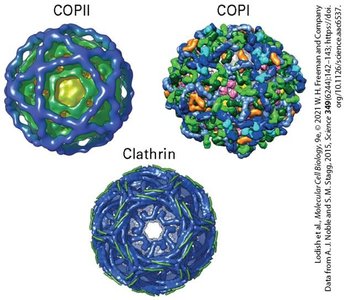
Vesicle Types: COPII, COPI, and Clathrin-coated vesicles are the main types involved in protein trafficking.
Directionality: COPII vesicles mediate anterograde transport (ER to Golgi), COPI vesicles mediate retrograde transport (Golgi to ER), and Clathrin vesicles are involved in endocytosis and transport from the Golgi to endosomes.
Coat Proteins: These proteins help shape the vesicle and select cargo for transport.
Overview of Secretory and Endocytic Pathways
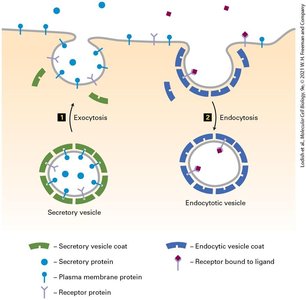
The secretory pathway involves the movement of proteins from the ER through the Golgi apparatus to their final destinations, such as the plasma membrane or extracellular space. The endocytic pathway involves the internalization of molecules from the plasma membrane into the cell.
Exocytosis: The process by which secretory vesicles fuse with the plasma membrane to release their contents.
Endocytosis: The process by which cells internalize molecules and particles from the extracellular environment.
Key Players: Secretory vesicle coats, endocytic vesicle coats, receptor proteins, and cargo proteins.
Intracellular Transport Pathways
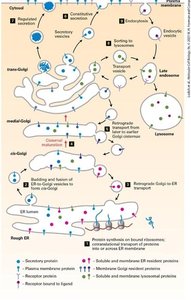
Proteins synthesized in the rough ER are transported to the Golgi apparatus for further processing and sorting. The Golgi is organized into cis, medial, and trans compartments, each with distinct functions.
Protein Sorting: Proteins are sorted in the Golgi for delivery to lysosomes, the plasma membrane, or for secretion.
Retrograde Transport: COPI vesicles return proteins to the ER or earlier Golgi compartments.
Endosomes and Lysosomes: Endocytic vesicles deliver cargo to endosomes, which mature into lysosomes for degradation.
The Life Cycle of a Transport Vesicle
Vesicle Formation and Fusion
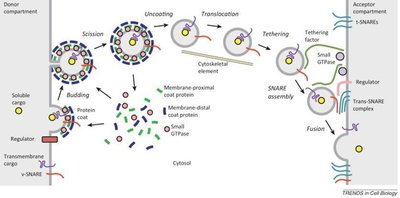
Transport vesicles are formed by budding from donor membranes, coated with specific proteins, and then uncoated before fusion with target membranes. The process involves several steps:
Budding: Coat proteins assemble on the donor membrane, causing it to curve and bud off.
Scission: The vesicle is pinched off from the donor membrane.
Uncoating: Coat proteins are removed, allowing the vesicle to interact with target membranes.
Translocation: Vesicles are transported along cytoskeletal elements.
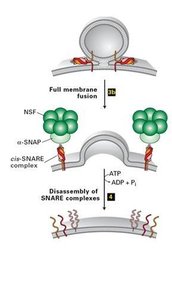
Tethering and Fusion: Vesicles are tethered to the target membrane, and SNARE proteins mediate membrane fusion.
Coated Vesicle Budding
Coated vesicle budding is initiated by the recruitment of coat proteins and GTP-binding proteins to the donor membrane. Cargo proteins and receptors are selected for transport.
GTP-binding Proteins: Regulate coat assembly and vesicle formation.
v-SNARE Proteins: Incorporated into vesicles for later fusion with target membranes.
Membrane Cargo-Receptor Proteins: Ensure specific cargo selection.
