 Back
BackAnalyzing Reaction Mechanisms Using Energy Diagrams
Study Guide - Smart Notes

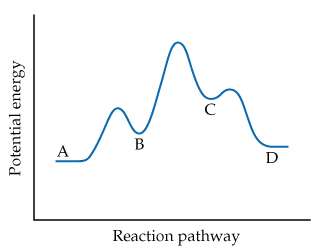
Q16. How many steps are in the reaction based on the energy diagram shown?
Background
Topic: Reaction Mechanisms & Energy Diagrams
This question tests your ability to interpret an energy diagram and determine the number of steps (elementary reactions) in a reaction mechanism. Each "step" corresponds to a distinct peak (activation energy barrier) in the diagram.
Key Terms and Concepts:
Reaction pathway: The progress of the reaction from reactants to products.
Potential energy: The energy of the system at each stage of the reaction.
Activation energy: The energy required to reach the transition state for each step.
Elementary step: A single stage in the reaction mechanism, often associated with a peak in the energy diagram.
Step-by-Step Guidance
Examine the energy diagram and identify the number of distinct peaks (high points) along the reaction pathway. Each peak represents an activation energy barrier for an elementary step.
Count the valleys (low points) between the peaks. These valleys often correspond to intermediates formed during the reaction.
Recall that the number of steps in the mechanism is equal to the number of activation energy barriers (peaks) in the diagram.
Compare the labeled points (A, B, C, D) to see which represent reactants, intermediates, transition states, and products.
Try solving on your own before revealing the answer!
