 Back
BackChapter 2: Biochemistry – Foundations for Anatomy & Physiology
Study Guide - Smart Notes

Biochemistry: The Chemical Basis of Life
Introduction to Biochemistry
Biochemistry is the study of the chemical processes and substances that occur within living organisms. Understanding biochemistry is essential for comprehending physiological processes, as all body functions depend on chemical reactions and molecular interactions.
Atoms, Ions, and Molecules
Atomic Structure
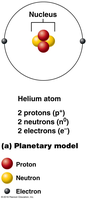
Atoms are the smallest units of matter that retain the properties of an element. Each atom consists of a nucleus containing protons (positively charged) and neutrons (neutral), with electrons (negatively charged) orbiting the nucleus.
Protons: Determine the atomic number and identity of the element.
Neutrons: Contribute to atomic mass and can vary, forming isotopes.
Electrons: Occupy energy levels (shells) and are involved in chemical bonding.
Ions and Isotopes
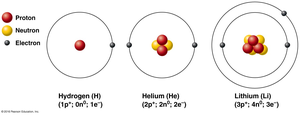
Ions: Atoms that gain or lose electrons become ions. Anions are negatively charged (gained electrons), and cations are positively charged (lost electrons).
Isotopes: Atoms with the same number of protons but different numbers of neutrons.
Chemical Bonds
Ionic Bonds
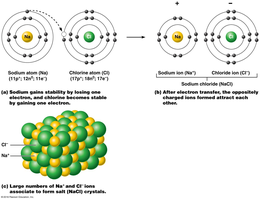
Ionic bonds are formed when electrons are transferred from one atom to another, resulting in the attraction between oppositely charged ions (cations and anions). For example, sodium (Na+) and chloride (Cl–) form sodium chloride (NaCl).
Electrolytes: Salts that dissociate in water to form ions capable of conducting electricity. Electrolyte balance is crucial for nerve and muscle function.
Covalent Bonds
Covalent bonds are formed when atoms share electrons. The number of bonds an atom can form depends on the number of electrons needed to fill its outer shell:
Hydrogen: 1 bond
Oxygen: 2 bonds
Nitrogen: 3 bonds
Carbon: 4 bonds
Polar and Nonpolar Molecules
When electrons are shared unequally, a polar molecule is formed, with partial positive and negative ends. Water is a key example of a polar molecule, making it an excellent solvent for ionic and polar substances.
Hydrogen Bonds
Hydrogen bonds are weak attractions between the positive end of one polar molecule and the negative end of another. They are important in stabilizing the structures of proteins and nucleic acids, and in the properties of water.
Acids, Bases, and pH
Definitions and Importance
Acid: Proton donor; releases H+ ions in water.
Base: Proton acceptor; accepts H+ ions or releases OH– ions in water.
pH: A measure of hydrogen ion concentration, calculated as .
The pH scale is logarithmic; a change of one unit represents a tenfold change in H+ concentration. Blood pH is tightly regulated between 7.35 and 7.45.
Buffers
Buffers are substances that resist changes in pH by absorbing or releasing hydrogen ions. They are essential for maintaining homeostasis in biological systems.
Organic and Inorganic Compounds
Classification
Organic molecules: Contain carbon and hydrogen, usually larger, and include carbohydrates, proteins, lipids, and nucleic acids.
Inorganic molecules: Generally do not contain carbon, are smaller, and include water, oxygen, carbon dioxide, and salts.
Macromolecules of Life
Carbohydrates
Carbohydrates are organic molecules with the general formula (CH2O)n. They serve as energy sources and structural components.
Monosaccharides: Simple sugars (e.g., glucose, fructose, ribose).
Disaccharides: Two monosaccharides joined (e.g., sucrose, lactose).
Polysaccharides: Long chains of monosaccharides (e.g., glycogen, starch).
Monomers are joined by dehydration synthesis and broken down by hydrolysis.
Lipids
Lipids are hydrophobic molecules composed mainly of carbon and hydrogen. They include fats, phospholipids, and steroids.
Fats (Triglycerides): Made of glycerol and three fatty acids; used for energy storage.
Phospholipids: Major components of cell membranes; contain a glycerol, two fatty acids, and a phosphate group.
Steroids: Four-ring structures; cholesterol is a precursor for steroid hormones.
Proteins
Proteins are polymers of amino acids linked by peptide bonds. Their structure determines their function in catalysis, transport, support, movement, and regulation.
Primary structure: Sequence of amino acids.
Secondary structure: Alpha helices and beta sheets formed by hydrogen bonding.
Tertiary structure: Three-dimensional folding driven by interactions among side chains.
Quaternary structure: Association of multiple polypeptide chains.
Enzymes are proteins that catalyze biochemical reactions by lowering activation energy and exhibit specificity for their substrates.
Nucleic Acids
Nucleic acids (DNA and RNA) are polymers of nucleotides, each consisting of a phosphate group, a sugar (ribose or deoxyribose), and a nitrogenous base (adenine, guanine, cytosine, thymine, or uracil).
DNA: Contains deoxyribose and the bases A, T, C, G; stores genetic information.
RNA: Contains ribose and the bases A, U, C, G; involved in protein synthesis.
Adenosine Triphosphate (ATP)
ATP is the primary energy carrier in cells. It stores energy in high-energy phosphate bonds and releases it during hydrolysis to power cellular processes.
ATP hydrolysis:
Phosphorylation: The transfer of a phosphate group to another molecule.
Summary Table: Major Classes of Biomolecules
Class | Monomer | Bond Type | Main Functions |
|---|---|---|---|
Carbohydrates | Monosaccharide | Glycosidic bond | Energy, structure |
Lipids | Fatty acid, glycerol | Ester bond | Energy storage, membranes |
Proteins | Amino acid | Peptide bond | Catalysis, structure, transport |
Nucleic Acids | Nucleotide | Phosphodiester bond | Genetic information, protein synthesis |
