 Back
BackAtoms, Molecules, and the Chemical Basis of Life: Study Notes
Study Guide - Smart Notes

Atoms and Molecules: The Chemical Basis of Life
Inorganic and Organic Compounds
Biological systems are composed of both inorganic and organic compounds, each playing distinct roles in cellular structure and function.
Inorganic Compounds: Small, simple substances such as water, acids, bases, and salts. These are crucial for physiological processes and maintaining cellular environments.
Organic Compounds: Generally large, complex molecules containing carbon. Carbon atoms form the backbone of these molecules, enabling the diversity of life’s chemistry.

Elements and Atoms
Principal Chemical Elements in Living Things
Living organisms are primarily composed of a few key elements, each with specific biological functions.
Oxygen, Carbon, Hydrogen, Nitrogen: Make up over 96% of the mass of most organisms.
Calcium, Phosphorus, Potassium, Sulfur, Sodium, Magnesium: Present in smaller quantities but essential for various physiological roles.
Trace Elements: Iodine and copper are required in minute amounts for proper function.

Atoms: Structure and Properties
An atom is the smallest unit of an element that retains its chemical properties. Atoms are composed of subatomic particles: protons, neutrons, and electrons.
Protons: Positively charged, located in the nucleus.
Neutrons: Neutral, located in the nucleus.
Electrons: Negatively charged, orbit the nucleus.
Matter: Anything that has mass and occupies space.

Atomic Number and Mass Number
The atomic number and mass number are fundamental properties of atoms, determining their identity and isotopic variation.
Atomic Number: Number of protons in the nucleus; defines the element.
Mass Number: Sum of protons and neutrons in the nucleus.
Atomic Mass Unit (AMU) or Dalton: Standard unit for measuring atomic and molecular mass.


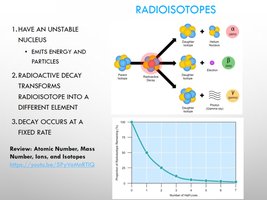
Isotopes and Radioisotopes
Isotopes are atoms of the same element with different numbers of neutrons. Radioisotopes are unstable isotopes that undergo radioactive decay.
Isotopes: Same number of protons, different number of neutrons.
Radioisotopes: Emit energy and particles, transform into different elements, decay at a fixed rate.
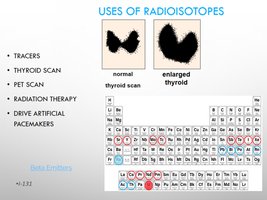
Uses: Tracers, medical imaging (thyroid scan, PET scan), radiation therapy, pacemakers.

Electron Structure and Chemical Behavior
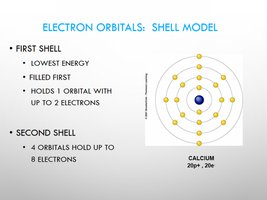
Electrons: Characteristics and Orbitals
Electrons determine the chemical behavior of atoms through their arrangement in orbitals and shells.
Electrons: Carry negative charge, repel each other, attracted to protons.
Orbitals: Volumes of space around the nucleus where electrons are likely to be found.
Shell Model: First shell holds up to 2 electrons; second shell holds up to 8 electrons.

Valence Electrons and Electron Vacancies
Valence electrons are those in the outermost shell and are crucial for chemical bonding. Atoms with unfilled valence shells tend to react to achieve stability.
Valence Electrons: Determine chemical reactivity and bonding.
Octet Rule: Atoms tend to fill their outer shell with 8 electrons (except hydrogen, which fills with 2).
Electron Vacancies: Atoms with unfilled shells are likely to gain, lose, or share electrons.


Chemical Bonds, Molecules, and Compounds
Chemical Bonds: Types and Formation
Chemical bonds are forces that hold atoms together in molecules and compounds. The main types are ionic, covalent, and hydrogen bonds.
Ionic Bonds: Formed by transfer of electrons, resulting in charged ions (cations and anions).
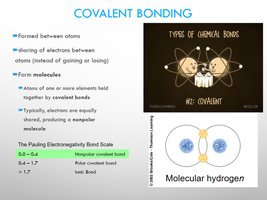
Covalent Bonds: Formed by sharing electrons between atoms.
Hydrogen Bonds: Weak attractions between a hydrogen atom and an electronegative atom (e.g., oxygen, nitrogen).

