 Back
BackChapters 2 & 3 Molecular Interactions and Cell Chemistry in Anatomy & Physiology
Study Guide - Smart Notes

Chapter 2 – Molecular Interactions
Why Study Chemistry in Anatomy & Physiology?
Understanding chemistry is essential for grasping physiological processes, as the body is composed of chemicals that interact continuously. These interactions are the foundation of all bodily functions, making chemistry the underlying basis of physiology.
Matter: Anything that takes up space and has mass.
Mass: Amount of matter in an object; constant regardless of location.
Weight: Gravity-dependent and can vary.
Forms of Matter
Solids: e.g., bones
Liquids: e.g., blood
Gases: e.g., oxygen
Energy and Its Forms
Energy is the capacity to do work and is measured by its effect on matter. It exists in two main types:
Kinetic Energy: Energy in action (e.g., a rolling rock).
Potential Energy: Stored energy (e.g., a rock at the top of a hill).
Forms of energy include:
Chemical: Stored in bonds; released during chemical reactions. ATP is the primary energy currency in cells.
Electrical: Movement of charged particles (e.g., ions across membranes in nerve impulses).
Mechanical: Directly involved in moving matter.
Radiant: Travels in waves (e.g., visible light, UV light).
Energy Conversions
Energy can be converted between forms, but some is always lost as heat, contributing to body temperature regulation.
Composition of Matter
Elements and Atoms
Elements are unique substances that cannot be broken down by ordinary chemical means. The human body is primarily composed of four elements: oxygen, carbon, hydrogen, and nitrogen.
Atoms: The smallest units of elements, consisting of protons, neutrons, and electrons.
Protons: Positive charge, mass = 1 amu.
Neutrons: Neutral charge, mass = 1 amu.
Electrons: Negative charge, negligible mass, orbit the nucleus.
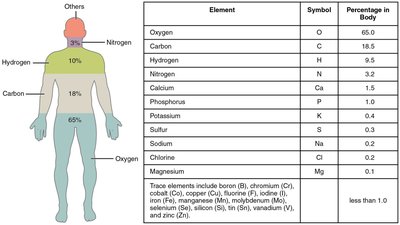
Table Purpose: This table and diagram show the major elements in the human body, their symbols, and their relative abundance. It highlights the importance of oxygen, carbon, hydrogen, and nitrogen, which together make up over 96% of body mass.
Atomic Structure and Characteristics
Atomic Number: Number of protons in an atom; unique to each element.
Mass Number: Sum of protons and neutrons.
Isotopes: Atoms with the same number of protons but different numbers of neutrons.
Atomic Weight: Average mass of all isotopes of an element.
Radioisotopes
Unstable isotopes that emit radiation as they decay to more stable forms.
Half-life: Time required for half of a radioisotope to decay.
Used in medical imaging and cancer treatment, but can damage living tissue.
Molecules, Compounds, and Mixtures
Definitions
Molecule: Two or more atoms bonded together (e.g., O2).
Compound: Two or more different atoms bonded together (e.g., H2O).
Mixture: Physical combination of substances without chemical bonding (e.g., sugar water).
Types of Mixtures
Solutions: Homogeneous mixtures; solute dissolved in solvent (e.g., saline solution).
Colloids: Heterogeneous, milky or translucent; particles do not settle (e.g., cytosol).
Suspensions: Heterogeneous, large particles settle out (e.g., blood cells in plasma).
Mixtures vs. Compounds
Mixtures can be separated physically; compounds require chemical reactions to separate.
All compounds are homogeneous; mixtures can be homogeneous or heterogeneous.
Chemical Bonds
Electron Shells and Bonding
Electrons occupy energy levels (shells) around the nucleus. The outermost shell (valence shell) determines chemical reactivity. Atoms are most stable when their outer shell is full (usually 8 electrons, except for the first shell, which holds 2).
Types of Chemical Bonds
Ionic Bonds: Formed by the transfer of electrons, resulting in oppositely charged ions (e.g., NaCl).
Covalent Bonds: Formed by sharing electrons. Can be nonpolar (equal sharing) or polar (unequal sharing, as in water).
Hydrogen Bonds: Weak attractions between a hydrogen atom and an electronegative atom (important in water and DNA structure).
Chemical Reactions
Types of Reactions
Synthesis (Anabolic): Building larger molecules from smaller ones ().
Decomposition (Catabolic): Breaking down molecules ().
Exchange: Both synthesis and decomposition ().
Redox (Oxidation-Reduction): Transfer of electrons; important in energy production ().
Reaction Reversibility and Rate
Most reactions are reversible but tend toward equilibrium.
Factors affecting rate: temperature, particle size, concentration, and catalysts (enzymes).
Biochemistry: Organic and Inorganic Compounds
Inorganic Compounds
Water: Most abundant; high heat capacity, solvent, reactivity, and cushioning properties.
Salts: Ionic compounds that dissociate into electrolytes (e.g., Na+, K+, Ca2+).
Acids and Bases: Acids release H+ (proton donors), bases accept H+ (proton acceptors). pH scale measures acidity/alkalinity.
Buffers: Resist changes in pH to maintain homeostasis (e.g., blood pH 7.35–7.45).
Organic Compounds
Carbohydrates: Sugars and starches; main energy source. Classified as monosaccharides, disaccharides, and polysaccharides.
Lipids: Fats, oils, phospholipids, steroids; energy storage, cell membranes, hormones.
Proteins: Made of amino acids; structural and functional roles (enzymes, antibodies, etc.).
Nucleic Acids: DNA and RNA; genetic information and protein synthesis.
Cells: The Basic Unit of Life
Cell Theory
All living things are composed of cells.
Cells are the basic unit of structure and function.
All cells arise from pre-existing cells.
Cell function depends on internal structures (organelles).
Cell Structure
Plasma Membrane: Phospholipid bilayer with embedded proteins; selectively permeable.
Cytoplasm: Semi-fluid matrix containing organelles.
Nucleus: Control center containing DNA.
Membrane Specializations
Microvilli: Increase surface area for absorption.
Membrane Junctions: Tight junctions, desmosomes, and gap junctions for cell-to-cell communication and adhesion.
Membrane Transport
Passive Transport: No energy required; includes diffusion, osmosis, facilitated diffusion, and filtration.
Active Transport: Requires ATP; includes solute pumps and bulk transport (endocytosis, exocytosis).
Resting Membrane Potential
Established by the Na+/K+ pump; inside of the cell is negative relative to the outside.
Essential for nerve and muscle function.
Cell Interactions
Glycoproteins on the cell surface mediate cell recognition, immune response, and tissue formation.
Cell Growth and Reproduction
Cell Cycle
Interphase: Growth, DNA replication, preparation for division.
Mitosis: Division of the nucleus into two identical nuclei (prophase, metaphase, anaphase, telophase).
Cytokinesis: Division of the cytoplasm, forming two new cells.
DNA Replication
DNA polymerases synthesize new DNA strands using original strands as templates (semi-conservative replication).
Protein Synthesis
Transcription: DNA code is transcribed to mRNA in the nucleus.
Translation: mRNA is translated into a protein at the ribosome with the help of tRNA and rRNA.
Summary Table: Major Elements in the Human Body
Element | Symbol | Percentage in Body |
|---|---|---|
Oxygen | O | 65.0 |
Carbon | C | 18.5 |
Hydrogen | H | 9.5 |
Nitrogen | N | 3.2 |
Calcium | Ca | 1.5 |
Phosphorus | P | 1.0 |
Potassium | K | 0.4 |
Sulfur | S | 0.3 |
Sodium | Na | 0.2 |
Chlorine | Cl | 0.2 |
Magnesium | Mg | 0.1 |
Trace elements (less than 1.0%): B, Cr, Co, Cu, F, I, Fe, Mn, Mo, Se, Si, Sn, V, Zn | ||
Additional info: This table is essential for understanding the elemental composition of the human body, which is foundational for all physiological processes.
