16. Chemical Equilibrium
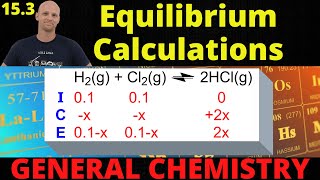
ICE Charts

Get help from an AI Tutor
Ask a question to get started.
Problem 154b
Textbook Question
Textbook QuestionAcetic acid tends to form dimers, 1CH3CO2H22, because of hydrogen bonding: The equilibrium constant Kc for this reaction is 1.51 * 102 in benzene solution but only 3.7 * 10-2 in water solution. (b) Calculate the ratio of dimers to monomers for 0.100 M acetic acid in water.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
571
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos