 Back
BackFluid, Electrolyte, and Acid-Base Balance: Core Concepts and Homeostatic Mechanisms
Study Guide - Smart Notes

Fluid, Electrolyte, and Acid-Base Balance
Introduction
The maintenance of fluid, electrolyte, and acid-base balance is essential for normal physiological function. These processes ensure the stability of body fluid volumes, solute concentrations, and pH within the extracellular fluid (ECF) and intracellular fluid (ICF) compartments.
Fluid balance: Regulation of water input and output to maintain homeostasis.
Electrolyte balance: Maintenance of ion concentrations for cellular function.
Acid-base balance: Control of hydrogen ion concentration to stabilize pH.
Body Fluid Compartments
Distribution of Body Water
Body water is distributed between two main compartments: the intracellular fluid (ICF) and the extracellular fluid (ECF). The ICF is the cytosol within cells, while the ECF includes interstitial fluid, plasma, and minor components such as lymph and cerebrospinal fluid.
ICF: Contains about two-thirds of total body water.
ECF: Contains about one-third of total body water, subdivided into plasma and interstitial fluid.
Water accounts for approximately 60% of adult male and 50% of adult female body weight.
Major Ions in Fluid Compartments
ECF: Dominated by sodium (Na+), chloride (Cl-), and bicarbonate (HCO3-).
ICF: Dominated by potassium (K+), magnesium (Mg2+), and phosphate (PO43-).
Negatively charged proteins are more abundant in the ICF.
Fluid Balance
Water Gains and Losses
Fluid balance is achieved when daily water gains equal losses. The digestive system is the primary source of water gain, while the urinary system is the main route of water loss.
Gains | Daily Input (mL) |
|---|---|
Water content of food | 1000 |
Water consumed as liquid | 1200 |
Metabolic water produced during catabolism | 300 |
Total | 2500 |
Losses | Daily Output (mL) |
|---|---|
Urination | 1200 |
Evaporation at skin | 750 |
Evaporation at lungs | 400 |
Loss in feces | 150 |
Total | 2500 |

Mechanisms of Fluid Movement
Water moves between ECF and ICF primarily by osmosis, maintaining osmotic equilibrium. Fluid shifts occur rapidly in response to changes in osmotic gradients, such as dehydration or overhydration.
If ECF becomes hypertonic, water moves from ICF to ECF.
If ECF becomes hypotonic, water moves from ECF to ICF.
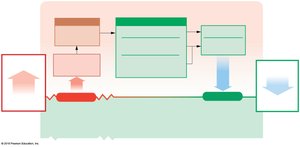
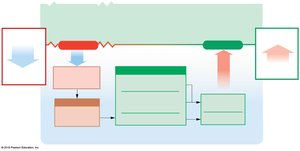
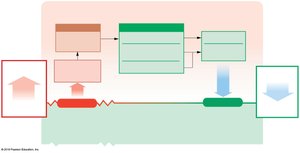
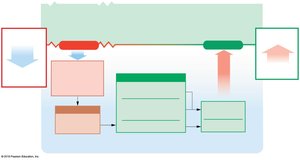
Regulation of Fluid and Electrolyte Balance
Three primary hormones regulate fluid and electrolyte balance:
Antidiuretic hormone (ADH): Promotes water conservation at the kidneys and stimulates thirst.
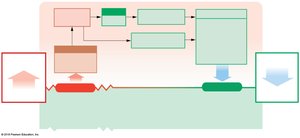
Aldosterone: Increases sodium reabsorption and potassium excretion in the kidneys; water follows sodium.
Natriuretic peptides (ANP and BNP): Promote sodium and water loss, reduce thirst, and inhibit ADH and aldosterone release.
Electrolyte Balance
Sodium Balance
Sodium is the dominant cation in the ECF and is crucial for maintaining osmotic pressure and fluid balance. Sodium balance is regulated by dietary intake and renal excretion.
Normal Na+ concentration in ECF: ~140 mEq/L.
Imbalances can lead to hyponatremia (Na+ < 135 mEq/L) or hypernatremia (Na+ > 145 mEq/L).
Potassium Balance
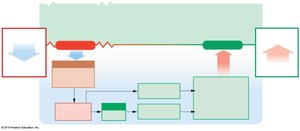
Potassium is the dominant cation in the ICF and is essential for normal neuromuscular function. Potassium balance is maintained by dietary intake and renal excretion, influenced by aldosterone.
Normal K+ concentration in ICF: ~160 mEq/L; in ECF: 3.5–5.5 mEq/L.
Imbalances can cause hypokalemia (K+ < 3.5 mEq/L) or hyperkalemia (K+ > 5.5 mEq/L).
Calcium, Magnesium, Phosphate, and Chloride Balance
Calcium: Most abundant mineral, regulated by PTH, calcitriol, and calcitonin.
Magnesium: Required for enzymatic reactions, mainly in ICF.
Phosphate: Important for bone mineralization and energy metabolism.
Chloride: Major anion in ECF, follows sodium movement.
Acid-Base Balance
pH Regulation
The pH of body fluids is tightly regulated between 7.35 and 7.45. Acidosis (pH < 7.35) and alkalosis (pH > 7.45) can disrupt cellular function, especially in the nervous and cardiovascular systems.
Fixed acids: Remain in body fluids until excreted by kidneys (e.g., sulfuric acid).
Metabolic acids: By-products of metabolism (e.g., lactic acid, ketone bodies).
Volatile acids: Can be excreted by the lungs (e.g., carbonic acid).
Buffer Systems
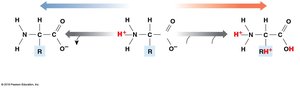
Buffers stabilize pH by binding or releasing H+ ions. The three major buffer systems are:
Phosphate buffer system: Important in ICF and urine.
Protein buffer systems: Rely on amino acids; function in both ECF and ICF.
Carbonic acid–bicarbonate buffer system: Most important in ECF.
Respiratory and Renal Compensation
Respiratory and renal mechanisms work with buffer systems to maintain acid-base balance:
Respiratory compensation: Adjusts CO2 exhalation to influence blood pH.
Renal compensation: Adjusts H+ secretion and HCO3- reabsorption.
Disorders of Acid-Base Balance
Respiratory acidosis: Caused by hypoventilation, leading to CO2 retention and decreased pH.
Respiratory alkalosis: Caused by hyperventilation, leading to CO2 loss and increased pH.
Metabolic acidosis: Due to increased acid production, impaired H+ excretion, or bicarbonate loss.
Metabolic alkalosis: Due to excessive loss of H+ or gain of HCO3-.
Effects of Aging
Age-Related Changes in Fluid and Acid-Base Balance
With aging, total body water content decreases, renal and respiratory compensatory mechanisms decline, and the risk of fluid, electrolyte, and acid-base imbalances increases. Maintaining adequate hydration and mineral intake becomes more important in older adults.
Decreased glomerular filtration rate and nephron number reduce pH regulation.
Reduced sensitivity to ADH and aldosterone impairs water conservation.
Loss of muscle and bone mass decreases mineral reserves.