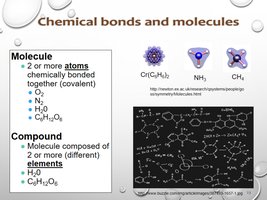
Molecules and Compounds
A molecule consists of two or more atoms bonded together. A compound is a molecule composed of different elements.
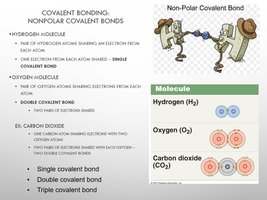
Molecule: O2, N2, H2 (covalent bonds).
Compound: H2O, C6H12O6 (different elements).
Chemical Bookkeeping: Molecular Formulas and Equations
Molecular formulas represent the composition of molecules, while chemical equations show reactions.
Molecular Formula: Indicates the number and type of atoms in a molecule (e.g., C6H12O6 for glucose).
Chemical Equation: Shows reactants and products. Example: Photosynthesis
Photosynthesis equation:

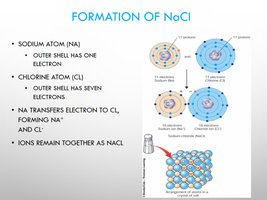
Ionic Bonding
Ionic bonds form when electrons are transferred from one atom to another, creating ions that attract each other.
Cations: Positively charged ions (lose electrons).
Anions: Negatively charged ions (gain electrons).
Example: Formation of NaCl (sodium chloride).

Covalent Bonding
Covalent bonds involve the sharing of electrons between atoms. They can be nonpolar (equal sharing) or polar (unequal sharing).
Nonpolar Covalent Bonds: Electrons shared equally (e.g., H2, O2).
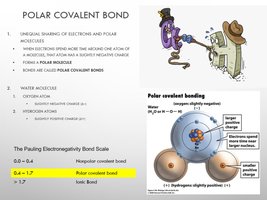
Polar Covalent Bonds: Electrons shared unequally, creating partial charges (e.g., H2O).
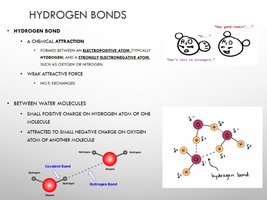
Hydrogen Bonds
Hydrogen bonds are weak attractions between a hydrogen atom and an electronegative atom. They are crucial for the structure of water and biological macromolecules.
Between Water Molecules: Hydrogen bonds form between the slightly positive hydrogen and slightly negative oxygen atoms of adjacent water molecules.
Biological Importance: Stabilize DNA, proteins, and contribute to water’s unique properties.

Properties of Water
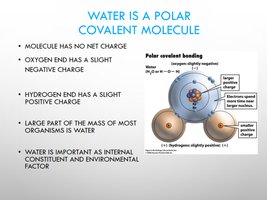
Water as a Polar Covalent Molecule
Water’s polarity results from the unequal sharing of electrons between hydrogen and oxygen, giving rise to its unique properties.
Oxygen: Slight negative charge.
Hydrogen: Slight positive charge.
Importance: Water is a major component of organisms and an essential environmental factor.
Key Properties of Water
Water exhibits several properties essential for life, including cohesion, adhesion, temperature stabilization, and solvent capacity.
Cohesion: Tendency of water molecules to stick together.
Adhesion: Tendency to stick to other substances.
Temperature Stabilizing: High heat capacity.
Solvent Capacity: Dissolves many substances.
Expands When Freezes: Ice is less dense than liquid water.

The Importance of Water in the Body
Water is the most important component of the human body, affecting all physiological systems.
Lubrication: Reduces friction in joints.
Reactivity: Participates in chemical reactions.
High Heat Capacity: Helps regulate body temperature.
Solubility: Solvent for many substances.

Cohesion and Adhesion
Cohesion and adhesion are responsible for water’s movement in plants and its ability to make surfaces wet.
Cohesion: Hydrogen bonds among molecules transmit force throughout a column of water.
Adhesion: Water sticks to other substances due to surface charges.

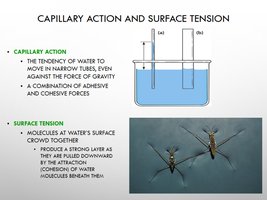
Capillary Action and Surface Tension
Capillary action and surface tension are phenomena resulting from water’s cohesive and adhesive properties.
Capillary Action: Water moves in narrow tubes against gravity.
Surface Tension: Molecules at the surface crowd together, forming a strong layer.
Hydrogen Bonding: Three Forms of Water
Water exists as a gas, liquid, or solid, with hydrogen bonds forming or breaking as water changes state.
Gas: Molecules move freely.
Liquid: Hydrogen bonds form and break rapidly.
Solid: Stable hydrogen bonds create a lattice.

Ionic Compounds in Water
Ionic compounds dissociate in water due to the polar nature of water molecules, which interact with ions.
Ionization: Process of breaking ionic bonds as ions interact with water.
Polarity: Water’s asymmetric hydrogen positions create positive and negative ends.

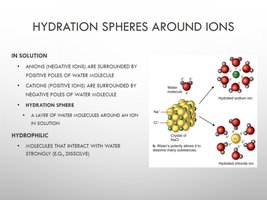
Hydration Spheres Around Ions
Hydration spheres form when water molecules surround ions in solution, facilitating dissolution.
Anions: Surrounded by positive poles of water.
Cations: Surrounded by negative poles of water.
Hydrophilic: Molecules that interact strongly with water.
Hydrophobic Molecules
Hydrophobic molecules do not readily interact with water and are typically nonpolar.
No Hydration Spheres: No attraction to water molecules.
Do Not Dissolve: Remain separate from aqueous solutions.

