 Back
BackChapter 24: Nutrition, Metabolism, and Energy Balance – Study Notes
Study Guide - Smart Notes

Nutrition, Metabolism, and Energy Balance
Introduction
This chapter explores the essential nutrients required by the human body, the metabolic pathways that convert these nutrients into energy, and the mechanisms that regulate energy balance. Understanding these processes is fundamental for students of anatomy and physiology, as they underpin cellular function, tissue maintenance, and overall health.
Essential Nutrients and Dietary Requirements
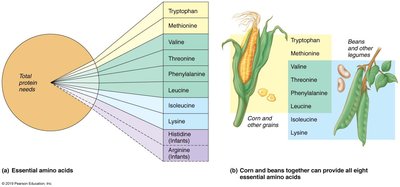
Essential Amino Acids
Essential amino acids are those that the body cannot synthesize in sufficient quantities and must be obtained from the diet. They are crucial for protein synthesis and various metabolic functions.
Definition: Amino acids that must be supplied by the diet because the body cannot produce them rapidly enough.
Examples: Tryptophan, methionine, valine, threonine, phenylalanine, leucine, isoleucine, lysine, histidine (infants), and arginine (infants).
Dietary Sources: Animal proteins (complete proteins), and combinations of plant proteins (e.g., legumes and cereal grains) can provide all essential amino acids for vegetarians.
Protein Requirements
Protein needs depend on age, size, metabolic rate, and nitrogen balance. The general recommendation is 0.8 g of protein per kg of body weight per day.
Calculation Example: For a 175 lb person: kg; g protein/day.
Energy Content: Proteins provide 4 kcal/gram.
Vitamins and Minerals
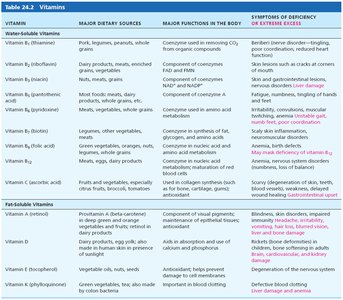
Vitamins
Vitamins are organic compounds required in small amounts for normal metabolism. They are classified based on solubility:
Water-soluble vitamins: B complex and C; absorbed with water, not stored in the body (except B12, which requires intrinsic factor for absorption).
Fat-soluble vitamins: A, D, E, K; absorbed with lipids, stored in the body (except K).
Antioxidants: Vitamins C, E, A, and the mineral selenium neutralize free radicals generated during metabolism.
Health Risks: Excessive intake (megadoses) can cause toxicity, especially with fat-soluble vitamins.
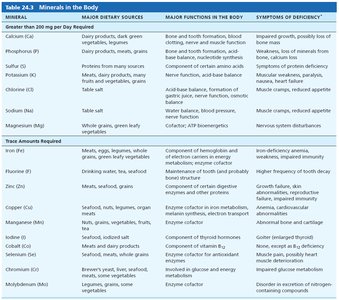
Minerals
Minerals are inorganic elements essential for body structure and function. They are required in varying amounts and play roles in bone formation, nerve function, and metabolic processes.
Major minerals: Calcium, phosphorus, potassium, sulfur, sodium, chloride, magnesium.
Trace elements: Iron, zinc, copper, manganese, iodine, fluoride, selenium, chromium, molybdenum.
Functions: Structural (bones, teeth), regulatory (enzyme cofactors, nerve transmission), and osmotic balance.
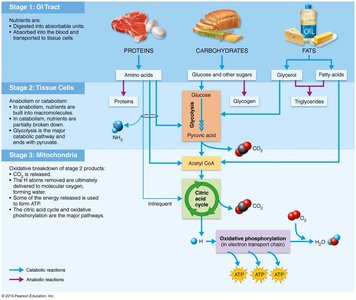
Metabolism: Anabolism and Catabolism
Overview of Metabolic Pathways
Metabolism encompasses all chemical reactions in the body, divided into:
Anabolism: Synthesis of large molecules from smaller ones (e.g., protein synthesis from amino acids).
Catabolism: Breakdown of complex molecules into simpler ones (e.g., protein breakdown into amino acids).
Cellular respiration is a catabolic process that captures energy from food to form ATP, the cell’s energy currency.
Oxidation-Reduction (Redox) Reactions and Coenzymes
Redox reactions are central to energy transfer in metabolism:
Oxidation: Loss of electrons or hydrogen atoms (OIL: Oxidation Is Loss).
Reduction: Gain of electrons or hydrogen atoms (RIG: Reduction Is Gain).
Coenzymes: NAD+ and FAD act as electron (hydrogen) acceptors in oxidative pathways.
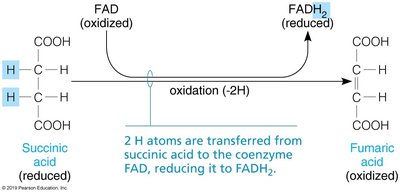
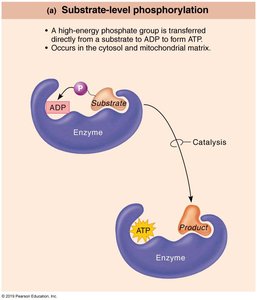
ATP Synthesis Mechanisms
Substrate-level phosphorylation: Direct transfer of a phosphate group to ADP from a phosphorylated substrate. Occurs in glycolysis and the Krebs cycle.
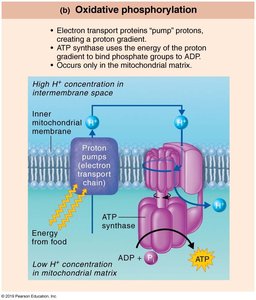
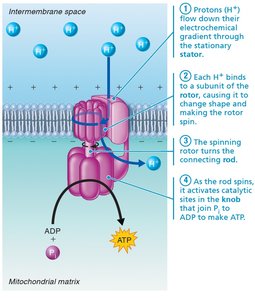
Oxidative phosphorylation: Indirect process using the electron transport chain and chemiosmosis to generate most cellular ATP. Involves the creation of a proton gradient across the mitochondrial membrane and ATP synthase.
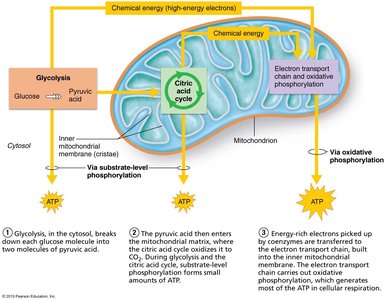
Carbohydrate Metabolism
Glucose Catabolism
Glucose is the primary fuel for ATP production. Its catabolism involves several key pathways:
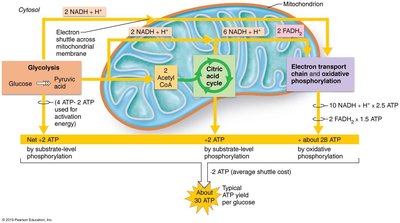
Glycolysis: Anaerobic breakdown of glucose to pyruvic acid in the cytosol.
Transition (Pyruvate Grooming): Conversion of pyruvic acid to acetyl CoA in mitochondria.
Citric Acid Cycle (Krebs Cycle): Aerobic processing of acetyl CoA to produce NADH, FADH2, and CO2.
Electron Transport Chain (ETC) and Oxidative Phosphorylation: Uses electrons from NADH and FADH2 to generate ATP.
The overall equation for glucose oxidation is:
Glycolysis: Three Major Phases
Phase 1: Sugar Activation – Glucose is phosphorylated twice by ATP, forming fructose-1,6-bisphosphate (energy investment phase).
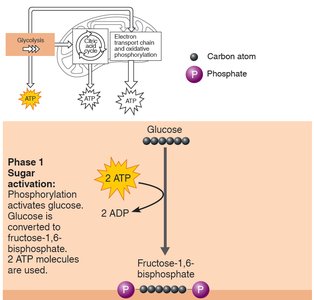
Phase 2: Sugar Cleavage – Fructose-1,6-bisphosphate splits into two 3-carbon fragments (dihydroxyacetone phosphate and glyceraldehyde 3-phosphate).
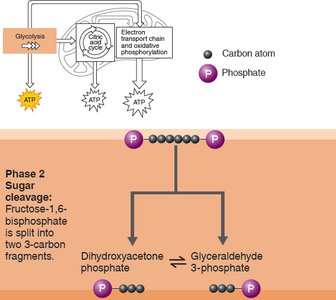
Phase 3: Sugar Oxidation and ATP Formation – Each 3-carbon fragment is oxidized, NAD+ is reduced to NADH, and ATP is generated by substrate-level phosphorylation. End products: 2 pyruvic acid, 2 NADH, 4 ATP (net gain: 2 ATP).
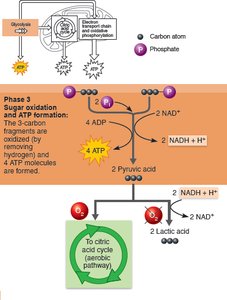
Fate of Pyruvic Acid
Aerobic conditions: Pyruvic acid enters mitochondria for further oxidation.
Anaerobic conditions: NADH reduces pyruvic acid to lactic acid, which can be converted back to glucose in the liver or oxidized when oxygen is available.
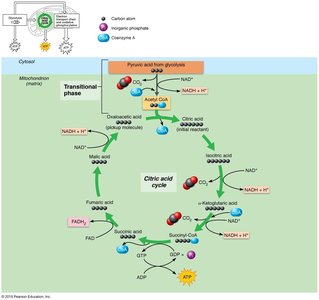
Citric Acid Cycle (Krebs Cycle)
Occurs in the mitochondrial matrix and is fueled by acetyl CoA derived from pyruvic acid and fatty acids. The cycle generates NADH, FADH2, and CO2.
Transitional Phase: Pyruvic acid is converted to acetyl CoA via decarboxylation, oxidation, and combination with coenzyme A.
Cycle Steps: Acetyl CoA combines with oxaloacetic acid to form citric acid, which is then decarboxylated and oxidized through a series of steps, regenerating oxaloacetic acid.
Intermediates: Fats and proteins can enter the cycle at various points; intermediates may be used for anabolic reactions.
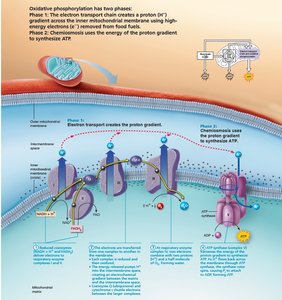
Electron Transport Chain and Oxidative Phosphorylation
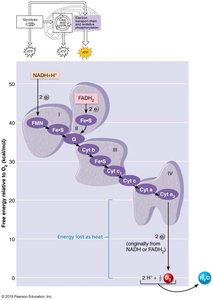
The ETC is located in the inner mitochondrial membrane and consists of a series of protein complexes that transfer electrons from NADH and FADH2 to oxygen, the final electron acceptor. This process creates a proton gradient used by ATP synthase to generate ATP.
Phase 1: Electron transport creates a proton (H+) gradient across the membrane.
Phase 2: Chemiosmosis uses the energy of the proton gradient to synthesize ATP.
ATP Yield and Efficiency
Typical ATP yield per glucose: About 30 ATP (varies with shuttle systems and cell type).
Efficiency: About 38% of glucose energy is captured as ATP; the rest is lost as heat.
Lipid Metabolism
Oxidation of Glycerol and Fatty Acids
Glycerol: Converted to glyceraldehyde 3-phosphate, enters glycolysis, yields about 15 ATP per glycerol.
Fatty acids: Undergo beta oxidation in mitochondria, producing acetyl CoA, NADH, and FADH2. Acetyl CoA enters the citric acid cycle.
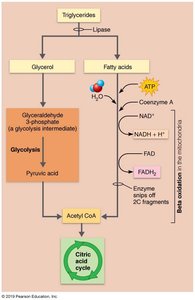
Lipolysis and Ketone Body Formation
Lipolysis: Breakdown of stored fats into glycerol and fatty acids, accelerated when carbohydrate intake is low.
Beta oxidation: Fatty acids are converted to acetyl CoA; if intermediates are insufficient, acetyl CoA is converted to ketone bodies (acetoacetic acid, β-hydroxybutyric acid, acetone) in the liver.
Lipoproteins and Cholesterol Metabolism
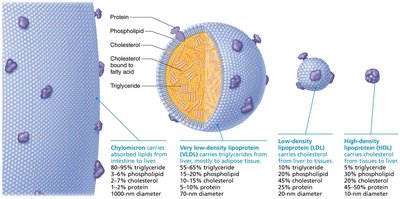
Composition and Function of Lipoproteins
Lipoproteins transport lipids in the blood. Their composition determines their function and health impact.
Chylomicrons: Carry absorbed lipids from intestine to liver.
VLDLs (Very Low-Density Lipoproteins): Transport triglycerides from liver to adipose tissue.
LDLs (Low-Density Lipoproteins): Deliver cholesterol to peripheral tissues; high levels are associated with atherosclerosis.
HDLs (High-Density Lipoproteins): Remove excess cholesterol from tissues and transport it to the liver for excretion; protective against heart disease.
Cholesterol Regulation
Recommended levels: Total cholesterol < 200 mg/dl; HDL > 60 mg/dl (good), LDL < 100 mg/dl (good).
Health implications: High LDL increases risk of cardiovascular disease; high HDL is protective.
Key Terms and Concepts
Essential nutrient: A nutrient the body cannot synthesize rapidly enough to meet its needs (must be obtained from the diet).
Glycogenesis: The process of storing excess glucose as glycogen in cells.
Beta oxidation: The process of breaking down fatty acids into acetyl CoA units in mitochondria.
ATP synthase: Enzyme complex that synthesizes ATP using the energy of a proton gradient.
Summary Table: Major Pathways of Cellular Respiration
Pathway | Location | Oxygen Required? | Main Products |
|---|---|---|---|
Glycolysis | Cytosol | No | 2 ATP, 2 NADH, 2 pyruvic acid |
Transition (Pyruvate Grooming) | Mitochondrial matrix | Yes | 2 acetyl CoA, 2 NADH, 2 CO2 |
Citric Acid Cycle | Mitochondrial matrix | Yes | 2 ATP, 6 NADH, 2 FADH2, 4 CO2 |
Electron Transport Chain | Inner mitochondrial membrane | Yes | ~28 ATP, H2O |
Additional info: This guide expands on the provided slides by integrating definitions, examples, and academic context for each process. For further study, refer to the full textbook chapter and practice with end-of-chapter questions.
