 Back
BackPrenatal Development and Newborn Assessment: Foundations of Developmental Psychology
Study Guide - Smart Notes

Prenatal Development: Stages and Processes
Overview of Prenatal Development
Prenatal development is the process from conception to birth, transforming a single cell into a fully formed human being. It is divided into three biological stages: germinal, embryonic, and fetal periods, each with distinct developmental milestones.
Germinal Period: Conception to ~2 weeks; cell division and implantation begin.
Embryonic Period: ~3–8 weeks; major organ systems and body structures form.
Fetal Period: ~9 weeks to birth; growth, refinement, and functional maturation.
Stage | Timeframe | Key Feature |
|---|---|---|
Germinal | Conception–2 weeks | Cell division, implantation |
Embryonic | 3–8 weeks | Organogenesis, vulnerability to teratogens |
Fetal | 9 weeks–birth | Growth, maturation, viability |
Germinal Period
The germinal period begins at fertilization, when sperm and egg unite to form a zygote. Rapid mitotic divisions (cleavage) increase cell number, leading to the blastocyst stage. Implantation occurs as the blastocyst embeds in the uterine wall.
Zygote: The single-cell formed at fertilization.
Blastocyst: Hollow ball of ~100 cells ready for implantation.
Implantation: Essential for pregnancy to continue; about half of zygotes do not survive this stage due to genetic abnormalities.
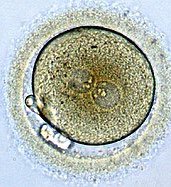
Embryonic Period
The embryonic period starts around week 3 post-fertilization. The blastocyst becomes an embryo, and organogenesis (formation of organs) occurs. The placenta and supporting structures develop, and this is the most vulnerable stage for teratogen effects.
Organogenesis: Formation of major organs and body structures.
Placenta: Develops to support nutrient and waste exchange.
Teratogen vulnerability: Exposure to harmful agents can cause structural defects.
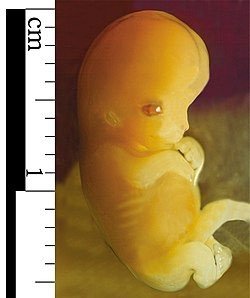
Fetal Period
The fetal period begins at week 9 and lasts until birth. This stage is characterized by rapid growth, maturation of body systems, and increasing viability (survival outside the womb becomes possible around 24 weeks).
Growth: Size and weight increase dramatically.
Viability: Survival possible with medical support from ~24 weeks.
Functional maturation: Organs and systems prepare for independent life.
Brain Development Before Birth
Foundations of Brain Development
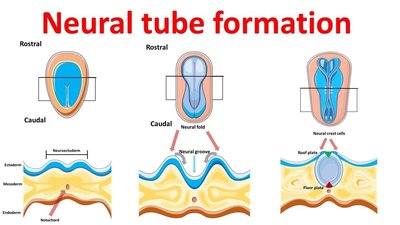
Prenatal brain development begins early in the embryonic period with the formation of the neural tube, which differentiates into the brain and spinal cord. Rapid neurogenesis and cell migration establish major brain structures and basic neural organization.
Neural tube: Forms around weeks 3–4; failure to close leads to neural tube defects.
Neurogenesis: Production of neurons; most occurs prenatally.
Cell migration: Neurons move to specific brain regions; errors can affect cognition and behavior.