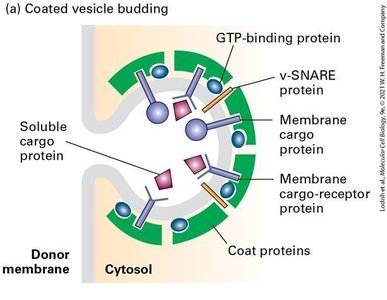
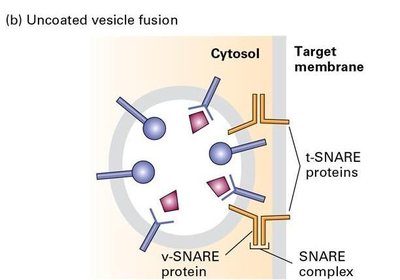
Uncoated Vesicle Fusion
After uncoating, vesicles fuse with their target membranes through the interaction of v-SNARE and t-SNARE proteins, forming a SNARE complex that brings the membranes together.
v-SNARE Proteins: Located on vesicles.
t-SNARE Proteins: Located on target membranes.
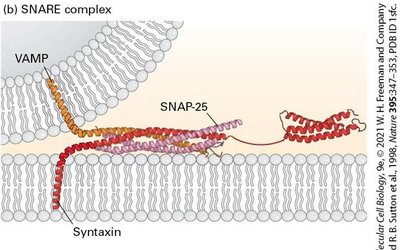
SNARE Complex: Drives membrane fusion.
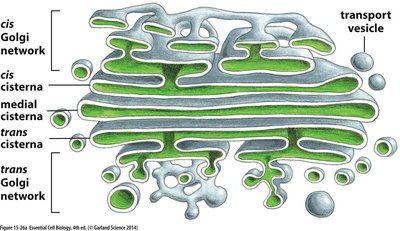
The Golgi Complex: Structure and Function
Golgi Apparatus Structure
The Golgi complex is a stack of flattened cisternae surrounded by vesicles. It is divided into distinct functional compartments: cis, medial, and trans regions, each with specific roles in protein processing and sorting.
Cis Face: Entry face closest to the ER; contains the cis Golgi network.
Medial Cisternae: Central region for further processing.
Trans Face: Exit face closest to the plasma membrane; contains the trans Golgi network.
Golgi Apparatus Functions
The Golgi apparatus is responsible for the modification, sorting, and packaging of proteins and lipids. Key functions include:
Modification of N-linked Oligosaccharides: Addition and processing of carbohydrate groups on proteins.
O-linked Glycosylation: Attachment of glycans to serine or threonine residues.
Phosphorylation and Sulfation: Addition of phosphate and sulfate groups to proteins.
Proteolytic Cleavage: Processing of secretory proteins.
Sphingolipid Synthesis: Production of complex lipids.
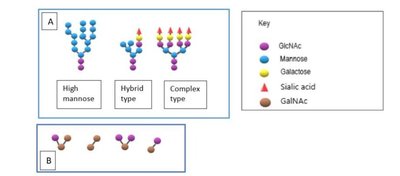
Glycosylation of Proteins
Glycosylation is a post-translational modification where carbohydrate groups are added to proteins, affecting their stability, activity, and localization.
N-linked Glycosylation: Occurs on the amide nitrogen of asparagine residues.
O-linked Glycosylation: Occurs on the hydroxyl group of serine or threonine residues.
Types of Glycans: High mannose, hybrid, and complex types for N-linked; various core structures for O-linked.
Coated Vesicles and Protein Trafficking
Types of Coated Vesicles
Coated vesicles are classified based on their coat proteins and the transport steps they mediate. The main types are COPII, COPI, and Clathrin-coated vesicles.
Vesicle Type | Transport Step Mediated | Coat Proteins | Associated GTPase |
|---|---|---|---|
COPII | ER to cis-Golgi | Sec23/Sec24, Sec13/Sec31, Sec16 | Sar1 |
COPI | cis-Golgi to ER, later to earlier Golgi cisternae | Coatomers (seven COP subunits) | ARF |
Clathrin | trans-Golgi to endosome, plasma membrane to endosome, Golgi to lysosome | Clathrin + AP complexes | ARF |
Assembly and Disassembly of COPII Coat
The assembly and disassembly of COPII-coated vesicles is regulated by the GTPase Sar1. The process involves:
Activation: Sar1-GDP interacts with Sec12, exchanging GDP for GTP.
