 Back
BackAcids, Bases, and Equilibrium: Comprehensive Study Notes
Study Guide - Smart Notes

Acids and Bases: Introduction and Clinical Relevance
Clinical Laboratory Applications
Acids and bases play a crucial role in clinical laboratory settings, where technicians analyze specimens and interpret test results for medical diagnoses, such as cancer detection and blood typing. Understanding acid-base chemistry is essential for accurate analysis and interpretation.

Key Math and Chemistry Skills for Acid-Base Chemistry
Solving Equations: Essential for quantitative analysis.
Scientific Notation: Used for expressing concentrations.
Writing Ionic Formulas: Important for representing acids and bases.
Balancing Chemical Equations: Fundamental for reaction stoichiometry.
Mole–Mole Factors: Used in titration and reaction calculations.
Concentration Conversions: Key for solution chemistry.
Acids and Bases: Definitions and Properties
Acids: Characteristics and Naming
Acids are molecular substances that produce ions in water, including the hydronium ion (H3O+). They are classified as electrolytes and are known for their sour taste, ability to turn blue litmus red, and their corrosive properties.

Arrhenius Acids: Produce H+ ions in water.
Brønsted–Lowry Acids: Donate H+ ions.
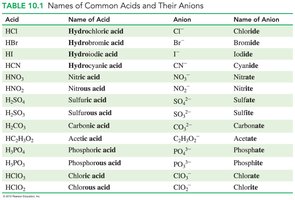
Naming Acids:
Acids with H+ and a nonmetal: Prefix "hydro" and suffix "ic acid" (e.g., HCl: hydrochloric acid).
Acids with H+ and a polyatomic ion: "ate" becomes "ic acid"; "ite" becomes "ous acid" (e.g., HNO3: nitric acid).
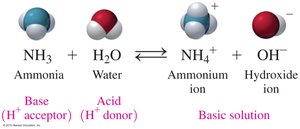
Bases: Characteristics and Naming
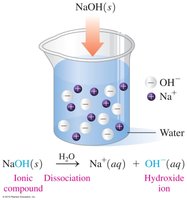
Bases are substances that produce hydroxide ions (OH−) in water. They are electrolytes, taste bitter or chalky, feel slippery, and turn litmus paper blue.
Arrhenius Bases: Produce OH− ions in water.
Brønsted–Lowry Bases: Accept H+ ions.
Naming Bases: Named as hydroxides (e.g., NaOH: sodium hydroxide).
Acid-Base Theories
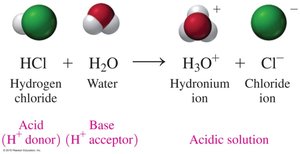
Brønsted–Lowry Theory
According to the Brønsted–Lowry theory, acids donate H+ ions, while bases accept H+ ions. This theory expands the definition of acids and bases beyond aqueous solutions.
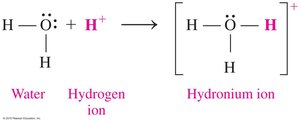
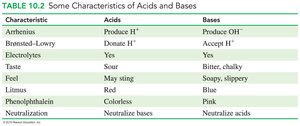
Characteristics of Acids and Bases
Acids and bases differ in their properties, including taste, feel, and reactions with indicators.
Characteristic | Acids | Bases |
|---|---|---|
Arrhenius | Produce H+ | Produce OH− |
Brønsted–Lowry | Donate H+ | Accept H+ |
Electrolytes | Yes | Yes |
Taste | Sour | Bitter, chalky |
Feel | May sting | Soapy, slippery |
Litmus | Red | Blue |
Phenolphthalein | Colorless | Pink |
Neutralization | Neutralize bases | Neutralize acids |
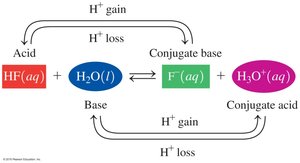
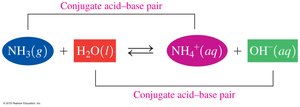
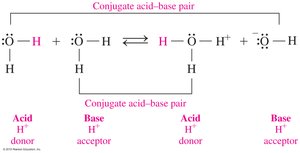
Conjugate Acid–Base Pairs
Definition and Identification
In acid–base reactions, conjugate pairs are formed by the loss or gain of H+. Each acid has a conjugate base, and each base has a conjugate acid.
Conjugate Acid–Base Pair Example: HF and F−; H2O and H3O+

