 Back
BackCell Metabolism and Glycolysis: Energy, Enzymes, and ATP
Study Guide - Smart Notes

Cell Metabolism - Glycolysis
Learning Objectives
This section introduces the foundational concepts of cell metabolism, focusing on glycolysis, energy transformations, and the role of enzymes and ATP in cellular processes. Understanding these topics is essential for grasping how cells generate and utilize energy.
Energy Types: Differentiate between kinetic and potential energy, and understand their roles in biological systems.
Enzymes: Comprehend how enzymes facilitate reactions by lowering activation energy barriers.
ATP: Learn the structure and function of ATP, and why its hydrolysis is vital for life.
Phosphocreatine: Understand the role of phosphocreatine in rapid ATP regeneration.
Glycolysis: Know the steps, substrates, and products of glycolysis, and its integration with other metabolic pathways.
Citric Acid Cycle: Recognize the link between glycolysis and the citric acid cycle, and the fate of glucose carbons.
Law of Mass Action: Apply this principle to physiological reactions, such as oxygen and carbon dioxide exchange.

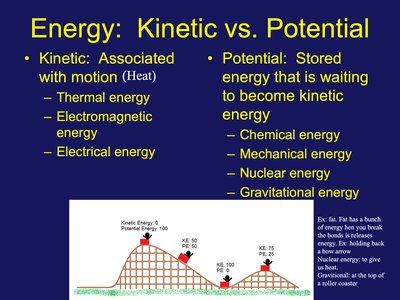
Energy: Kinetic vs. Potential
Definitions and Examples
Energy exists in two primary forms: kinetic and potential. Kinetic energy is associated with motion, while potential energy is stored and can be converted to kinetic energy.
Kinetic Energy: Energy of movement (e.g., thermal, electromagnetic, electrical).
Potential Energy: Stored energy (e.g., chemical, mechanical, nuclear, gravitational).
Example: Fat stores chemical energy; a roller coaster at the top has gravitational potential energy.
Laws of Thermodynamics
Energy Equilibration and Entropy
The laws of thermodynamics govern energy transformations in biological systems. In a closed system, energy equilibrates and entropy increases, leading to a state where no net energy is available for work.
First Law: Energy cannot be created or destroyed, only transformed.
Second Law: Entropy (disorder) increases in closed systems.
Biological Implication: Cells must constantly input energy to maintain order and perform work.

Diffusion and Entropy
Why Substances Spread Out
Diffusion is driven by the tendency of systems to move toward higher entropy. Molecules spread from areas of high concentration to low concentration, seeking equilibrium.
Entropy: A measure of disorder; systems naturally progress toward higher entropy.
Example: Dye diffusing in water illustrates entropy-driven mixing.

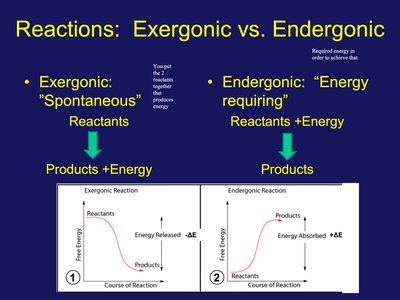
Reactions: Exergonic vs. Endergonic
Energy Changes in Chemical Reactions
Chemical reactions are classified based on their energy profiles. Exergonic reactions release energy and are spontaneous, while endergonic reactions require energy input.
Exergonic: Reactants produce energy; spontaneous ().
Endergonic: Energy is absorbed; not spontaneous ().
Equation:
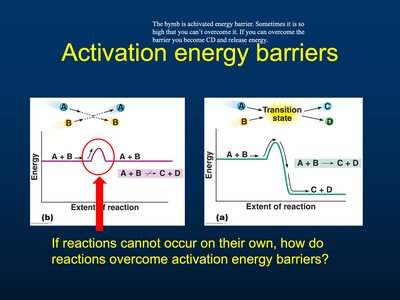
Activation Energy Barriers
Transition States and Reaction Rates
Activation energy is the minimum energy required to initiate a chemical reaction. The transition state is a high-energy intermediate that must be overcome for products to form.
Activation Energy: Energy barrier separating reactants and products.
Transition State: Unstable, high-energy state during a reaction.
Overcoming Barriers: Cells use enzymes to lower activation energy.
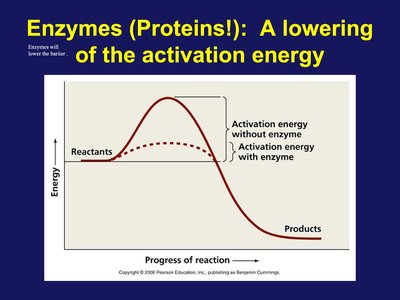
Enzymes: Lowering Activation Energy
Role of Enzymes in Metabolism
Enzymes are biological catalysts that accelerate reactions by lowering the activation energy required. This enables essential cellular processes to occur rapidly and efficiently.
Enzyme Function: Lower activation energy, increase reaction rate.
Example: Digestive enzymes break down food molecules.
Law of Mass Action
Reaction Directionality and Equilibrium
The law of mass action states that the rate of a chemical reaction is proportional to the concentrations of the reactants. By altering reactant or product concentrations, reactions can be pushed forward or backward.
Equation:
Application: Used to explain physiological processes like oxygen and carbon dioxide exchange.




