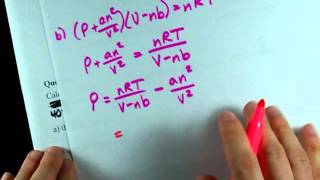7. Gases
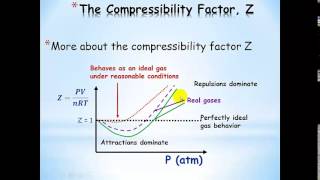
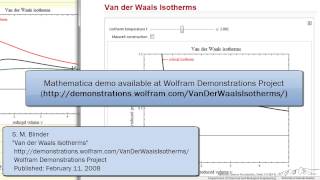
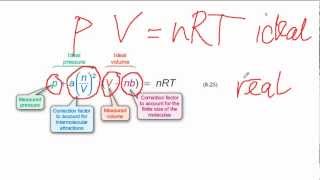
Van der Waals Equation

Get help from an AI Tutor
Ask a question to get started.
Problem 93
Textbook Question
Textbook QuestionUse the van der Waals equation and the ideal gas equation to calculate the volume of 1.000 mol of neon at a pressure of 500.0 atm and a temperature of 355.0 K. Explain why the two values are different. (Hint: One way to solve the van der Waals equation for V is to use successive approximations. Use the ideal gas law to get a preliminary estimate for V.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
3052
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos