Back
BackCarbon and the Molecular Diversity of Life
Study Guide - Smart Notes

Carbon: The Backbone of Life
Importance of Carbon in Biological Molecules
Carbon is the foundational element in organic chemistry and biology, forming the backbone of the vast majority of molecules essential for life. Its unique ability to form four covalent bonds allows for the construction of large, complex, and diverse molecules, including those that make up living organisms.
Organic compounds are defined as molecules containing carbon-hydrogen bonds.
Carbon's versatility enables the formation of chains, rings, and branching structures, supporting molecular diversity.
Are there other elements capable of forming 4 covalent bonds? While silicon can also form four covalent bonds, carbon's smaller size and ability to form stable double and triple bonds make it uniquely suited for the chemistry of life.
Carbon Chains and Skeletons
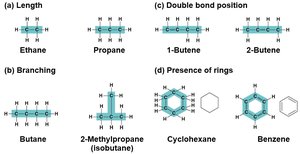
Carbon chains form the skeletons of most organic molecules. These chains can vary in length, branching, double bond position, and the presence of rings, contributing to the diversity of organic compounds.
Length: Chains can be short or long.
Branching: Chains may be straight or branched.
Double bond position: Double bonds can occur at different locations.
Rings: Chains can form closed ring structures.
Hydrocarbons
Definition and Biological Importance
Hydrocarbons are organic molecules consisting entirely of carbon and hydrogen. They are hydrophobic and can store significant amounts of energy, making them important components of fats and other biological molecules.
Hydrocarbons are found in many biological molecules, such as fatty acids.
They can undergo reactions that release large amounts of energy, important for metabolism.
Isomers: Molecular Diversity
Types of Isomers
Isomers are compounds with the same molecular formula but different structures and properties. The three main types are:
Structural isomers: Differ in the covalent arrangement of atoms.
Cis-trans isomers: Differ in spatial arrangement due to inflexible double bonds.
Enantiomers: Mirror images due to an asymmetric carbon.
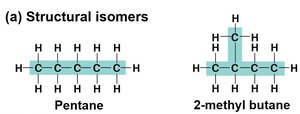
Structural Isomers
Structural isomers have different covalent arrangements of their atoms. The number of possible isomers increases with the size of the carbon skeleton.
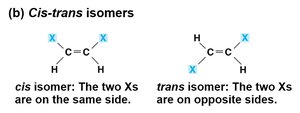
Cis-trans Isomers
In cis-trans isomers, atoms are connected in the same order but differ in spatial arrangement around a double bond. The rigidity of the double bond prevents rotation, leading to distinct isomers with different properties.
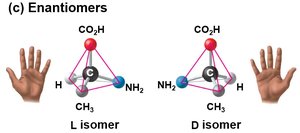
Enantiomers
Enantiomers are isomers that are non-superimposable mirror images of each other, typically due to the presence of an asymmetric (chiral) carbon. Usually, only one enantiomer is biologically active.
Example: Identifying Isomer Types
Given a pair of molecules, determine if they are structural isomers, cis-trans isomers, or enantiomers by analyzing their connectivity and spatial arrangement.

Functional Groups
Definition and Importance
Functional groups are specific groups of atoms attached to the carbon skeleton of organic molecules that participate in chemical reactions in characteristic ways. The properties of organic molecules depend on both the arrangement of the carbon skeleton and the functional groups attached.
Functional groups can replace hydrogens on the carbon skeleton.
They determine the reactivity and function of organic molecules.
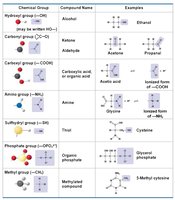
The Seven Most Important Functional Groups in Biology
Hydroxyl group (–OH)
Carbonyl group (C=O)
Carboxyl group (–COOH)
Amino group (–NH2)
Sulfhydryl group (–SH)
Phosphate group (–OPO32–)
Methyl group (–CH3)
Example: Identifying Functional Groups
Given a molecular structure, identify the functional group present (e.g., methyl, carbonyl, carboxyl, phosphate).

ATP: The Energy Currency of the Cell
Structure and Function
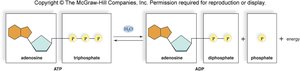
Adenosine triphosphate (ATP) is the primary energy carrier in cells. Energy is stored in the bonds between phosphate groups and is released when these bonds are broken, typically by hydrolysis.
ATP consists of adenosine (adenine + ribose) and three phosphate groups.
Breaking a phosphate bond releases energy for cellular work.
Macromolecules: Polymers and Monomers
Definition and Types
Macromolecules are large organic molecules composed of smaller subunits called monomers. The four major classes are carbohydrates, lipids, proteins, and nucleic acids.
Polymers are long molecules made of repeating monomers.
Some monomers have functions beyond serving as building blocks.
Polymerization and Depolymerization
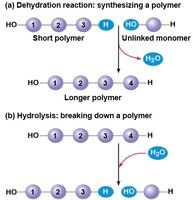
Cells build and break down polymers using dehydration and hydrolysis reactions, respectively. Enzymes catalyze these processes.
Dehydration reaction: Joins two monomers by removing a water molecule.
Hydrolysis: Breaks a polymer into monomers by adding water.
Carbohydrates
Overview and Classification
Carbohydrates include sugars and their polymers. They serve as fuel and building material in cells.
Monosaccharides: Simple sugars (e.g., glucose, C6H12O6).
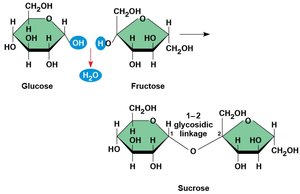
Disaccharides: Two monosaccharides joined by a glycosidic linkage.
Polysaccharides: Polymers of many monosaccharides with storage or structural roles.
Monosaccharides
Monosaccharides are classified by the number of carbons and the position of the carbonyl group. They serve as fuel for cellular processes.
Disaccharides
Disaccharides are formed by dehydration reactions between two monosaccharides, creating a glycosidic bond.
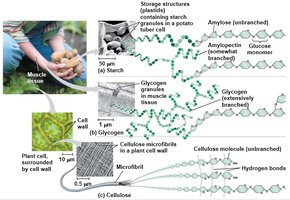
Polysaccharides
Polysaccharides serve as storage (e.g., starch in plants, glycogen in animals) or structural (e.g., cellulose in plants, chitin in fungi and arthropods) molecules. Their properties depend on the types of monomers and the linkages between them.
Proteins
Structure and Function
Proteins are polymers of amino acids and account for more than half the dry mass of most cells. They perform a wide variety of functions, including catalysis, defense, transport, support, movement, and regulation.
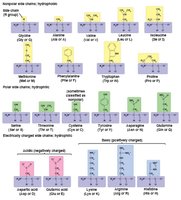
Amino acids: Organic molecules with amino and carboxyl groups, differing in their side chains (R groups).
Polypeptides: Chains of amino acids linked by peptide bonds.
Levels of Protein Structure
Primary structure: Linear sequence of amino acids.
Secondary structure: Local folding into α-helices and β-pleated sheets due to hydrogen bonding.
Tertiary structure: Overall 3D shape due to interactions among side chains.
Quaternary structure: Association of multiple polypeptide chains.
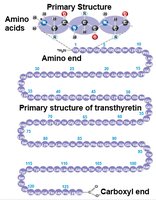
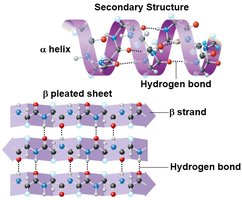
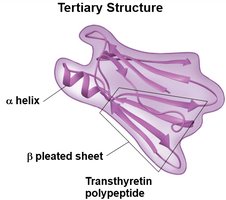
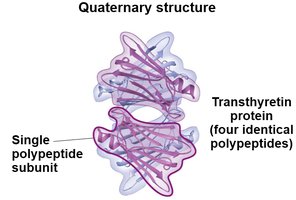
Protein Denaturation
Physical and chemical conditions such as pH, salt concentration, and temperature can cause proteins to lose their native structure, a process called denaturation. Denatured proteins are biologically inactive.
Nucleic Acids
Structure and Function
Nucleic acids (DNA and RNA) are polymers of nucleotides that store and transmit genetic information and direct protein synthesis.
Nucleotide: Composed of a phosphate group, a five-carbon sugar, and a nitrogenous base.
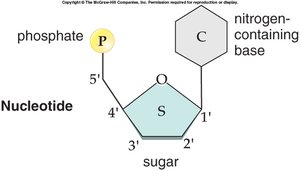
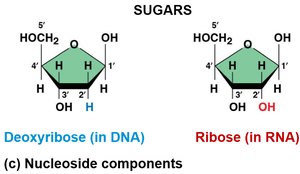
Nitrogenous Bases
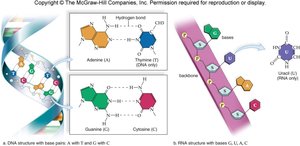
Pyrimidines: Cytosine (C), Thymine (T), Uracil (U) – single ring
Purines: Adenine (A), Guanine (G) – double ring
Base pairing: In DNA, A pairs with T, G with C; in RNA, A pairs with U, G with C.
Nucleotide Polymers
Nucleotides are joined by covalent bonds between the 3' hydroxyl of one sugar and the 5' phosphate of the next, forming a sugar-phosphate backbone. DNA is double-stranded (double helix), while RNA is usually single-stranded.
Lipids
Overview and Types
Lipids are hydrophobic molecules that do not form true polymers. The main types are fats, phospholipids, and steroids.
Lipids are hydrophobic due to their hydrocarbon content.
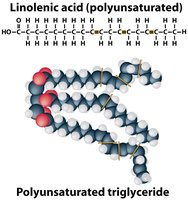
Fats (Triglycerides)
Fats are composed of a glycerol molecule and three fatty acids. Their main function is energy storage.

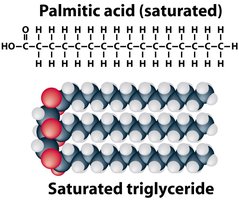
Saturated vs. Unsaturated Fats
Saturated fats: No double bonds, straight chains, solid at room temperature, usually animal origin.
Unsaturated fats: One or more double bonds, kinked chains, liquid at room temperature, usually plant or fish origin.
Phospholipids
Phospholipids are similar to fats but have a phosphate group replacing one fatty acid. They are the primary component of cell membranes, forming bilayers in aqueous environments due to their amphipathic nature (hydrophilic head, hydrophobic tails).
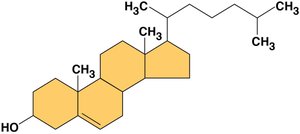
Steroids
Steroids are lipids with a structure of four fused carbon rings. Cholesterol is a key steroid in animal cell membranes. Despite their different structure from fats and phospholipids, steroids are classified as lipids due to their hydrophobic nature.