Law of Mass Action in Physiology
Examples: Oxygen and Carbon Dioxide Exchange
Physiological reactions, such as oxygen binding to hemoglobin and carbon dioxide exchange with bicarbonate, are governed by the law of mass action. These reactions are essential for respiratory function.
Oxygen Exchange:
CO2 Exchange:

ATP: The Cellular Energy Currency
ATP Hydrolysis and Energy Release
ATP (adenosine triphosphate) is the primary energy carrier in cells. Hydrolysis of ATP releases energy used for cellular work.
ATP Hydrolysis:
Energy Use: Powers movement, synthesis, and transport.
Fact: Daily ATP turnover equals body weight.

Cellular Activities Requiring ATP
ATP Utilization in Cells
ATP is rapidly consumed during cellular activities such as movement, molecular synthesis, and membrane transport. Cells must continually regenerate ATP to sustain these processes.
Glucose Oxidation:
ATP Use: Fast and essential for life.
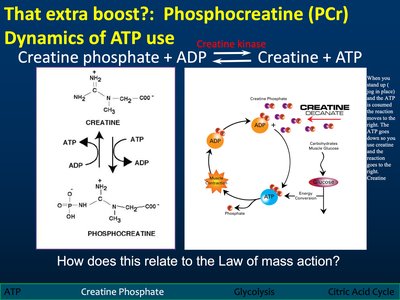
Phosphocreatine (PCr): Dynamics of ATP Use
Creatine Phosphate System
Phosphocreatine acts as a rapid reserve for ATP regeneration during intense cellular activity. The creatine kinase reaction quickly replenishes ATP from ADP.
Reaction:
Law of Mass Action: The reaction direction depends on concentrations of reactants and products.
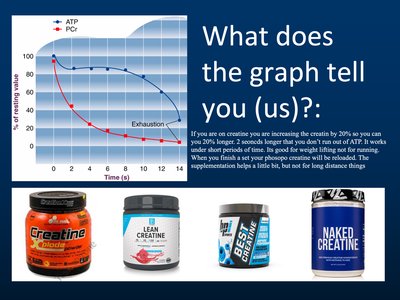
Phosphocreatine Supplementation
Graph Interpretation and Applications
Supplementing creatine increases phosphocreatine stores, allowing for prolonged ATP production during high-intensity activity. The graph shows ATP and PCr levels over time, highlighting the benefit of supplementation.
Application: Useful for short bursts of activity; limited effect for endurance.
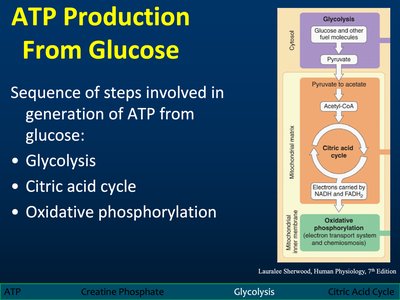
ATP Production from Glucose
Metabolic Pathways
ATP is generated from glucose through three main pathways: glycolysis, the citric acid cycle, and oxidative phosphorylation. Each pathway contributes to the overall energy yield.
Glycolysis: Breakdown of glucose to pyruvate.
Citric Acid Cycle: Oxidation of acetyl-CoA to CO2 and NADH/FADH2.
Oxidative Phosphorylation: Electron transport and ATP synthesis.
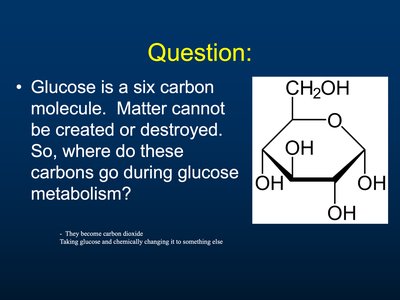
Fate of Glucose Carbons
Carbon Tracking in Metabolism
Glucose is a six-carbon molecule. During metabolism, these carbons are ultimately converted to carbon dioxide, which is expelled from the body.
Conservation of Matter: Matter is not created or destroyed; carbons are transformed.
Example: Glucose carbons become CO2 during cellular respiration.
