 Back
BackCarbon and the Molecular Diversity of Life: Structure, Bonding, and Functional Groups
Study Guide - Smart Notes

Carbon and the Molecular Diversity of Life
Introduction
Carbon is the foundational element of organic chemistry and the molecular diversity of life. Its unique bonding properties allow for the formation of a vast array of complex molecules essential for biological structure and function. This chapter explores the origin of organic compounds, the bonding behavior of carbon, the concept of isomerism, and the significance of functional groups in organic molecules.
Organic Compounds and Their Origin
Definition and Historical Context
Organic Chemistry is the study of carbon-containing compounds, ranging from simple molecules like methane to complex macromolecules such as proteins.
Organic Compounds are defined as molecules containing carbon atoms, typically bonded to hydrogen, oxygen, nitrogen, or other elements.
Historically, it was believed that organic compounds could only be produced by living organisms, but the synthesis of urea in the 19th century disproved this notion.

Origin of Organic Compounds
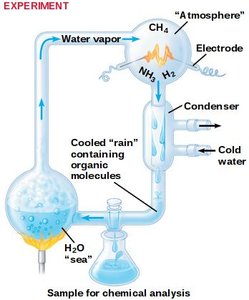
Organic molecules can arise from inorganic precursors under certain conditions, as demonstrated by the Stanley Miller experiment (1953).
This experiment simulated early Earth conditions and produced amino acids and other organic molecules from simple gases and electrical sparks.
Carbon Bonding and Molecular Structure
Valence and Bonding Properties
Carbon has an atomic number of 6, with 4 valence electrons, allowing it to form up to four covalent bonds with other atoms.
This tetravalency enables carbon to act as a versatile backbone for organic molecules, forming single, double, or triple bonds.

Shapes of Carbon Compounds
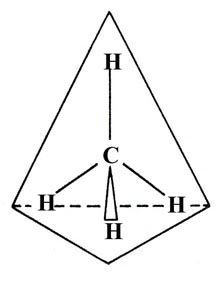
When carbon forms four single bonds (e.g., methane, CH4), the molecule adopts a tetrahedral geometry.
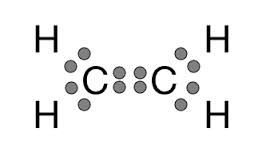
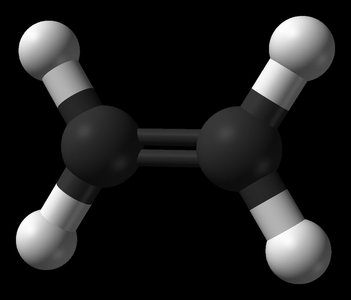
Double bonds (e.g., ethene, C2H4) result in a planar structure.
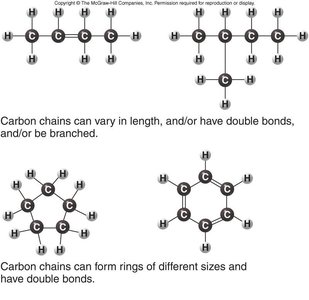
Carbon Backbones
Carbon chains can vary in length, branching, and the presence of rings or double bonds, contributing to molecular diversity.
These variations form the basis for the structural complexity of organic molecules.
Hydrocarbons
Definition and Properties
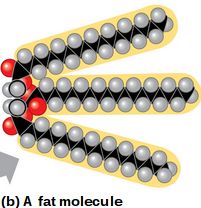
Hydrocarbons are organic molecules consisting entirely of carbon and hydrogen.
They are hydrophobic due to nonpolar C-H bonds and serve as major components of fossil fuels and biological molecules like fats.
Isomers
Types of Isomers
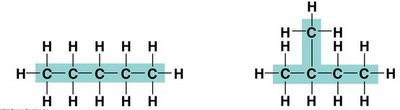
Isomers are compounds with the same molecular formula but different structures and properties.
Structural Isomers: Differ in the covalent arrangement of atoms (e.g., pentane vs. 2-methylbutane).
Cis-Trans Isomers: Differ in spatial arrangement around a double bond.
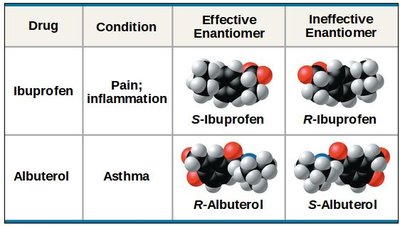
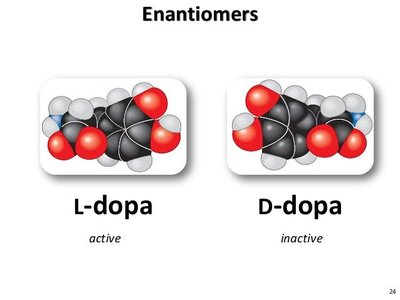
Enantiomers: Mirror images due to an asymmetric carbon atom bonded to four different groups.

Biological Importance of Isomers
Enantiomers can have drastically different biological activities (e.g., L-dopa is active in Parkinson's treatment, D-dopa is inactive).
Chemical (Functional) Groups
Overview
Functional groups are specific groups of atoms attached to the carbon backbone that confer distinct chemical properties and reactivity.
They are critical in determining the behavior of organic molecules in biological systems.
Major Functional Groups
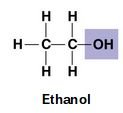
Hydroxyl (-OH): Polar, forms hydrogen bonds, increases solubility (e.g., ethanol).
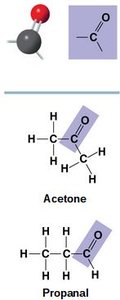
Carbonyl (C=O): Polar, found in ketones (within carbon skeleton) and aldehydes (at the end).
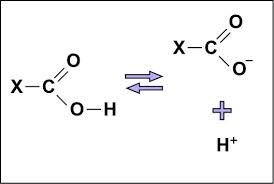
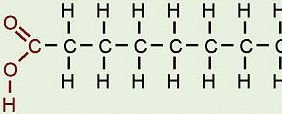
Carboxyl (-COOH): Acts as an acid, can donate H+, found in carboxylic acids.
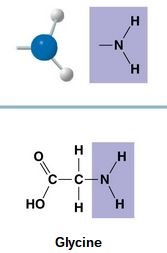
Amino (-NH2): Acts as a base, can accept H+, found in amino acids.
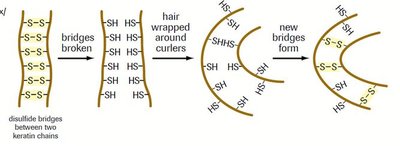
Sulfhydryl (-SH): Forms disulfide bonds, important in protein structure (e.g., cysteine).
Phosphate (-PO42-): Contributes negative charge, involved in energy transfer (e.g., ATP).
Methyl (-CH3): Nonpolar, affects gene expression and molecular recognition.
Functional Groups and Biological Function
Functional groups determine the chemical reactivity and interactions of organic molecules, influencing processes such as enzyme activity, hormone function, and genetic regulation.
For example, the difference between estradiol and testosterone is due to the presence of different functional groups.
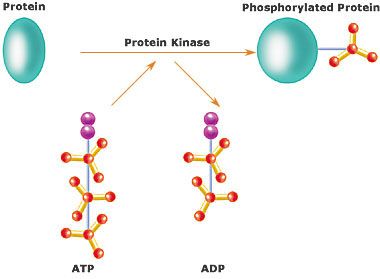
ATP: The Energy Currency of the Cell
Structure and Function
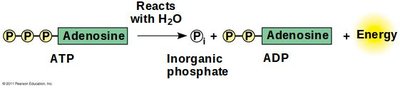
Adenosine Triphosphate (ATP) consists of adenosine attached to three phosphate groups.
Hydrolysis of ATP releases energy by removing a phosphate group, forming ADP (adenosine diphosphate).
Summary Table: Functional Groups
Group | Structure | Properties | Example |
|---|---|---|---|
Hydroxyl | -OH | Polar, forms H-bonds | Ethanol |
Carbonyl | C=O | Polar, reactive | Acetone, Propanal |
Carboxyl | -COOH | Acidic, donates H+ | Acetic acid |
Amino | -NH2 | Basic, accepts H+ | Glycine |
Sulfhydryl | -SH | Forms disulfide bonds | Cysteine |
Phosphate | -PO42- | Negative charge, energy transfer | ATP |
Methyl | -CH3 | Nonpolar, gene regulation | 5-Methyl cytidine |
Key Equations
ATP Hydrolysis:
Conclusion
Understanding the bonding properties of carbon, the diversity of isomers, and the role of functional groups is essential for grasping the molecular basis of life. These concepts form the foundation for further study in biochemistry, molecular biology, and related fields.