Coat Assembly: Sar1-GTP recruits Sec23/Sec24 and Sec13/Sec31 complexes.
GTP Hydrolysis: Sec23 stimulates Sar1 GTP hydrolysis, leading to coat disassembly.
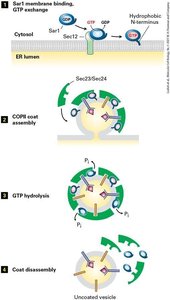
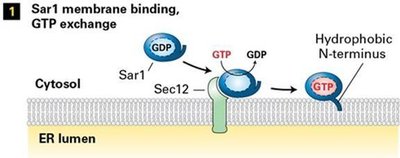
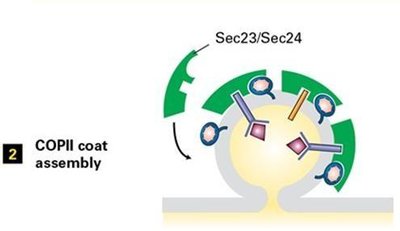
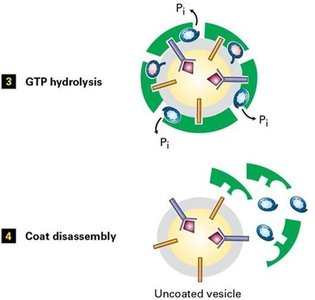
Vesicle Docking and Fusion
Rab GTPases and SNARE Proteins
Rab GTPases and SNARE proteins are essential for the docking and fusion of transport vesicles with their target membranes.
Rab GTPases: Control vesicle docking by binding to effector proteins on target membranes.
v-SNARE and t-SNARE Proteins: Form stable coiled-coil interactions, bringing vesicle and target membranes together.
SNARE Complex: Four α helices (two from SNAP-25, one each from syntaxin and VAMP) form a coiled-coil structure.
Sorting Signals and Protein Targeting
Known Sorting Signals
Sorting signals are short amino acid sequences that direct proteins to specific transport vesicles. These signals are recognized by coat protein complexes and ensure proper protein targeting.
Signal Sequence | Signal-Bearing Protein | Proteins with Signal | Signal Receptor |
|---|---|---|---|
Lys-Lys-X-X (KKXX) | ER-resident membrane proteins | COPI α and β subunits | COPI |
Di-arginine (X-Arg-Arg-X) | ER-resident membrane proteins | COPI α and β subunits | COPI |
Di-acidic (Asp-X-Glu) | Cargo membrane proteins in ER | COPII Sec24 subunit | COPII |
Asn-Pro-X-Tyr (NPXY) | LDL receptor in plasma membrane | AP2 complex | Clathrin/AP2 |
Tyr-X-X-Φ (YXXΦ) | Membrane proteins in trans-Golgi | AP1 (μ1 subunit) | Clathrin/AP1 |
Leu-Leu (LL) | Plasma membrane proteins | AP2 complexes | Clathrin/AP2 |
Lys-Asp-Glu-Leu (KDEL) | ER-resident soluble proteins | KDEL receptor in cis-Golgi membrane | COPI |
Mannose 6-phosphate (M6P) | Soluble lysosomal enzymes | M6P receptor in trans-Golgi membrane | Clathrin/AP1 |
Summary
Vesicular traffic, secretion, and endocytosis are central to cell biology, enabling the transport, processing, and sorting of proteins and lipids. The coordinated action of coat proteins, GTPases, SNAREs, and sorting signals ensures the fidelity and directionality of intracellular transport.
Key Equations:
GTP Hydrolysis:
SNARE Complex Formation:
Additional info: The notes include expanded explanations and context for clarity and completeness, suitable for exam preparation in a cell biology course.
