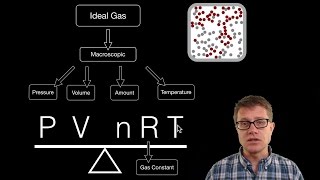
7. Gases
The Ideal Gas Law

Get help from an AI Tutor
Ask a question to get started.
Problem 120
Textbook Question
Textbook QuestionThe radius of a xenon atom is 1.3 * 10 - 8 cm. A 100-mL flask is filled with Xe at a pressure of 1.0 atm and a temperature of 273 K. Calculate the fraction of the volume that is occupied by Xe atoms. (Hint: The atoms are spheres.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
1941
views
2
comments
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos