 Back
BackCarbohydrates: Structure, Function, and Biological Roles
Study Guide - Smart Notes

Carbohydrates
Overview of Carbohydrates
Carbohydrates are essential biomolecules that serve as fuel, building material, components of the extracellular matrix (ECM), and play significant roles in disease and cell interactions. They include simple sugars and their polymers, ranging from monomers to complex polysaccharides.
Monomers: Single sugar units (monosaccharides).
Oligosaccharides: Short chains of 3–9 monosaccharide units.
Polysaccharides: Long chains of monosaccharide units, forming macromolecules.
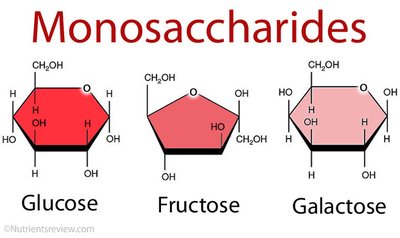
Monosaccharides
Monosaccharides are the simplest carbohydrates, typically with molecular formulas that are multiples of CH2O. Glucose (C6H12O6) is the most common example. They are classified based on the location of the carbonyl group (aldose or ketose) and the number of carbon atoms in their skeleton.
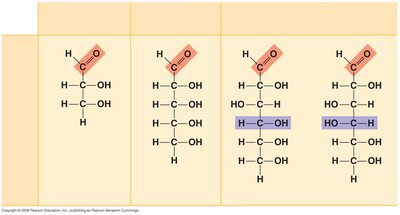
Aldoses: Monosaccharides with an aldehyde group (e.g., glucose, galactose, ribose).
Ketoses: Monosaccharides with a ketone group (e.g., fructose, ribulose).
Classification by carbon number: Trioses (3C), Pentoses (5C), Hexoses (6C).
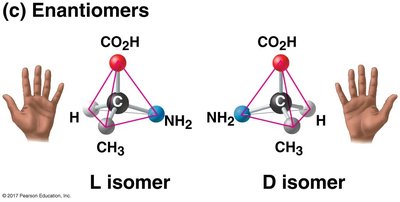
Chirality: Many monosaccharides have chiral centers, leading to isomerism.
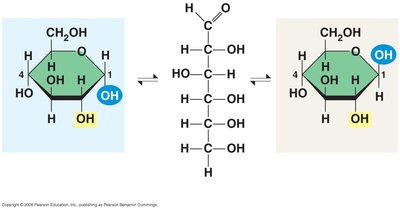
Structure and Isomerism of Monosaccharides
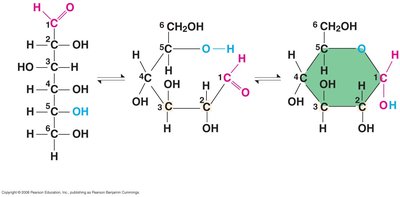
Monosaccharides can exist in linear or ring forms, especially in aqueous solutions. The ring formation is a result of intramolecular reactions between the carbonyl and hydroxyl groups.
Linear form: Shows the open-chain structure.
Ring form: Most common in biological systems.
Isomerism: Includes structural isomers and enantiomers (mirror-image forms).
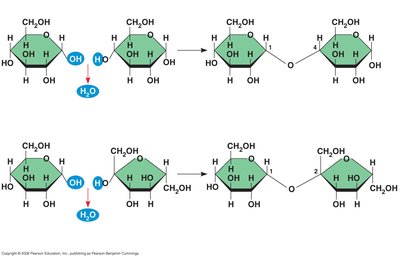
Disaccharides
Disaccharides are formed by joining two monosaccharides via a dehydration reaction, resulting in a covalent bond called a glycosidic linkage. Common examples include sucrose (glucose + fructose) and maltose (glucose + glucose).
Dehydration reaction: Removes a water molecule to form the bond.
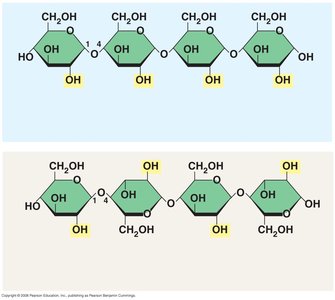
Glycosidic linkage: The specific carbon atoms involved determine the type (e.g., 1–4 or 1–2 linkage).
Polysaccharides
Polysaccharides are polymers of sugars with storage and structural roles. Their properties depend on the types of sugar monomers and the positions of glycosidic linkages.
Storage polysaccharides: Starch (plants) and glycogen (animals).
Structural polysaccharides: Cellulose (plants), chitin (arthropods and fungi).
Storage Polysaccharides
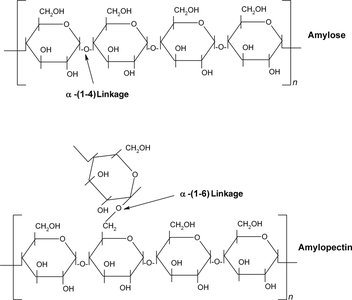
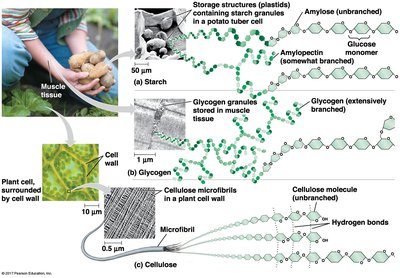
Starch is a plant storage polysaccharide composed entirely of glucose monomers. It is stored as granules in chloroplasts and other plastids. Starch exists in two forms: amylose (unbranched, helical) and amylopectin (branched).
Amylose: Linear, forms a helical structure due to α(1–4) linkages.
Amylopectin: Branched, contains α(1–6) linkages at branch points.

