 Back
BackBIOL_1233 Exam I Study Guide: Foundations of Biology
Study Guide - Smart Notes

Chapter 1: Themes of Biology & Scientific Inquiry
The Scientific Method
The scientific method is a systematic approach used to investigate natural phenomena and answer scientific questions. It involves forming hypotheses, conducting experiments, and analyzing data.
Hypothesis: A testable statement that explains an observation.
Theory: A well-substantiated explanation based on a body of evidence.
Steps: Observation, Question, Hypothesis, Experiment, Data Collection, Analysis, Conclusion.
Data Types: Qualitative (descriptive) vs. Quantitative (numerical).
Variables: Independent (manipulated) vs. Dependent (measured).
Controls: Experimental controls and control groups ensure reliability.
Chapter 2 & 3: The Chemical Context of Life, Water and Life
The Elements of Life
Living organisms are composed primarily of four essential elements: carbon (C), hydrogen (H), oxygen (O), and nitrogen (N). Trace elements are required in smaller amounts.
Chemical Bonds: Covalent (shared electrons), Ionic (transferred electrons), Hydrogen (weak attraction between H and electronegative atom).
Single vs. Double Bonds: Single bonds share one pair of electrons; double bonds share two pairs.
Electronegativity: The tendency of an atom to attract electrons. Polar bonds result from unequal sharing; nonpolar bonds from equal sharing.
Molecular Shape: The shape of a molecule determines its function.
Water as the Solvent of Life
Water's unique properties make it essential for life, including cohesion, adhesion, high specific heat, expansion upon freezing, and its ability to dissolve a wide range of substances.
Cohesion: Attraction between water molecules.
Adhesion: Attraction between water and other substances.
High Specific Heat: Water resists temperature changes.
Expansion Upon Freezing: Hydrogen bonds cause ice to be less dense than liquid water.
Solvent Diversity: Water forms hydration shells around ions and polar molecules.
Acids and Bases
Acids increase H+ concentration; bases increase OH-. The pH scale measures acidity.
Reversible Reaction:
Buffer: A substance that minimizes changes in pH. Example: blood pH regulation.
Chapter 4 & 5: Carbon and Large Biological Molecules
Carbon Atoms and Molecular Diversity
Carbon's ability to form four covalent bonds allows for diverse molecules, including hydrocarbons and isomers.
Valence Electrons: C (4), H (1), O (6), N (5).
Isomers: Structural, geometric (cis/trans), and enantiomers.
Functional Groups
Functional groups are chemically reactive and determine biomolecule behavior.
Hydroxyl, Carbonyl (ketone/aldehyde), Carboxyl (acidic), Amino (basic), Sulfhydryl (hydrophobic), Phosphate, Methyl (inert).
ATP: Generates energy by reacting with water.
Synthesis and Breakdown of Polymers
Polymers are built from monomers via dehydration synthesis and broken down by hydrolysis. Enzymes catalyze these reactions.
Dehydration Synthesis: Removes water to form bonds.
Hydrolysis: Adds water to break bonds.
Carbohydrates
Carbohydrates are sugars and their polymers. Monosaccharides form polysaccharides via glycosidic linkages.
Storage: Starch (plants), glycogen (animals).
Structural: Cellulose (plants), chitin (fungi/animals).
Fats, Phospholipids, and Steroids
Fats are composed of glycerol and fatty acids, formed by ester bonds. Phospholipids have hydrophilic heads and hydrophobic tails, forming bilayers. Steroids have four carbon rings.
Unsaturated: C=C double bonds, liquid at room temperature.
Saturated: No C=C, solid at room temperature.
Phospholipid Bilayer: Spontaneously forms membranes.
Steroids: Cholesterol, sex hormones.
Proteins
Proteins are polymers of amino acids, each with a unique R group. They perform diverse functions and have four structural levels.
Primary: Amino acid sequence.
Secondary: Alpha helix, beta sheets (H-bonding).
Tertiary: R-group interactions (ionic, covalent, hydrophobic).
Quaternary: Multiple polypeptides.
Functions: Enzymes, transport, receptors, motility.
Nucleic Acids
Nucleic acids are polymers of nucleotides, composed of a nitrogenous base, pentose sugar, and phosphate group. DNA and RNA differ in structure and function.
Pyrimidines: Cytosine, Thymine (DNA), Uracil (RNA).
Purines: Adenine, Guanine.
Central Dogma: DNA → RNA → Protein.
DNA: Double helix, antiparallel strands.
RNA: Single stranded.
Chapter 6: Tour of the Cell
Microscopy
Microscopy allows visualization of cells and their structures. Light microscopy uses visible light; electron microscopy uses electron beams for higher resolution.
Light Microscopy: Up to 1000x magnification; specimens can be alive or stained.
Fluorescence Microscopy: Uses fluorescent tags.
Electron Microscopy: SEM (surface), TEM (internal structures).
Prokaryotes vs. Eukaryotes
All cells share basic components: plasma membrane, ribosomes, cytosol, and DNA. Eukaryotes have membrane-bound organelles; prokaryotes do not.
Prokaryotes: No nucleus, no organelles.
Eukaryotes: Nucleus, organelles.
Organelles of the Eukaryotic Cell
Eukaryotic cells contain specialized organelles for various functions.
Plasma Membrane: Phospholipid bilayer; surface area is important.
Nucleus: Nuclear membrane, lamina, pores, chromosomes.
Endomembrane System: ER (smooth: lipids, detox; rough: glycoproteins), Golgi apparatus (shipping/receiving), lysosomes (hydrolytic enzymes), vacuoles (storage).
Mitochondria: Cellular respiration, ATP generation; matrix contains ribosomes and mtDNA.
Chloroplasts: Photosynthesis; thylakoids, stroma.
Peroxisomes: Metabolic compartment; forms H2O2.
Endosymbiont Theory: Mitochondria and chloroplasts originated from engulfed bacteria.
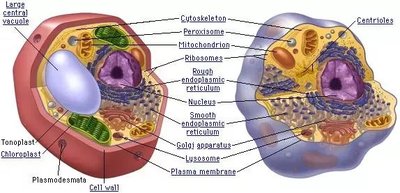
The Cytoskeleton
The cytoskeleton provides structural support and motility, composed of microtubules, microfilaments, and intermediate filaments.
Microtubules: Tubulin; transport, flagella, cilia, centrosome/centrioles.
Microfilaments: Actin; cortex, motility, muscle contraction (myosin).
Intermediate Filaments: Keratin; nuclear lamina, permanent structure.
Extracellular Components
Cells interact with their environment via cell walls (plants/fungi), extracellular matrix (animals), and cell junctions.
Cell Wall: Structure, water balance.
ECM: Collagen, fibronectin, integrins.
Cell Junctions: Plasmodesmata (plants), tight junctions, gap junctions, desmosomes (animals).
Chapter 7: Membrane Structure & Function
Membrane Fluidity
Cell membranes are selectively permeable and composed of amphipathic phospholipids and proteins. The fluid mosaic model describes their dynamic nature.
Cholesterol: Modulates fluidity in animal membranes.
Membrane Proteins and Functions
Membrane proteins serve various functions, including transport, enzymatic activity, signal transduction, cell recognition, joining, and attachment.
Integral: Penetrate bilayer.
Peripheral: Loosely bound to surface.
Aquaporins: Transport water.
Transport Across Membranes
Substances move across membranes via passive (diffusion, osmosis, facilitated diffusion) or active transport (requires energy).
Diffusion: High to low concentration.
Osmosis: Water movement; tonicity (isotonic, hypertonic, hypotonic).
Facilitated Diffusion: Transport proteins assist.
Active Transport: Carrier proteins, sodium-potassium pump, cotransport.
Bulk Transport: Exocytosis, endocytosis.
Chapter 8: An Introduction to Metabolism
Metabolism and Energy
Metabolism encompasses all chemical reactions in cells, divided into catabolic (breakdown) and anabolic (building) pathways. Bioenergetics studies energy flow.
Kinetic Energy: Motion; thermal energy.
Potential Energy: Position/structure; chemical energy.
Laws of Energy Transformation
1st Law: Energy cannot be created or destroyed.
2nd Law: Entropy increases; cells must use energy to maintain order.
Free Energy and Reactions
Free energy () determines whether a reaction is spontaneous.
Equation:
Exergonic: Releases energy, spontaneous (e.g., cellular respiration).
Endergonic: Absorbs energy, non-spontaneous (e.g., photosynthesis).
ATP and Energy Coupling
ATP couples exergonic and endergonic reactions, powering cellular work. It is regenerated from ADP using energy from exergonic reactions.
Enzymes and Regulation
Enzymes lower activation energy, speeding up reactions without being consumed. They are regulated by allosteric, competitive, and feedback inhibition.
Optimal Conditions: pH, temperature.
Regulation: Allosteric (inhibitor binds elsewhere), competitive (inhibitor blocks active site), feedback (product inhibits earlier enzyme).