Strengths of Acids and Bases
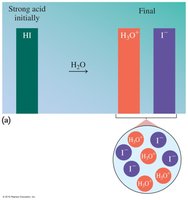
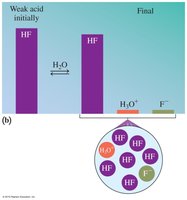
Strong vs. Weak Acids
Strong acids completely ionize in water, producing high concentrations of H3O+. Weak acids only partially dissociate, resulting in lower ion concentrations.
Strong Acids: HCl, HBr, HI, HNO3, H2SO4, HClO4
Weak Acids: HF, H2CO3, CH3COOH




Strong vs. Weak Bases
Strong bases, such as NaOH and KOH, dissociate completely in water. Weak bases, like NH3 and Cu(OH)2, only partially ionize.

Acid–Base Equilibrium
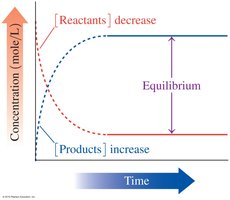
Reversible Reactions and Equilibrium
Acid–base reactions can reach equilibrium, where the rates of the forward and reverse reactions are equal and concentrations remain constant.

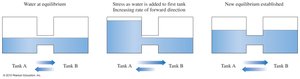
Le Châtelier’s Principle
Le Châtelier’s principle states that a system at equilibrium will adjust to relieve any stress caused by changes in concentration, temperature, or pressure.
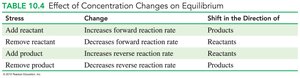
Stress | Change | Shift in Direction of |
|---|---|---|
Add reactant | Increases forward reaction rate | Products |
Remove reactant | Decreases forward reaction rate | Reactants |
Add product | Increases reverse reaction rate | Reactants |
Remove product | Decreases reverse reaction rate | Products |
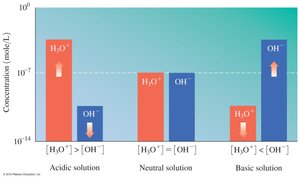
Dissociation of Water and Ion Product Constant (Kw)
Ionization of Water
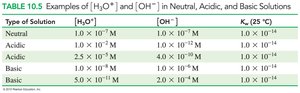
Water is amphoteric and can act as both an acid and a base. The dissociation of water produces equal concentrations of H3O+ and OH− ions.
Ion Product Constant: at 25°C
![Guide to calculating [H3O+] and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/62627a34_image_26.png)
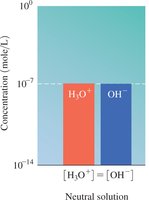
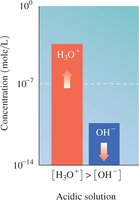
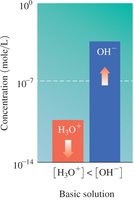
![Comparison of [H3O+] and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/62627a34_image_30.png)
![Guide to calculating [H3O+] and [OH-]](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/62627a34_image_32.png)
The pH Scale
Definition and Calculation
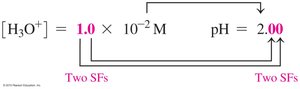
The pH scale measures the acidity or basicity of a solution, ranging from 0 (very acidic) to 14 (very basic). pH is calculated as the negative logarithm of the hydronium ion concentration:
pH Equation:




![Guide to calculating [H3O+] from pH](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/62627a34_image_44.png)
![Comparison of [H3O+], [OH-], and pH](https://static.studychannel.pearsonprd.tech/study_guide_files/gob/sub_images/62627a34_image_45.png)
Reactions of Acids and Bases
Acids Reacting with Metals, Carbonates, and Bases
Acids react with metals to produce hydrogen gas and salts, with carbonates and bicarbonates to produce carbon dioxide, and with bases in neutralization reactions to form water and salts.



Acid–Base Titration
Principles and Calculations
Titration is used to determine the concentration of an acid or base by neutralizing it with a solution of known concentration. The endpoint is identified using an indicator such as phenolphthalein.

Chemistry Link to Health: Antacids
Antacids neutralize excess stomach acid and are composed of aluminum hydroxide and magnesium hydroxide. These compounds are only slightly soluble, minimizing adverse effects on the digestive tract.
Buffers
Definition and Function
A buffer solution maintains pH by neutralizing small amounts of added acid or base. It contains a weak acid and its conjugate base, or a weak base and its conjugate acid.
Acids and Bases—Concept Map
Additional info: These notes cover all major aspects of Chapter 10: Acids and Bases and Equilibrium, including definitions, properties, reactions, calculations, and clinical relevance. All images included are directly relevant to the adjacent content and reinforce key concepts for college-level chemistry students.
