 Back
BackAn Introduction to Metabolism: Energy, Enzymes, and Regulation in Biological Systems
Study Guide - Smart Notes

An Introduction to Metabolism
The Energy of Life
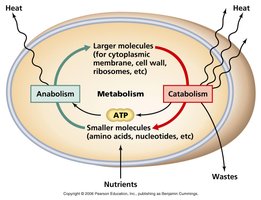
Metabolism encompasses all chemical reactions within a living organism, enabling the transformation of matter and energy. These reactions are essential for cellular processes such as growth, maintenance, and response to environmental changes.
Metabolism: The sum total of an organism’s chemical reactions.
Cellular respiration extracts energy from sugars and fuels, which is then used to perform cellular work.
Metabolism is an emergent property resulting from the orderly interactions between molecules.
Organization of Metabolic Pathways
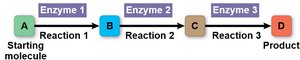
Metabolic pathways are sequences of chemical reactions, each catalyzed by a specific enzyme, that transform a starting molecule into a product. These pathways are tightly regulated to balance supply and demand within the cell.
Metabolic pathway: A series of reactions that build (anabolic) or break down (catabolic) molecules.
Each step is catalyzed by a specific enzyme.
Regulation ensures efficient use of resources.
Catabolic and Anabolic Pathways
Metabolic pathways are classified as catabolic or anabolic based on their function and energy flow.
Catabolic pathways: Release energy by breaking down complex molecules (e.g., cellular respiration).
Anabolic pathways: Consume energy to build complex molecules (e.g., protein synthesis).
Bioenergetics: The study of how energy flows through living organisms.
Forms and Transformations of Energy
Types of Energy
Energy is the capacity to cause change and exists in various forms relevant to biological systems.
Kinetic energy: Energy of motion.
Thermal energy: Kinetic energy due to random movement of atoms or molecules.
Heat: Transfer of thermal energy between objects.
Potential energy: Stored energy due to position or structure.
Chemical energy: Potential energy available for release in chemical reactions.


The Laws of Thermodynamics
Biological systems obey the laws of thermodynamics, which govern energy transformations.
First Law (Conservation of Energy): Energy can be transferred and transformed, but not created or destroyed.
Second Law: Every energy transfer increases the entropy (disorder) of the universe; some energy is lost as heat.
Organisms are open systems, exchanging energy and matter with their surroundings.
Entropy and Spontaneous Processes
Entropy is a measure of disorder. Spontaneous processes increase the entropy of the universe and do not require energy input, while nonspontaneous processes require energy.
Living cells increase local order but contribute to overall entropy by releasing heat and waste.
Organisms are islands of low entropy in a universe trending toward disorder.
Free Energy and Biological Reactions
Free-Energy Change (ΔG)
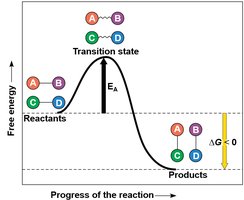
Free energy (G) is the portion of a system’s energy available to do work at constant temperature and pressure. The change in free energy (ΔG) predicts whether a process is spontaneous.
ΔG = ΔH − TΔS
ΔG < 0: Spontaneous process
ΔG > 0: Nonspontaneous process
ΔH: Change in enthalpy (total energy); ΔS: Change in entropy; T: Temperature in Kelvin

Stability, Equilibrium, and Work
Systems tend toward greater stability (lower free energy). At equilibrium, no work can be done, and metabolic reactions in living cells are kept away from equilibrium to sustain life.
Spontaneous changes decrease free energy and increase stability.
Cells maintain disequilibrium by expelling waste and taking in nutrients.
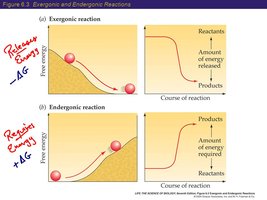
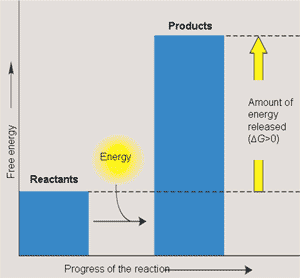
Exergonic and Endergonic Reactions
Reactions are classified by their free energy change:
Exergonic reactions: Release free energy (ΔG < 0), spontaneous.
Endergonic reactions: Absorb free energy (ΔG > 0), nonspontaneous.
ATP: The Energy Currency of the Cell
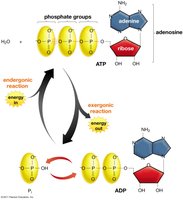
ATP Structure and Hydrolysis
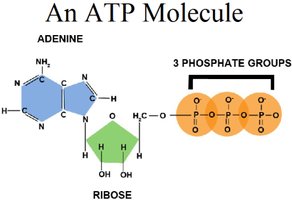
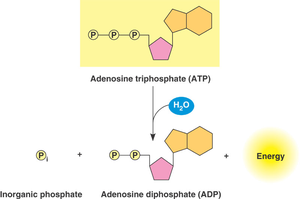
ATP (adenosine triphosphate) is the primary energy carrier in cells. Hydrolysis of ATP releases energy by converting ATP to ADP and inorganic phosphate (Pi).
ATP consists of adenine, ribose, and three phosphate groups.
Hydrolysis of the terminal phosphate releases energy (ΔG ≈ -7.3 kcal/mol under standard conditions).
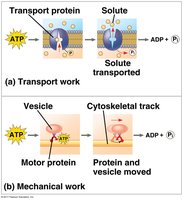
ATP and Energy Coupling
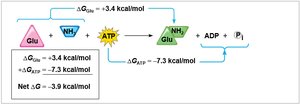
Cells use ATP hydrolysis to drive endergonic reactions by transferring a phosphate group to a reactant (phosphorylation), making it more reactive.
Energy coupling links exergonic and endergonic reactions.
Phosphorylated intermediates are more reactive and less stable.
Enzymes and Metabolic Regulation
Enzymes as Biological Catalysts
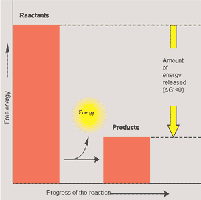
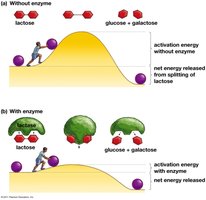
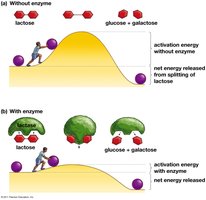
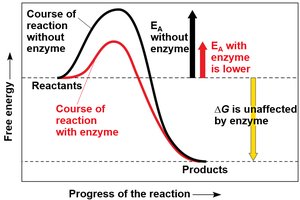
Enzymes are proteins that speed up metabolic reactions by lowering the activation energy (EA) barrier, allowing reactions to occur at moderate temperatures.
Catalyst: Substance that increases reaction rate without being consumed.
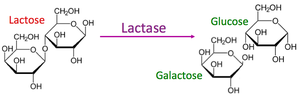
Enzyme: Biological catalyst, usually a protein (e.g., lactase).
Activation Energy and the Transition State
Activation energy is the initial energy required to start a reaction. Enzymes lower this barrier, enabling reactants to reach the transition state more easily.
Activation energy (EA) is often supplied as heat.
Enzymes do not affect ΔG; they only speed up the rate of reaction.
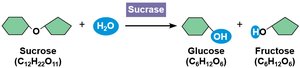
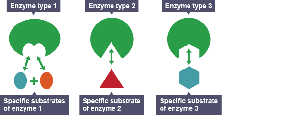
Enzyme Specificity and Mechanism
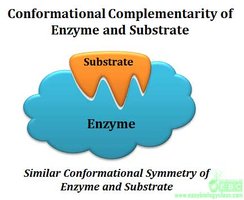
Enzymes are highly specific, recognizing their substrates through a unique three-dimensional active site. The induced fit model describes how enzyme binding changes shape to enhance catalysis.
Substrate: The reactant an enzyme acts upon.
Active site: Region on the enzyme where the substrate binds.
Induced fit: Enzyme changes shape for optimal substrate binding.

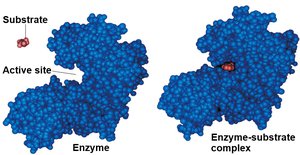
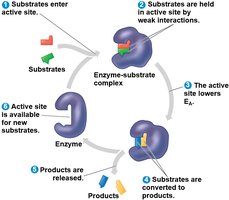
Catalysis in the Active Site
The active site lowers activation energy by orienting substrates, straining bonds, providing a favorable microenvironment, or forming temporary covalent bonds with the substrate.
Factors Affecting Enzyme Activity
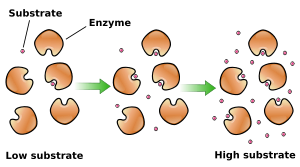
Enzyme activity is influenced by substrate concentration, temperature, pH, and the presence of cofactors or inhibitors.
Enzymes have optimal temperature and pH ranges.
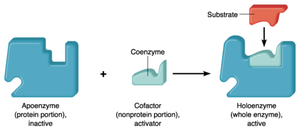
Cofactors: Nonprotein helpers (inorganic or organic, e.g., vitamins).

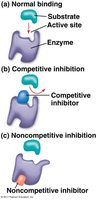
Enzyme Inhibition
Enzyme inhibitors can be competitive (binding the active site) or noncompetitive (binding elsewhere and altering enzyme shape). Inhibition is a key regulatory mechanism in metabolism.
Competitive inhibitors: Compete with substrate for active site.
Noncompetitive inhibitors: Bind elsewhere, changing enzyme conformation.
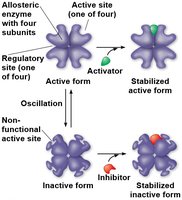
Regulation of Enzyme Activity
Cells regulate metabolism by controlling enzyme activity through allosteric regulation, cooperativity, and feedback inhibition.
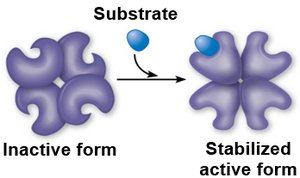
Allosteric regulation: Regulatory molecules bind to sites other than the active site, stabilizing active or inactive forms.
Cooperativity: Substrate binding to one active site increases activity at other sites.
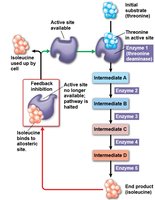
Feedback inhibition: End product of a pathway inhibits an upstream enzyme, preventing overproduction.
Localization of Enzymes
Enzymes are often compartmentalized within cells, residing in specific organelles or as part of membrane structures, to organize and regulate metabolic pathways efficiently.
