 Back
BackChapter 2: The Chemical Level of Organization – Study Notes
Study Guide - Smart Notes

Chapter 2: The Chemical Level of Organization
Atoms and Atomic Structure
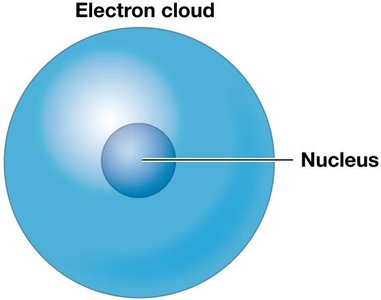
The atom is the smallest stable unit of matter, composed of subatomic particles: protons (positively charged), neutrons (neutral), and electrons (negatively charged). Protons and neutrons are similar in size and mass and are located in the nucleus, while electrons orbit the nucleus in an electron cloud.
Atomic number: The number of protons in an atom, unique for each element.
Atoms are usually electrically neutral, containing equal numbers of protons and electrons (except ions).
Electron cloud: The region around the nucleus where electrons are likely to be found.
Elements and Isotopes
An element is a pure substance consisting of atoms with the same number of protons. Each element has a unique chemical symbol, often derived from English or Latin names (e.g., Na for sodium from 'natrium').
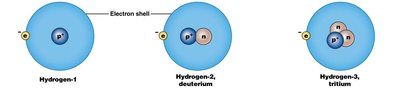
Isotopes: Atoms of the same element with different numbers of neutrons, thus different mass numbers.
Radioisotopes: Unstable isotopes that emit radiation as they decay. The rate of decay is measured as half-life.
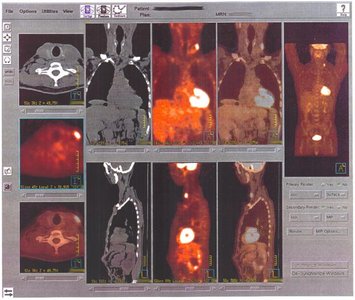
Radioisotopes are used in diagnostic procedures, such as PET scans, to track metabolic activity.
Electrons and Energy Levels
Electrons occupy energy levels or shells around the nucleus. The arrangement of electrons determines the chemical properties of an atom.
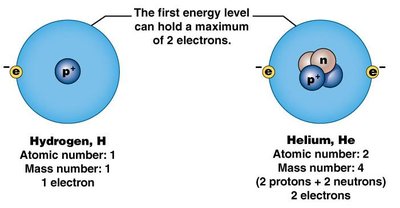
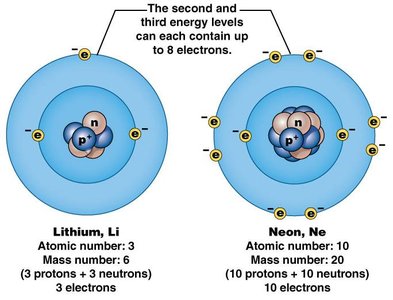
The first shell (closest to the nucleus) can hold up to 2 electrons.
The second and third shells can each hold up to 8 electrons.
The valence shell is the outermost shell; its electron count determines reactivity.
Atoms with unfilled valence shells are reactive; those with filled shells are stable (inert).
Molecules and Compounds
Chemical bonds form when atoms interact to fill their valence shells. These interactions result in molecules and compounds.
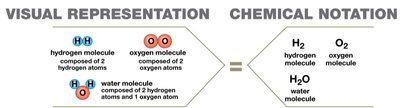
Molecule: Two or more atoms held together by shared electrons (e.g., H2, O2, H2O).
Compound: A chemical substance composed of atoms of two or more different elements (e.g., NaCl, C6H12O6).
Not all molecules are compounds (e.g., H2), and not all compounds are molecules (e.g., NaCl, which is ionic).
Ions and Ionic Bonds
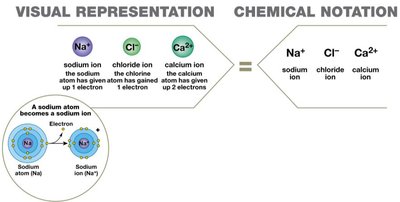
An ion is an atom or group of atoms with an electrical charge, formed by gaining or losing electrons.