Synaptogenesis and Early Connectivity
Formation of synapses begins prenatally, with overproduction of connections. Early circuits are broadly tuned and inefficient; experience after birth shapes the strengthening of pathways.
Synaptogenesis: Creation of synaptic connections between neurons.
Experience-dependent: Postnatal experiences refine neural circuits.
Brain Maturation Before Birth
Subcortical systems mature earlier than the cortex. Cortical development accelerates in late gestation, and myelination (insulation of nerve fibers) begins prenatally. Brain organization continues long after birth.
Subcortical maturation: Early development of basic regulatory systems.
Cortical development: Accelerates in third trimester.
Myelination: Begins before birth, continues postnatally.
Teratogens and Environmental Risks
Definition and Key Factors
Teratogens are environmental agents that can cause prenatal harm. Effects depend on timing, dose, genetics, and number of exposures. The embryonic period is the highest-risk window for structural abnormalities.
Timing: Most severe effects during organ formation (weeks 3–8).
Dose and duration: Higher levels or longer exposure increase risk.
Genetics: Maternal and fetal genotype influence susceptibility.
Number of teratogens: Multiple exposures add risk.
Common Teratogens and Effects
Alcohol: Causes Fetal Alcohol Spectrum Disorders (FASD), including intellectual disability and characteristic facial/CNS effects.
Tobacco/Nicotine: Reduces oxygen, linked to low birth weight, preterm birth, and developmental issues.
Medications: Some drugs (e.g., isotretinoin, anticonvulsants) are known teratogens; others require careful risk–benefit analysis.
Illicit substances: Cocaine, heroin, methamphetamine increase risks of low birth weight, preterm birth, and neonatal withdrawal syndromes.
Heavy metals: Lead, mercury cause neurotoxicity.
Infections: Rubella, cytomegalovirus, toxoplasmosis, syphilis, Zika virus can cause structural and neurological defects.
Critical Periods for Teratogenic Risks
Embryonic period (weeks 3–8): Highest risk for structural defects.
Fetal period: Functional and growth effects more likely.
Prevention: Avoid alcohol, tobacco, and known harmful drugs; ensure vaccination and proper nutrition.
Maternal and Paternal Factors in Prenatal Development
Maternal Age and Health
Maternal age, health, lifestyle, and environment play major roles in prenatal outcomes. Advanced maternal age (35+) increases risks for chromosomal conditions and pregnancy complications, while teen pregnancy is linked to higher risk of preterm birth and low birth weight.
Advanced maternal age: Higher risk of Down syndrome, gestational diabetes, preterm birth.
Teen pregnancy: Increased risk due to social factors (nutrition, access to care).
Chronic illnesses: Gestational diabetes, hypertension, pre-existing conditions require careful management.
Nutrition: Adequate intake of folate, iron, iodine is crucial; deficiencies can cause neural tube defects, anemia, developmental delays.
Paternal Factors
Paternal age: Linked to higher rates of new genetic mutations and small increased risk for conditions such as autism and schizophrenia.
Epigenetic effects: Sperm health and lifestyle can influence conception and fetal development.
Prenatal Assessment and Newborn Screening
Prenatal Assessment
Prenatal assessment involves medical evaluations to monitor the health of both the pregnant person and the developing fetus. Routine components include medical checkups, blood and urine analyses, education, and counseling.
Ultrasound imaging: Monitors fetal growth and detects anomalies.
Genetic screening: Blood tests for chromosomal anomalies, anemia, metabolic concerns.
Diagnostic testing: Chorionic villus sampling (CVS), amniocentesis for genetic defects.
Newborn Assessment
Newborn assessment evaluates how well the infant is adapting to life outside the womb and identifies those needing immediate support or follow-up care.
APGAR score: Rapid screening of vital functioning at 1 and 5 minutes (heart rate, respiration, reflexes, muscle tone, color).
Neonatal Behavioral Assessment Scale (NBAS): Examines reflexes, motor control, responsiveness, and self-regulation.
Newborn Genetic Screening
Newborn genetic screening involves routine blood tests to detect inherited metabolic and genetic disorders before symptoms appear. Early detection allows timely intervention, changing developmental trajectories.
Phenylketonuria (PKU): Early treatment prevents brain damage and developmental delay.
Spinal Muscular Atrophy (SMA): Early identification allows presymptomatic treatments, improving motor outcomes.
Common Birth Complications and Outcomes
Birth Complications
Preterm birth: Before 37 weeks; associated with immature organ systems and increased risk for cognitive, motor, and attention difficulties.
Low birth weight: Under 2,500 g; linked to higher rates of neonatal complications and long-term risks for learning and health problems.
Birth asphyxia: Oxygen deprivation can affect the central nervous system and lead to motor impairments or neurological injury.
Breech delivery: Feet or buttocks down; increases risk for umbilical cord prolapse and difficult delivery.
Meconium aspiration: Inhalation of meconium-stained fluid can cause respiratory distress.
Neonatal infection or distress: Early physiological instability can influence feeding, regulation, and early caregiver–infant interaction.
Prematurity, Hypoxia, and Cerebral Palsy
Hypoxia: Insufficient oxygen to the brain can disrupt normal development and result in neurological injury.
Cerebral palsy (CP): Non-progressive motor disorders caused by early brain injury, affecting movement, posture, and coordination.
Premature infants: Higher risk for CP due to vulnerable developing brains.
Neural Tube Defects and Prevention
Neural Tube Defects (NTDs)
Neural tube defects are serious congenital abnormalities that occur when the neural tube fails to close properly during early embryonic development (weeks 3–4). Common NTDs include spina bifida and anencephaly.
Folate deficiency: Major, preventable risk factor; adequate folic acid intake before and early in pregnancy reduces NTD risk.
Developmental takeaway: NTDs highlight the importance of critical periods and early prenatal nutrition in brain and spinal development.

COVID-19 and Prenatal Development
COVID-19 Effects
Maternal COVID-19 infection has been linked to greater risks of adverse outcomes (e.g., preterm birth, low birth weight) and potential effects on fetal health. Psychological stress during the pandemic may influence fetal brain development and later child behavior.
Vaccination: Protects both pregnant person and fetus; maternal antibodies provide passive immunity to the newborn.
Research: No evidence of increased risk for miscarriage, congenital anomalies, or adverse developmental outcomes from recommended vaccines.
Newborn Reflexes and Evolutionary Significance
Newborn Reflexes
Newborn reflexes (e.g., grasp, rooting, sucking, Moro) are automatic, survival-oriented responses present at birth that support feeding, protection, and early interaction. Many reflexes are thought to have evolutionary benefits.
Grasp reflex: May reflect ancestral adaptation for clinging to caregivers.
Rooting and sucking: Support feeding and survival.
Developmental perspective: Reflexes provide insight into early neural functioning and fade as voluntary motor control develops.
Birth Contexts and Neonatal Outcomes
Birth Settings
Free birthing: Birth without medical professionals; emphasizes autonomy but carries higher risk due to lack of monitoring and emergency response.
Midwife-led birth: Low-intervention care with trained professionals; positive experiences for low-risk pregnancies.
Hospital birth: Immediate access to medical technology and specialists; reduces risk in high-risk situations.
Doula: Trained non-medical support person providing emotional, physical, and informational support during pregnancy, labor, and postpartum.
Key Takeaways
Prenatal development is structured in three biological stages.
Timing of environmental exposures critically shapes outcomes.
Both maternal and paternal factors influence development.
Prenatal assessment and newborn screening help optimize healthy outcomes.
Events during pregnancy and the neonatal period can have lifelong impact on development.