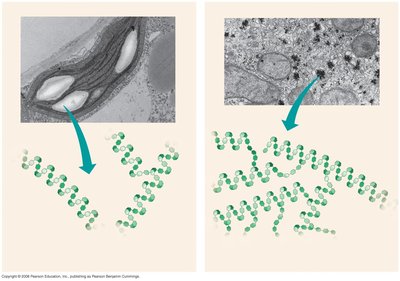
Glycogen is the storage polysaccharide in animals, stored mainly in liver and muscle cells. It is highly branched, allowing rapid mobilization of glucose.
Structural Polysaccharides
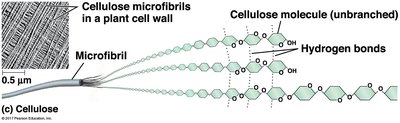
Cellulose is a major component of plant cell walls. It is a polymer of glucose, but differs from starch in the type of glycosidic linkage (β instead of α). Cellulose molecules are straight and form microfibrils through hydrogen bonding, providing structural strength.
α-glucose: Forms helical polymers (starch).
β-glucose: Forms straight polymers (cellulose).
Microfibrils: Bundles of cellulose molecules held together by hydrogen bonds.
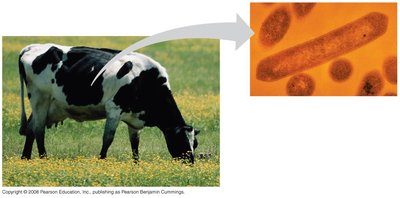
Enzymes that digest starch (α linkages) cannot hydrolyze β linkages in cellulose. Thus, cellulose acts as insoluble fiber in human diets, but some microbes can digest it, enabling herbivores to utilize cellulose through symbiotic relationships.
Chitin is a structural polysaccharide found in the exoskeleton of arthropods and cell walls of fungi. It is composed of N-acetylglucosamine units and forms strong, flexible materials.
Applications: Used in surgical threads that decompose naturally.

Other Carbohydrate Molecules
Peptidoglycan is a structural carbohydrate in bacterial cell walls, consisting of a carbohydrate backbone linked by peptides.

Proteoglycans are complexes of proteins and carbohydrates, important in the extracellular matrix and cell interactions.
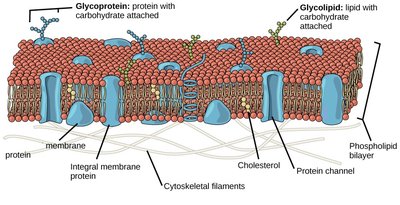
Role of Sugars in Cell Interactions and Disease
Sugars are involved in cell–cell interactions and the extracellular matrix. They modify proteins and lipids on cell surfaces, affecting processes such as immune function and cell recognition. These modifications are carried out in the endoplasmic reticulum and Golgi apparatus, and broken down in lysosomes. Sugars play roles in diseases ranging from viral infections to cancer progression.
Glycoproteins: Proteins with carbohydrate attachments.
Glycolipids: Lipids with carbohydrate attachments.
Cell recognition: Cells appear as "sugar-coated," facilitating communication and immune responses.
Summary Table: Types of Carbohydrates
Type | Structure | Function | Example |
|---|---|---|---|
Monosaccharide | Single sugar unit | Fuel, building block | Glucose |
Disaccharide | Two sugar units | Transport, energy | Sucrose |
Polysaccharide | Many sugar units | Storage, structure | Starch, cellulose, glycogen, chitin |
Proteoglycan | Protein + carbohydrate | ECM, cell signaling | Proteoglycan complex |
Peptidoglycan | Carbohydrate + peptide | Bacterial cell wall | Peptidoglycan |
Key Equations
General formula for monosaccharides:
Dehydration reaction:
Additional info:
Chirality and isomerism are crucial for biological specificity in carbohydrate function.
Cellulose's β linkages make it indigestible for most animals, but symbiotic microbes enable digestion in herbivores.
Chitin's unique properties make it valuable in medical applications.