Cation: Positively charged ion (loss of electrons).
Anion: Negatively charged ion (gain of electrons).
Ionic bond: Attraction between cations and anions, forming ionic compounds (e.g., NaCl).
Covalent Bonds
Covalent bonds are formed when atoms share electrons to fill their valence shells. These bonds can be single, double, or triple, depending on the number of shared electron pairs.
Nonpolar covalent bond: Equal sharing of electrons (e.g., H2, O2).
Polar covalent bond: Unequal sharing of electrons, resulting in partial charges (e.g., H2O).
Hydrogen Bonds
Hydrogen bonds are weak attractions between the partial positive charge of a hydrogen atom in a polar covalent bond and the partial negative charge of another atom (O, N, or F) in a different polar covalent bond. These are important in water, proteins, and nucleic acids.
Chemical Reactions
Chemical reactions involve the formation or breaking of bonds between atoms. They are represented by chemical equations, with reactants on the left and products on the right.
Decomposition reaction: AB → A + B
Hydrolysis: AB + H2O → AH + BOH
Synthesis reaction: A + B → AB
Dehydration synthesis: AH + BOH → AB + H2O
Exchange reaction: AB + CD → AD + CB
Reversible reaction: A + B \leftrightarrow AB
pH and Homeostasis
pH measures the concentration of hydrogen ions (H+) in a solution. Maintaining proper pH is critical for physiological function.
Neutral: pH = 7 (equal H+ and OH-)
Acidic: pH < 7 (more H+)
Basic (alkaline): pH > 7 (less H+)
Normal blood pH: 7.35–7.45
Acidosis: Blood pH < 7.35
Alkalosis: Blood pH > 7.45
Blood pH is regulated by respiration and kidney function.
Macromolecules
Macromolecules are large, complex molecules essential for life. There are four main types: carbohydrates, lipids, proteins, and nucleic acids.
Monomer: The basic subunit of a macromolecule.
Polymer: A molecule composed of repeating monomers.
Monomers are joined by dehydration synthesis and separated by hydrolysis.
Carbohydrates
Carbohydrates are organic molecules with a 1:2:1 ratio of carbon, hydrogen, and oxygen. They serve as energy sources.
Monosaccharide: Simple sugar (e.g., glucose).
Disaccharide: Two monosaccharides joined (e.g., sucrose).
Polysaccharide: Many monosaccharides joined (e.g., starch, glycogen, cellulose).
Humans can digest starch and glycogen but not cellulose.
Lipids
Lipids are organic molecules with a high ratio of carbon and hydrogen to oxygen. They include fatty acids, glycerides, steroids, and phospholipids.
Fatty acids: Saturated (no double bonds) or unsaturated (one or more double bonds).
Glycerides: Fatty acids attached to glycerol (mono-, di-, triglycerides).
Steroids: Four-ring structure (e.g., cholesterol, hormones).
Phospholipids: Major component of cell membranes.
Proteins
Proteins are polymers of amino acids and are the most abundant macromolecules in the body. They perform structural, regulatory, transport, and catalytic functions.
Amino acids: 20 types, each with a central carbon, hydrogen, amino group, carboxyl group, and R group.
Peptide bond: Covalent bond linking amino acids.
Protein structure: primary (sequence), secondary (alpha helix, beta sheet), tertiary (3D folding), quaternary (multiple polypeptides).
Enzymes: Proteins that catalyze biochemical reactions by lowering activation energy.
Nucleic Acids
Nucleic acids store and process genetic information. The two main types are DNA and RNA.
Nucleotide: Monomer of nucleic acids, consisting of a pentose sugar, phosphate group, and nitrogenous base (A, G, C, T, U).
DNA: Double-stranded, contains A, T, C, G; stores genetic information.
RNA: Single-stranded, contains A, U, C, G; involved in protein synthesis.
High-Energy Compounds
Cells use high-energy compounds, such as ATP, to perform work. These compounds contain high-energy phosphate bonds.
ATP (Adenosine triphosphate): Main energy currency of the cell.
ATP is formed from ADP and a phosphate group, catalyzed by ATPase.
Equation:
