 Back
BackFluid, Electrolyte, and Acid-Base Balance: Study Notes for Anatomy & Physiology II
Study Guide - Smart Notes

Fluid, Electrolyte, and Acid-Base Balance
Introduction
Fluid, electrolyte, and acid-base balance are essential for maintaining homeostasis in the human body. These processes ensure that the body's internal environment remains stable, supporting cellular function and overall health. This chapter covers the distribution and regulation of body fluids, the roles of key electrolytes, and the mechanisms that maintain acid-base balance.
Body Water Content
Variation by Age, Sex, and Body Composition
Total body water varies with age, body mass, sex, and body fat percentage.
Infants: ~75-80% of body weight is water.
Young adults: ~50-60% (Females: ~50-55%, Males: ~55-60%).
Elderly: ~45% of body weight is water.
Fluid Compartments in the Body
Major Fluid Compartments
Intracellular fluid (ICF): ~65% of body fluids, contained within cells.
Extracellular fluid (ECF): ~35% of body fluids, includes:
Intravascular fluid (plasma, lymph)
Interstitial fluid (between cells)
Transcellular fluid (cerebrospinal fluid, synovial fluid, serous fluid, etc.)
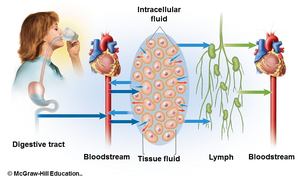
Fluid Balance
Fluid Gains and Losses
For proper hydration, fluid intake must equal fluid output.
Fluid gains:
Beverage ingestion
Food ingestion
Cellular respiration (metabolic water)
Fluid losses:
Sensible losses: sweat, urine, feces
Insensible losses: evaporation from skin and respiratory tract

Regulation of Fluid Intake
Thirst Mechanism
Fluid intake is primarily regulated by the hypothalamic thirst center.
Stimuli for thirst include increased plasma osmolality, decreased blood volume/pressure, and dry mouth.
Activation of the thirst center leads to decreased salivation, dry mouth, and the sensation of thirst.
Regulation of Fluid Output
Hormonal Control of Urine Volume
Fluid output is mainly regulated by urine volume, which is controlled by hormones such as antidiuretic hormone (ADH), aldosterone, and atrial natriuretic peptide (ANP).
During dehydration: increased ADH, renin/angiotensin, and aldosterone promote water reabsorption.
During overhydration: decreased ADH, renin/angiotensin, and aldosterone reduce water reabsorption.
Electrolyte Balance
Sodium (Na+)
Most abundant extracellular ion (90-95%).
Functions:
Drives osmosis between fluid compartments.
Essential for Na+/K+ pump, resting membrane potential, and action potentials.
Drives cotransport of many molecules across membranes.
Sodium bicarbonate acts as a buffer in ECF.
Imbalances:
Excess sodium: Hypernatremia
Deficiency: Hyponatremia
Hormonal regulation: Aldosterone increases sodium reabsorption in kidney tubules. ADH indirectly affects sodium concentration by regulating water reabsorption. ANP decreases sodium reabsorption.
Potassium (K+)
Most abundant intracellular cation.
Functions:
Drives osmosis into/out of cells.
Essential for Na+/K+ pump, resting membrane potential, action potentials, and pH balance.
Imbalances:
Excess potassium: Hyperkalemia
Deficiency: Hypokalemia
Imbalances can be life-threatening due to effects on nerve and muscle excitability, especially cardiac muscle.
Potassium homeostasis is regulated by kidney tubules in response to ECF potassium levels and influenced by aldosterone.
Calcium (Ca2+)
99% of calcium is stored in bones.
Functions:
Bone formation, muscle contraction, neuron signaling, blood coagulation, cell division, exocytosis, cell signaling, and enzyme function.
Imbalances:
Excess calcium: Hypercalcemia
Deficiency: Hypocalcemia
Parathyroid hormone (PTH) is the main regulator, raising blood calcium by stimulating bone resorption, increasing intestinal absorption, and enhancing renal reabsorption.
Acid-Base Balance
Generation of Acids and Buffer Types
Acids are produced by protein breakdown (phosphoric acid), anaerobic respiration (lactic acid), fat catabolism (fatty acids), and CO2 transport (H+ release).
Two buffer types:
Chemical buffer systems: Act rapidly to resist pH changes by binding or releasing H+.
Physiologic buffer systems: Respiratory and urinary systems that remove acids or bases from the body.
Chemical Buffer Systems
Buffers minimize pH changes by binding or releasing H+.
Major systems:
Bicarbonate buffer system: Active in ICF and ECF; most important in ECF.
Phosphate buffer system: Active in ICF and urine.
Protein buffer system: Active in ICF; proteins act as buffers due to their amino and carboxyl groups.
Bicarbonate buffer equation:
Phosphate buffer equation:
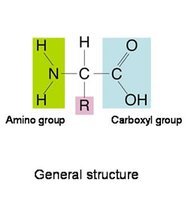
Physiologic Buffer Systems
Respiratory System
Regulates pH by controlling CO2 exhalation; acts within minutes.
Increased ventilation removes CO2, raising pH (compensates for acidosis).
Decreased ventilation retains CO2, lowering pH (compensates for alkalosis).
Urinary System
Regulates pH by secreting H+ and reabsorbing/generating HCO3-; acts over hours to days.
Kidneys are the only organs that can eliminate metabolic acids and renew chemical buffers.
Impairment leads to acid-base imbalances.
Renal Regulation of pH
High blood CO2 lowers pH; kidneys respond by reabsorbing HCO3- and secreting H+ into urine.
This process alkalinizes blood and acidifies urine.
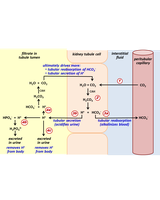
Acid-Base Disorders
Normal pH and Definitions
Normal arterial blood pH: 7.35–7.45
Acidosis (acidemia): pH < 7.35
Alkalosis (alkalemia): pH > 7.45
Types of Acid-Base Disorders
Respiratory acidosis/alkalosis: Caused by changes in CO2 due to respiratory dysfunction.
Acidosis: Slow/shallow breathing, impaired gas exchange, CNS depression.
Alkalosis: Hyperventilation, stress, pain, panic attacks.
Metabolic acidosis/alkalosis: Caused by changes in HCO3- or acid production/excretion.
Acidosis: Excess alcohol, loss of HCO3-, lactic acid buildup, lipid catabolism, renal disease.
Alkalosis: Loss of H+ (vomiting), excess antacid intake.
