 Back
BackBiomolecules, Cell Structure, Membrane Function, and Metabolism: Study Notes for Biology College Students
Study Guide - Smart Notes

The Structure and Function of Large Biological Molecules
Biomolecules: Classes and Organization
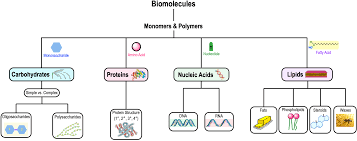
Biomolecules are organic molecules essential to living organisms, classified into four primary groups: carbohydrates, proteins, nucleic acids, and lipids. These molecules are often organized as monomers (single units) and polymers (chains of monomers), except for lipids, which do not form true polymers.
Monomers: Individual building blocks (e.g., glucose, amino acids, nucleotides).
Polymers: Long chains of monomers linked by covalent bonds (e.g., starch, proteins, DNA).
Building and Breaking Down Polymers
Dehydration Synthesis: Forms covalent bonds between monomers, building polymers by removing water.
Hydrolysis: Breaks covalent bonds in polymers by adding water, releasing energy.
Carbohydrates
Carbohydrates are carbon-based molecules with many hydroxyl groups, also known as saccharides. They are classified by size:
Monosaccharides: Single carbohydrate units (e.g., glucose).
Oligosaccharides: 2–20 covalently linked monosaccharides.
Polysaccharides: More than 20 monosaccharides (e.g., starch, cellulose).
Glycosidic bonds link monosaccharides in polysaccharides. Functions:
Structural support: Cellulose in plant cell walls, chitin in exoskeletons.
Energy storage: Starch in plants, glycogen in animals.
Proteins
Proteins are functional molecules made of one or more polypeptides, each folded into a specific three-dimensional structure.
Amino acids: Monomers of proteins, each with a central carbon, hydrogen, amino group, carboxyl group, and unique R-group.
Peptide bonds: Covalent bonds linking adjacent amino acids.
Directionality: Proteins have N-terminal and C-terminal ends.

Classification of Amino Acid Chains
Term | Length of Amino Acid Chain |
|---|---|
Amino acid | Single protein unit or monomer |
Oligopeptide | 2–20 covalently linked amino acids |
Peptide | Less than 50 covalently linked amino acids |
Polypeptide | Greater than 50 covalently linked amino acids |
Protein | One or multiple polypeptide chains in their folded functional forms |

Denatured Proteins & Chaperones
Denatured proteins: Lose their shape and function due to environmental changes (pH, temperature, salt concentration).
Chaperone proteins: Assist in refolding denatured proteins.
Protein misfolding: Implicated in diseases like cystic fibrosis, Alzheimer's, Parkinson's, and mad cow disease.
Nucleic Acids
Nucleic acids (DNA and RNA) are polymers that store and encode genetic information.
Nucleotides: Monomers consisting of a phosphate group, pentose sugar, and nitrogenous base.
Phosphodiester bonds: Covalent bonds linking nucleotides.
Directionality: 5' phosphate end and 3' hydroxyl end.
Nitrogenous bases:
Pyrimidines: Single-ringed (cytosine, thymine, uracil).
Purines: Double-ringed (adenine, guanine).
DNA: Double helix, stores hereditary information, uses deoxyribose sugar, bases: A, T, C, G. RNA: Single-stranded, uses ribose sugar, bases: A, U, C, G.
Lipids
Lipids are hydrophobic biomolecules, highly diverse in structure and function.
Fatty acids: Hydrocarbon chains with a carboxyl group; can be saturated (solid, only single bonds), unsaturated (liquid, double bonds), or trans fats (artificial, unhealthy).
Triglycerides: Three fatty acids linked to glycerol; main fat storage in animals.
Phospholipids: Major component of cell membranes; amphipathic with hydrophilic head and hydrophobic tails.
Steroids: Four fused carbon rings; cholesterol is a key membrane component.
Waxes: Fatty acids bound to long-chain alcohols; protection and water loss prevention.


A Tour of the Cell
Microscopy
Light microscopes: Use light to magnify small objects.
Electron microscopes: Use electrons for higher magnification; SEM for external surfaces, TEM for internal structures.
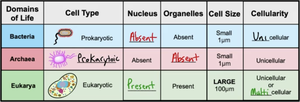
Prokaryotic vs. Eukaryotic Cells
Domains of Life | Cell Type | Nucleus | Organelles | Cell Size | Cellularity |
|---|---|---|---|---|---|
Bacteria | Prokaryotic | Absent | Absent | Small (1 μm) | Unicellular |
Archaea | Prokaryotic | Absent | Absent | Small (1 μm) | Unicellular |
Eukarya | Eukaryotic | Present | Present | Large (100 μm) | Unicellular or Multicellular |
Cellular Organelles
Nucleus: Stores DNA, surrounded by nuclear envelope, contains nucleolus.
Endoplasmic Reticulum (ER): Rough ER (ribosome-coated, protein synthesis), Smooth ER (lipid synthesis).
Golgi Apparatus: Modifies, sorts, and packages proteins.
Lysosomes: Digestive enzymes, found in animal cells.
Peroxisomes: Break down toxic compounds, found in all eukaryotes.
Central Vacuole: Storage and turgor pressure in plant cells.
Energy-Related Organelles
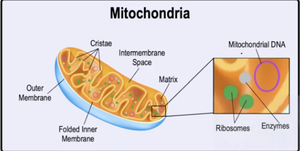
Mitochondria
Mitochondria are the site of cellular respiration, producing ATP from food sources.
Two membranes: outer and folded inner (cristae).
Matrix: contains enzymes, ribosomes, and mitochondrial DNA.
Intermembrane space: region between membranes.
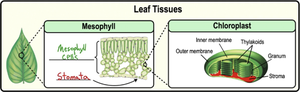
Chloroplasts
Chloroplasts are the site of photosynthesis in plant cells, converting sunlight into sugars.
Two membranes: inner and outer.
Stroma: matrix with DNA, ribosomes, enzymes.
Thylakoids: sacs containing chlorophyll.
Granum: stacks of thylakoids.
Stomata: pores for gas exchange.
Cytoskeleton
The cytoskeleton is a network of proteins providing cell shape, structure, movement, and transport.
Microfilaments (Actin): Smallest, cellular movement/division.
Intermediate Filaments: Variable proteins, structural support.
Microtubules: Largest, transport vesicles.
Cell Junctions
Cell junctions allow direct interaction between neighboring cells.
Junction Type | Description |
|---|---|
Tight | Membrane protein links cells, creating a leak-proof barrier (animal cells). |
Anchoring (Desmosomes) | Intermediate filaments anchor neighboring cells (animal cells). |
Gap | Protein channels connect cytoplasms of two animal cells. |
Plasmodesmata | Gaps in cell walls connect cytoplasms of two plant cells. |

Membrane Structure and Function
Biological Membranes
Biological membranes are phospholipid bilayers with embedded proteins and cholesterol, forming a fluid mosaic.
Phospholipids: Amphipathic, major component.
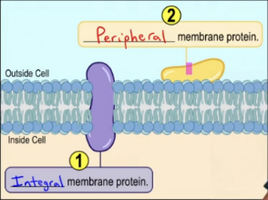
Integral membrane proteins: Span the bilayer.
Peripheral membrane proteins: Located on the membrane's perimeter.
Membrane Protein Functions
Membrane proteins perform diverse functions:
Recognition: Cell identification.
Anchorage: Cytoskeleton and ECM anchoring.
Transduction: Signal molecule receptors.
Transport: Molecular transport across membrane.
Linkage: Connects cells via protein linkage.
Enzymes: Catalyze enzymatic processes.

Membrane Fluidity
Factors affecting membrane fluidity:
Temperature
Saturation of fatty acids
Cholesterol content (fluidity buffer)
Concentration Gradients and Diffusion
Concentration gradient: Difference in substance concentration between two areas.
Diffusion: Movement from high to low concentration (down gradient).

Membrane Transport
Biological membranes are selectively permeable.
Passive transport: No energy required; includes simple and facilitated diffusion.
Active transport: Requires energy (ATP); moves molecules against gradient.
Transport proteins: Uniporters (one molecule, one direction), symporters (multiple molecules, same direction), antiporters (multiple molecules, opposite directions).
Osmosis
Passive diffusion of water across membranes; direction depends on tonicity (hypotonic, isotonic, hypertonic).
Water moves from hypotonic to hypertonic solutions.
Osmoregulation maintains solute and water balance.
Bulk Transport
Exocytosis: Movement out of the cell via vesicle fusion.
Endocytosis: Movement into the cell via vesicle formation; includes phagocytosis (solid), pinocytosis (liquid), receptor-mediated endocytosis (specific).
An Introduction to Metabolism
Metabolism and Metabolic Pathways
Metabolism is the sum of all chemical reactions in an organism, organized into metabolic pathways.
Catabolic pathways: Release energy by breaking down complex molecules (e.g., cellular respiration).
Anabolic pathways: Use energy to build complex molecules (e.g., photosynthesis, protein synthesis).
Energy and Thermodynamics
Potential energy: Stored energy (e.g., chemical bonds).
Kinetic energy: Energy of motion.
First Law: Energy cannot be created or destroyed, only transformed.
Second Law: Energy conversions increase entropy (disorder).
Chemical Reactions
Endergonic reactions: Require energy input (nonspontaneous).
Exergonic reactions: Release energy (spontaneous).
Free energy (G): Portion of a system's energy available to do work.
Change in free energy () determines spontaneity:
ATP: Cellular Energy Currency
ATP (adenosine triphosphate) powers cellular activity.
Three components: chain of three phosphate groups, pentose sugar, adenine base.
ATP hydrolysis releases energy, forming ADP and inorganic phosphate.
Energy coupling: Exergonic reactions drive endergonic reactions via ATP hydrolysis.
Phosphorylation: Transfer of phosphate group to another molecule, activating it.
Enzymes
Enzymes are biological catalysts that speed up reactions without being consumed.
Substrates: Reactants catalyzed by enzymes.
Activation energy (): Minimum energy required to start a reaction; enzymes lower .
Enzyme-substrate complex: Substrate binds at active site.
Cofactors: Non-protein substances required for catalysis (e.g., metal ions, coenzymes).
Enzyme Inhibition
Competitive inhibitors: Compete for active site, block substrate binding.
Noncompetitive inhibitors: Bind at allosteric site, alter enzyme function.
Metabolic Pathways and Feedback
Catabolic pathways: Break down molecules, release energy.
Anabolic pathways: Build up molecules, require energy.
Negative feedback: Final product inhibits earlier step, decreases production.
Positive feedback: Final product stimulates earlier step, increases production.
Additional info: Genomics and proteomics are fields studying the structure, function, and mapping of genomes and proteomes, respectively.
