 Back
BackAcid-Base Balance and Buffering in Human Physiology
Study Guide - Smart Notes

Q10. How does the equation below shift when the plasma CO2 concentration rises?
Background
Topic: Acid-Base Balance and Buffering
This question tests your understanding of the carbonic acid-bicarbonate buffer system and how changes in CO2 levels affect acid-base equilibrium in the body.
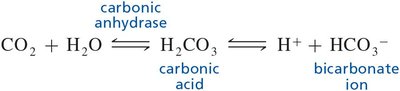
Key Terms and Formulas
CO2: Carbon dioxide, a waste product of cellular respiration.
H2O: Water.
H2CO3: Carbonic acid, formed from CO2 and water.
H+: Hydrogen ion, contributes to acidity.
HCO3-: Bicarbonate ion, acts as a buffer.
Carbonic anhydrase: Enzyme that catalyzes the conversion between CO2 and H2CO3.
Key formula:
Step-by-Step Guidance
Recognize that an increase in plasma CO2 means more substrate is available for the reaction.
Recall Le Chatelier's Principle: When the concentration of a reactant increases, the equilibrium shifts to consume that reactant.
Apply this principle to the equation: More CO2 will drive the reaction toward the formation of H2CO3 and subsequently toward H+ and HCO3-.
Consider the physiological effect: Increased H+ production can lower blood pH, leading to acidosis.
Try solving on your own before revealing the answer!
Final Answer: Shifts to the right
When plasma CO2 concentration rises, the equilibrium shifts to the right, producing more H2CO3, H+, and HCO3-. This increases acidity and can contribute to respiratory acidosis.
